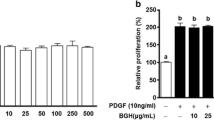
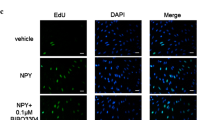
Abstract
Arterial injury-induced vascular smooth muscle cell (VSMC) proliferation in intima is the important etiologic factor in vascular proliferative disorders such as atherosclerosis, hypertension and restenosis after balloon angioplasty. Butyrate, a naturally occurring short chain fatty acid, is produced by bacterial fermentation of dietary fiber and by mammary glands of certain mammals. Studies have shown that butyrate at millimolar concentrations, which are physiological, induces growth arrest, differentiation and apoptosis. We examined the effect of physiological concentrations of butyrate on rat VSMC proliferation and proliferation-induced PCNA expression to determine anti-atherogenic potential of butyrate. Butyrate concentrations, closer to physiological range, exhibited antiproliferative effects on both serum-induced proliferation of serum-starved quiescent VSMCs and actively proliferating non-confluent VSMCs. Treatment of serum-starved quiescent VSMCs with 1-8 mmol/l concentration of butyrate caused a concentration-dependent decrease in serum-induced VSMC proliferation and cell proliferation-associated increase in total cellular proteins and RNA levels. Similarly, exposure of actively growing VSMCs to 5 mmol/l butyrate resulted in the inhibition of cell proliferation and proliferation-induced increase in cellular proteins and RNA levels. Furthermore, cellular morphology was significantly altered. Analysis of cell cycle regulatory proteins indicated that levels of PCNA, an excellent marker for cell proliferation, was significantly altered by butyrate both in actively proliferating and serum-induced quiescent VSMCs. These observations suggest that butyrate exhibits potential antiatherogenic capability by inhibiting VSMC proliferation and proliferation-associated increase in PCNA expression and thus merits further investigations regarding therapeutic significance of butyrate in vascular proliferative disorders.
Similar content being viewed by others
References
Ross R: The pathogenesis of atherosclerosis: A perspective for the 1990's. Nature 362: 801–809, 1993
Schwartz SM, deBlois D, O'Brien ERM: The intima: Soil for atherosclerosis and restenosis. Circ Res 77: 445–465, 1995
Ferns GAA, Raines EW, Sprugel KH, Motani AS, Reidy MA, Ross R: Inhibition of neointimal smooth muscle accumulation after angioplasty by an antibody to PDGF. Science 253: 1129–1132, 1991
Lindner V, Reidy MA: Proliferation of smooth muscle cells after vascular injury is inhibited by an antibody against basic fibroblast growth factor. Proc Natl Acad Sci USA 88: 3739–3743, 1991
Casscells W, Lappi D, Olwin B, Wai C, Siegman M, Speir EH, Sasse J, Baird A: Elimination of smooth muscle cells in experimental restenosis: Targeting of fibroblast growth factor receptors. Proc Natl Acad Sci USA 89: 7159–7163, 1992
Pastore CJ, Isner JM, Bacha PA, Kearney M, Pickering JG: Epidermal growth factor receptor-targeted cytotoxin inhibits neointimal hyperplasia in vivo. Results of local versus systemic administration. Circ Res 77: 519–529, 1995
Simons M, Edelman ER, Rosenberg RD: Antisense proliferating cell nuclear antigen oligonucleotides inhibit intimal hyperplasia in a rat carotid artery injury model. J Clin Invest 93: 2351–2356, 1994
Simons M, Edelman ER, DeKeyser JL, Langer RS, Rosenberg RD: Antisense c-myb oligonucleotides inhibit intimal arterial smooth muscle cell accumulation in vivo. Nature 359: 67–70, 1992
