Abstract
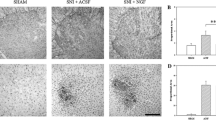
Small, circumscribed electrolytic lesions were made in the upper cervical corticospinal tract in adult rats. In the centre of the lesion, the axons and all other tissue elements were totally destroyed. Surrounding this region of destruction is an area of tissue which is only partially damaged. In this area TUNEL positive staining of contiguous rows of tract glial cells indicates massive oligodendrocytic apoptosis at 1–3 days after operation, but axons, astrocytes and blood vessels survive. From around 4 days, the corticospinal axons in this area are demyelinated, and the microglia contain ingested myelin, identified in electron micrographs as characteristic MBP immunoreactive laminar cytoplasmic bodies. After around 3 weeks, large numbers of Schwann cells, continuous with those on the pial surface of the spinal cord, accumulate along the lesion track and selectively infiltrate the perilesional reactive area, where they mingle intimately with the phagocytic microglia. Electron micrographs show that at this time basal lamina-enclosed Schwann cell processes establish non-myelinated ensheathment of axons. From around 4 weeks after operation, prominent Schwann cell myelination is indicated by P0 immunoreactivity, and peripheral type, one-to-one myelination in electron micrographs. Thus the effect of the selective loss of oligodendrocytes is to first activate microglia, and then to induce a replacement of myelin by Schwann cells.
Similar content being viewed by others
References
Adelman, E. S. & Aronson, S. M. (1972) Intramedullary nerve fiber and Schwann cell proliferation within the spinal cord (Schwannosis). Neurology 22, 726–731.
Anton, E. S., Weskamp, G., Reichardt, L. F. & Matthew, W. D. (1994) Nerve growth factor and its low-affinity receptor promote Schwann cell migration. Proc. Natl. Acad. Sci. USA 91, 2795–2799.
Baichwal, R. R., Bigbee, J. W. & Devries, G. H. (1988) Macrophage-mediated myelin-related mitogenic factor for cultured Schwann cells. Proc. Natl. Acad. Sci. USA 85, 1701–1705.
Baichwal, R. R. & Devries, G. H. (1989) A mitogen for Schwann cells derived from myelin basic protein. Biochem. Biophys. Res. Commun. 164, 883–888.
Baron-van evercooren, A., Duhamel-clerin, E., Boutry, J. M., Hauw, J. J. & Gumpel, M. (1993) Pathways of migration of transplanted Schwann cells in the demyelinated mouse spinal cord. J. Neurosci. Res. 35, 428–438.
Blight, A. R. & Young, W. (1989) Central axons in injured cat spinal cord recover electrophysiological function following remyelination by Schwann cells. J. Neurol. Sci. 91, 15–34.
Bolin, L. M. & Shooter, E. M. (1993) Neurons regulate Schwann cell genes by diffusible molecules. J. Cell Biol. 123, 237–243.
Brook, G. A., Plate, D., Franzen, R., Martin, D., Moonen, G., Schoenen, J., Schmitt, A. B., Noth, J. & Nacimiento, W. (1998) Spontaneous longitudinally orientated axonal regeneration is associated with the Schwann cell framework within the lesion site following spinal cord compression injury of the rat. J. Neurosci. Res. 53, 51–65.
Crowe, M. J., Bresnahan, J. C., Shuman, S. L., Masters, J. N. & Beattie, M. S. (1997) Apoptosis and delayed degeneration after spinal cord injury in rats and monkeys. Nature Med. 3, 73–76.
Davis, J. B. & Stroobant, P. (1990) Platelet-derived growth factors and fibroblast growth factors are mitogens for rat Schwann cells. J. Cell Biol. 110, 1353–1360.
Dusart, I., Marty, S. & Peschanski, M. (1992) Demyelination, and remyelination by Schwann cells and oligodendrocytes after kainate-induced neuronal depletion in the central nervous system. Neuroscience 51, 137–148.
Felts, P. A. & Smith, K. J. (1992) Conduction properties of central nerve fibers remyelinated by Schwann cells. Brain Res. 574, 178–192.
Gilmore, S. A. & Sims, T. J. (1993) Patterns of Schwann cell myelination of axons within the spinal cord. J. Chem. Neuroanat. 6, 191–199.
Hardy, R. J. & Friedrich, V. L., JR. (1996) Progressive remodeling of the oligodendrocyte process arbor during myelinogenesis. Dev. Neurosci. 18, 243–254.
