Abstract
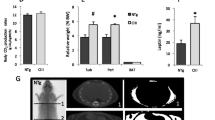
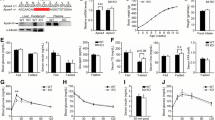
The levels of plasma apolipoprotein (apo) E, an anti-atherogenic protein involved in mammalian cholesterol transport, were found to be 2-3 fold lower in mice over-expressing human apoA-I gene. ApoE is mainly associated with VLDL and HDL-size particles, but in mice the majority of the apoE is associated with the HDL particles. Over-expression of the human apoA-I in mice increases the levels of human apoA-I-rich HDL particles by displacing mouse apoA-I from HDL. This results in lowering of plasma levels of mouse apoA-I. Since plasma levels of apoE also decreased in the apoA-I transgenic mice, the mechanism of apoE lowering was investigated. Although plasma levels of apoE decreased by 2-3 fold, apoB levels remained unchanged. As expected, the plasma levels of human apoA-I were almost 5-fold higher in the apoAI-Tg mice compared to mouse apoA-I in WT mice. If the over-expression of human apoA-I caused displacement of apoE from the HDL, the levels of hepatic apoE mRNA should remain the same in WT and the apoAI-Tg mice. However, the measurements of apoE mRNA in the liver showed 3-fold decreases of apoE mRNA in apoAI-Tg mice as compared to WT mice, suggesting that the decreased apoE mRNA expression, but not the displacement of the apoE from HDL, resulted in the lowering of plasma apoE in apoAI-Tg mice. As expected, the levels of hepatic apoA-I mRNA (transgene) were 5-fold higher in the apoAI-Tg mice. ApoE synthesis measured in hepatocytes also showed lower synthesis of apoE in the apoAI-Tg mice. These studies suggest that the integration of human apoA-I transgene in mouse genome occurred at a site that affected apoE gene expression. Identification of this locus may provide further understanding of the apoE gene expression.
Similar content being viewed by others
References
Shimano H, Yamada N, Katsuki M, Shimada M, Gotoda T, Harada K, Murase T, Fukazawa C, Takaku F, Yazaki Y: Overexpression of apolipoprotein E in transgenic mice: Marked reduction in plasma lipoproteins except high density lipoproteins and resistance against diet-induced hypercholesterolemia. Proc Natl Acad Sci USA 89: 1750-1754, 1992
Smith JD, Plump AS, Hayek T, Walsh A, Breslow JL: Accumulation of human apolipoprotein E in the plasma of transgenic mice. J Biol Chem 265: 14709-14712, 1990
Rubin EM, Krauss RM, Spangler EA, Verstuyft JG, Clift SM: Inhibition of early atherogenesis in transgenic mice by human apolipoprotein AI. Nature 353: 265-267, 1991
Walsh A, Ito Y, Breslow J: High levels of human apolipoprotein A-I in transgenic mice results in increased plasma levels of small high density lipoprotein (HDL) particles comparable to human HDL3. J Biol Chem 264: 6488-6494, 1989
Innerarity TL, Mahley RW: Enhanced binding of cultured human fibroblasts of apoE-containing lipoproteins as compared with low density lipoproteins. Biochemistry 17: 1440-1447, 1978
Zhang W, Gaynor PM, Kruth HS: Apolipoprotein E produced by human monocyte-derived macrophages mediates cholesterol efflux that occurs in the absence of added cholesterol acceptors. J Biol Chem 271: 28641-28646, 1996
Zhang S, Reddick RL, Piedrahita JA, Maeda N: Spontaneous hypercholesterolemia and arterial lesions in mice lacking apolipoprotein E. Science 258: 468-471, 1992
Plump AS, Smith JD, Hayek T, Aalto-Setala K, Walsh A, Verstuyft JG, Rubin EM, Breslow JL: Severe hypercholesterolemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell 71: 343-353, 1992
Sorci-Thomas M, Prack MM, Dashti N, Johnson F, Rudel LL, Williams DL: Apolipoprotein (apo) A-I production and mRNA abundance explain plasma apoA-I and high density lipoprotein differences between two non-human primate species with high and low susceptibilities to diet-induced hypercholesterolemia. J Biol Chem 263: 5183-5189, 1988
Miller GJ, Miller NE: Plasma high density lipoproteins and the development of ischaemic heart disease. Lancet I: 16-19, 1975
Srivastava RAK, Tang J, Krul ES, Pfleger B, Kitchens RT, Schonfeld G: Dietary fatty acids and dietary cholesterol differ in their effects on the in vivo regulation of apoA-I and A-II gene expression. Biochim Biophys Acta 1125: 251-261, 1992
Srivastava RAK, Baumann D, Schonfeld G: Regulation of low density lipoprotein receptor by estrogen differs in rat and mouse. Eur J Biochem 216: 527-538.