The Multicenter European Research Trial with Cilazapril after angioplasty to prevent transluminal coronary obstruction and restenosis (Mercator) study group. Does the new angiotensin converting enzyme inhibitor cilazapril prevent restenosis after percutaneous transluminal coronary angioplasty? Circulation 86: 100–110, 1992
Clowes AW, Karnovsky MJ: Suppression by heparin of smooth muscle cell proliferation in injured arteries. Nature 265: 625–626, 1977
Guyton JR, Rosenberg RD, Clowes AW, Karnovsky MJ: Inhibition of rat arterial cell proliferation by heparin. In vivo studies with anticoagulant and non-anticoagulant heparin. Circ Res 46: 625–634, 1980
Cummings JH, Pomare EW, Branch WJ, Naylor CPE, MacFarlane GT: Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 28: 1221–1227, 1987
Smith JG, German JB: Molecular and genetic effects of dietary derived butyric acid. Food Technol 49: 87–90, 1995
Prasad KN: Butyric acid: A small fatty acid with diverse biological functions. Life Sci 27: 1351–1358, 1980
Kruh J: Effects of sodium butyrate, a new pharmacological agent, on cells in culture. Mol Cell Biol 42: 65–82, 1982
Ranganna K, Joshi T, Yatsu FM: Sodium butyrate inhibits plateletderived growth factor-induced proliferation of vascular smooth muscle cells. Atheroscler Thromb Vasc Biol 15: 2273–2283, 1995
Boffa LC, Lupton JR, Mariani MR, Ceppi M, Newmark HL, Scalmati A, Lipkin M: Modulation of colonic epithelial cell proliferation, histone acetylation and luminal short chain fatty acids by variation of dietary fiber (wheat bran) in rats. Cancer Res 52: 5906–5912, 1992
Csordas A: On the biological role of histone acetylation. Biochem J 265: 23–38, 1990
Kruh J, Tichonicky L, Defer N: Effect of butyrate on gene expression. In: H.J. Binder, J.H. Cummings, K. Soergel (eds). Short Chain Fatty Acids. Kluwer, Dordrecht, 1994, pp 135–147
Steinman RA, Shiff SJ, Huang J, Yaroslavskiy B, Nalesnik M: Activation of a tumor suppressor gene by a nutrient derivative. Am Instit Cancer Res:(abstr) 1996
Hague A, Manning AM, Hanlon KA, Huschtscha LI, Hart D, Paraskeva C: Sodium butyrate induces apoptosis in human colonic tumor cell lines in a p53-independent pathway. Implications for the possible role of dietary fiber in the prevention of large-bowel cancer. Int J Cancer 55: 498–505, 1993
Scheppach W, Sommer H, Kirchner T, Paganelli GM, Bartram P, Christl S, Richter F, Dusel G, Kasper H: Effect of butyrate enemas on the colonic mucosa in distal ulcerative colitis. Gastroenterology 103: 51–56, 1992
Miller AA, Kurschel E, Osieka R, Schmidt CG: Clinical pharmacology of sodium butyrate in patients with acute leukemia. Eur J Cancer Clin Oncol 23: 1283–1287, 1987
Ikuta T, Kan YW, Swerdlow PS, Faller DV, Perrine SP: Alterations in protein-DNA interactions in the gamma-globin gene promoter in response to butyrate therapy. Blood 92: 2924–2933, 1998
Yanagi S, Yamashita M, Imai S: Sodium butyrate inhibits the enhancing effect of high fat diet on mammary tumorigenesis. Oncology 50: 201–204, 1993
Ranganna K, Yatsu FM: Inhibition of platelet-derived growth factor-BB-induced expression of glyceraldehyde-3-phosphate dehydrogenase by sodium butyrate in rat vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 17: 3420–3427, 1997
Barnard JA, Warwick G: Butyrate rapidly induces growth inhibition and differentiation in HT-29 cells. Cell Growth Diff 4: 495–501, 1993
Bravo R: Synthesis of the nuclear protein cyclin (PCNA) and its relationship with DNA replication. Exp Cell Res 163: 287–293, 1986
Kelman Z: PCNA: Structure, functions and interactions. Oncogene 14: 629–640, 1997
Hunter T, Pines J: Cyclins and cancer II: Cyclin D and cdk inhibitors come of age. Cell 79: 573–582, 1994
Hall PA, Levison DA, Woods AL, Yu CC, Kellock DB, Watkins JA, Barnes DM, Gillett CE, Camplejohn R, Dover R, Waseem NH, Lane DP: Proliferating cell nuclear antigen (PCNA) immunolocalization in paraffin sections: An index of cell proliferation with evidence of deregulated expression in some neoplasms. J Pathol 162: 285–294, 1990
Witzgall R, Brown D, Schwarz C, Bonventre JV: Localization of proliferating cell nuclear antigen, vimentin, c-fos, and clusterin in the postischemic kidney. Evidence for a heterogeneous genetic response among nephron segments, and a large pool of mitotically active and dedifferentiated cells. J Clin Invest 93: 2175–2188, 1994
Chuang LS-H, Ian H-I, Koh, T-W, Ng H-H, Xu G, Li BFL: Human DNA-(cytosine-5) methyltransferase-PCNA complex as a target for p21/WAF1. Science 277: 1996–2000, 1997
Jaskulski D, Deriel JK, Mercer WE, Calabretta B, Baserga R: Inhibition of cellular proliferation by antisense oligonucleotides to PCNA cyclin. Science 240: 1545–1546, 1988
Liu YC, Marraccino RL, Keng PC, Bambara RA, Lord EM, Chou WG, Zain SB: Requirement for proliferating cell nuclear antigen expression during stages of the Chinese hamster ovary cell cycle. Biochem 28: 2967–2974, 1989
Maeshima Y, Kashihara N, Sugiyama H, Makino H, Ota Z: Antisense oligonucleotides to proliferating cell nuclear antigen and ki-67 inhibit human mesangial cell proliferation. J Am Soc Nephrol 7: 2219–2229, 1996
Speir E, Epstein SE: Inhibition of smooth muscle cell proliferation by an antisense oligodeoxynucleotide targeting the messenger RNA encoding proliferating cell nuclear antigen. Circulation 86: 538–547, 1992
Geisterfer AA, Peach MJ, Owens GK: Angiotensin II induces hypertrophy, not hyperplasia, of cultured rat aortic smooth muscle cells. Circ Res 62: 749–758, 1988
Ramos K, Cox LR: Primary cultures of rat aortic endothelial and smooth muscle cells. An in vitro model to study xenobiotic-induced vascular cytotoxicity. In Vitro Cell Dev Biol 23: 288–296, 1987
Clowes AW, Schwartz SM: Significance of quiescent smooth muscle migration in the injured rat carotid artery. Circ Res 56: 139–145, 1985
Perrine SP, Olivieri NF, Faller DV, Vichinsky EP, Dover GJ, Ginder GD: Butyrate derivatives: New agents for stimulating fetal globin production in the beta-globin disorders. Am J Pediatr Hematol Oncol 16: 67–71, 1994
Morris GF, Mathews MB: Regulation of proliferating cell nuclear antigen during the cell cycle. J Biol Chem 264: 13856–13864, 1989
Jaskulski D, Gatti C, Travali S, Calabretta B, Baserga R: Regulation of the proliferating cell nuclear antigen cyclin and thymidine kinase mRNA levels by growth factors. J Biol Chem 263: 10175–10179, 1988
Wong RL, Katz ME, Tan EM, Cohen, S: Inhibition of nuclear DNA synthesis by an autoantibody to proliferating cell nuclear antigen/cyclin. Cell Immunol 110: 443–448, 1987
Tan CK, Sullivan K, Li XY, Tan EM, Downey KM, So AG: Autoantibody to the proliferating cell nuclear antigen neutralizes the activity of the auxiliary protein for DNA polymerase delta. Nucl Acid Res 15: 9299–9308, 1987
Clausen MR, Mortensen PB: Kinetic studies on the metabolism of short-chain fatty acids and glucose by the isolated rat colonocytes. Gastroenterology 106: 423–432, 1994
Fitch MD, Fleming SE: Metabolism of short-chain fatty acids by rat colonic mucosa in vivo. Am J Physiol 277: G31–G40, 1999