Strobl W, Chan L, Patsch W: Differential regulation of hepatic apolipoprotein A-I and A-II gene expression by thyroid hormone in rat liver. Atherosclerosis 97: 161-170, 1993
Paszty C, Maeda N, Verstuyft JG, Rubin EM: Apolipoprotein A-I transgene corrects apolipoprtoein E deficiency-induced atherosclerosis in mice. J Clin Invest 94: 899-903, 1994
Kronenberg F, Steinmetz A, Kostner GM, Dieplinger H: Lipoprotein[a] in health and disease. Crit Rev Clin Lab Sci 33: 495-543, 1996
Liu AC, Lawn RM, Verstuyft JG, Rubin EM: Human apolipoprotein A-I prevents atherosclerosis associated with apolipoprotein[a] in transgenic mice. J Lipid Res 35: 2263-2267, 1994
deSilva HV, Mas-Oliva J, Taylor JM, Mahley RW: Identification of apolipoprotein B-100 low density lipoproteins, apolipoprotein B-48 remnants, and apolipoprotein-rich high density lipoproteins in the mouse. J Lipid Res 35: 1297-1310, 1994
Srivastava RAK, Jiao S, Tang J, Pfleger B, Kitchens RT, Schonfeld G: In vivo regulation of low density lipoprotein receptor and apolipoprotein B gene expression by dietary fat and cholesterol in inbred strains of mice. Biochim Biophys Acta 1086: 29-43, 1991
Srivastava RAK, Srivastava N, Schonfeld G: Expression of low density lipoprotein receptor, apolipoprotein A-I, apolipoprotein A-II, and apolipoprotein A-IV in various rat organs utilizing an efficient and rapid method of RNA isolation. Biochem Intern 27: 85-95, 1992
Srivastava RAK: Using riboprobes for Northern blotting analysis. Meth Mol Biol 86: 103-112, 1998
Srivastava RAK, Pfleger B, Schonfeld G: Expression of low density lipoprotein receptor, apolipoprotein B, apolipoprotein A-I, and apolipoprotein A-IV messenger RNA in various mouse organs as determined by a novel RNA-excess solution hybridization assay. Biochim Biophys Acta 1090: 95-101, 1991
Srivastava RAK: Regulation of the apolipoprotein E by dietary lipids occurs by transcriptional and posttranscriptional mechanisms. Mol Cell Biochem 155: 153-162, 1996
Srivastava RAK, Srivastava N, Averna A, Lin RC, Korach K, Lubahn D, Schonfeld G: Estrogen upregulates apolipoprotein E (apoE) gene expression by increasing apoE mRNA in the translating pool via the estrogen receptor a-mediated pathway. J Biol Chem 272: 33360-33366, 1997
Srivastava RAK: Dietary saturated fat, but not cholesterol, regulates apolipoprotein A-I gene expression by posttranscriptional mechanism. Biochem Mol Biol Intern 34: 393-402, 1994
Azrolan N, Odaka H, Breslow JL, Fischer EA: Dietary fat elevates hepatic apoA-I production by increasing the fraction of apolipoprotein A-I mRNA in the translating pool. J Biol Chem 270: 19833-19838, 1995
Williamson R, Lee D, Hagaman J, Maeda N: Marked reduction of high density lipoprotein cholesterol in mice genetically modified to lack apolipoprotein A-I. Proc Natl Acad USA 89: 7134-7138, 1992
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Srivastava, R.A.K. Apolipoprotein E gene expression is reduced in apolipoprotein A-I transgenic mice. Mol Cell Biochem 209, 125–129 (2000). https://doi.org/10.1023/A:1007107712725
Issue Date:
DOI: https://doi.org/10.1023/A:1007107712725