Vinores SA, Marangos PJ, Ko L: Butyrate-induced increase in neuronspecific enolase and ornithine decarboxylase in anaplastic glioma cells. Brain Res 281: 23–28, 1982
Tang S-J, Ko L-W, Lee Y-HW, Wang F-F: Induction of fos and sis proto-oncogenes and genes of the extracellular matrix proteins during butyrate induced glioma differentiation. Biochemica et Biophysica Acta 1048: 59–65, 1990
Schwartz B, Avivi-Green C, Polak-Charcon S: Sodium butyrate induces retinoblastoma protein dephosphorylation, p16 expression and growth arrest of colon cancer cells. Mol Cell Biochem 188: 21–30, 1998
Coleman WB, Smith GJ, Grisham JW: Development of dexamethasone-inducible tyrosine aminotransferase activity in WB-F344 rat liver epithelial stemlike cells cultured in the presence of sodium butyrate. J Cell Physiol 161: 463–469, 1994
Majesky MW, Benditt EP, Schwartz SM: Expression and developmental control of platelet-derived growth factor A-chain and B-chain/Sis genes in rat aortic smooth muscle cells. Proc Natl Acad Sci USA 85: 1524–1528, 1988
Han DKM, Liau G: Identification and characterization of developmentally regulated genes in vascular smooth muscle cells. Circ Res 71: 711–719, 1992
Munro E, Patel M, Chan P, Betteridge L, Gallagher K, Schachter M, Wolfe J, Sever P: Growth inhibition of human vascular smooth muscle cells by fenofibrate: A possible therapy for restenosis. Cardiovasc Res 28: 615–620, 1994
Hershey JC, Hautmann M, Thompson MM, Rothblum LI, Haystead TAJ, Owens GK: Angiotensin II-induced hypertrophy of rat vascular smooth muscle is associated with increased 18S rRNA synthesis and phosphorylation of the rRNA transcription factor, upstream binding factor. J Biol Chem 270: 25096–25101, 1995
Owens G: Differential effects of antihypertensive drug therapy on vascular smooth muscle cell hypertrophy, hyperploidy and hyperplasia in the spontaneously hypertensive rat. Circ Res 56: 525–536, 1985
Higgins PJ, Chaudhari P, Ryan MP: Cell-shape regulation and matrix protein p52 content in phenotypic variants of ras-transformed rat kidney fibroblasts. Biochem J 273: 651–658, 1991
Higgins PJ, Staiano-Coico L, Ryan MP: Cell-shape-dependent modulation of p52 (PAI-1) gene expression involves a secondary response pathway. Biochem J 306: 497–504, 1995
Powell JS, Clozel JP, Muller RK, Kuhn H, Hefti F, Hosang M, Baumgartner HR: Inhibitors of angiotensin-converting enzyme prevent myointimal proliferation after vascular injury. Science 245: 186–188, 1989
Morishita R, Gibbons GH, Ellison KE, Nakajima M, Zhang L, Kaneda Y, Ogihara T, Dzau VJ: Single intraluminal delivery of antisense cdc2 kinase and proliferating cell nuclear antigen oligonucleotides results in chronic inhibition of neointimal hyperplasia. Proc Natl Acad Sci USA 90: 8474–8478, 1993
Ohno T, Gordon D, San H, Pompili VJ, Imperiale MJ, Nabel GJ, Nabel, EG: Gene therapy for vascular smooth muscle cell proliferation after arterial injury. Science 265: 781–784, 1994
Gordon D, Reidy MA, Benditt EP, Schwartz SM: Cell proliferation in human coronary arteries. Proc Natl Acad Sci USA 87: 4600–4604, 1990
Libby P, Hansson GK: Involvement of the immune system in human atherogenesis: Current knowledge and unanswered questions. Lab Invest 64: 5–15, 1991
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ranganna, K., Yatsu, F.M., Hayes, B.E. et al. Butyrate inhibits proliferation-induced Proliferating Cell Nuclear Antigen expression (PCNA) in rat vascular smooth muscle cells. Mol Cell Biochem 205, 149–161 (2000). https://doi.org/10.1023/A:1007078200482
Issue Date:
DOI: https://doi.org/10.1023/A:1007078200482