Harrison, B. M. (1987) Schwann cells divide in a demyelinating lesion of the central nervous system. Brain Res. 409, 163–168.
Inoue, Y., Kagawa, T., Matsumura, Y., Ikenaka, K. & Mikoshiba, K. (1996) Cell death of oligodendrocytes or demyelination induced by overexpression of proteolipid protein depending on expressed gene dosage. Neurosci. Res. 25, 161–172.
Khan, S. & Wigley, C. (1994) Different effects of a macrophage cytokine on proliferation in astrocytes and Schwann cells. NeuroReport 5, 1381–1385.
Kreutzberg, G. W. (1996) Microglia: A sensor for pathological events in the CNS. Trends Neurosci. 19, 312–318.
Li, Y. & Raisman, G. (1994) Schwann cells induce sprouting in motor and sensory axons in the adult rat spinal cord. J. Neurosci. 14, 4050–4063.
Li, Y. & Raisman, G. (1995) Sprouts fromcut corticospinal axons persist in the presence of astrocytic scarring in long-term lesions of the adult rat spinal cord. Exp. Neurol. 134, 102–111.
Li, Y. & Raisman, G. (1997) Integration of transplanted cultured Schwann cells into the long myelinated fibre tracts of the adult spinal cord. Exp. Neurol. 145, 397–411.
Mahanthappa, N. K., Anton, E. S. & Matthew, W. D. (1996) Glial growth factor 2, a soluble neuregulin, directly increases Schwann cell motility and indirectly promotes neurite outgrowth. J. Neurosci. 16, 4673–4683.
Martin, J. R. & Webster, H. D. (1973) Mitotic Schwann cells in developing nerve: their changes in shape, fine structure, and axon relationships. Dev. Biol. 32, 417–431.
Mcdonald, J. W., Althomsons, S. P., Hyrc, K. L., Choi, D. W. & Goldberg, M. P. (1998) Oligodendrocytes from forebrain are highly vulnerable to AMPA/kainate receptor-mediated excitotoxicity. Nature Med. 4, 291–297.
Moore, S. & Thanos, S. (1996) The concept of microglia in relation to central nervous system disease and regeneration. Prog. Neurobiol. 48, 441–449.
Oka, A., Belliveau, M. J., Rosenberg, P. A. & Volpe, J. J. (1993) Vulnerability of oligodendroglia to glutamate: Pharmacology, mechanisms,andprevention. J. Neurosci. 13, 1441–1453.
Olby, N. J. & Blakemore, W. F. (1996) Primary demyelination and regeneration of ascending axons in the dorsal funiculus of the rat spinal cord following photochemically induced injury. J. Neurocytol. 25, 465–480.
Perry, V. H., Bell, M. D., Brown, H. C. & Matyszak, M. K. (1995) Inflammation in the nervous system. Curr. Opin. Neurobiol. 5, 636–641.
Ratner, N., Hong, D., Lieberman, M. A., Bunge, R. P. & Glaser, L. (1988) The neuronal cell-surface molecule mitogenic for Schwann cells is a heparinbinding protein. Proc. Natl. Acad. Sci. USA 85, 6992–6996.
Remahl, S. & Hildebrand, C. (1990) Relation between axons and oligodendroglial cells during initial myelination. I The glial unit. J. Neurocytol. 19, 313–328.
Ridley, A. J., Davis, J. B., Stroobant, P. & Land, H. (1989) Transforming growth factors-b1 and b2 are mitogens for rat Schwann cells. J. Cell Biol. 109, 3419–3424.
Rosenbluth, J., Hasegawa, M., Shirsaki, N., Rosen, C. L. & Liu, Z. (1990) Myelin formation following transplantation of normal fetal glia into myelindeficient rat spinal cord. J. Neurocytol. 19, 718–730.
Scolding, N. J., Morgan, B. P., Houston, W. A. J., Linington, C., Campbell, A. K. & Compston, D. A. S. (1989) Vesicular removal by oligodendrocytes of membrane attack complexes formed by activated complement. Nature 339, 620–622.
Shuman, S. L., Bresnahan, J. C. & Beattie, M. S. (1997) Apoptosis of microglia and oligodendrocytes after spinal cord contusion in rats. J. Neurosci. Res. 50, 798–808.
Sims, T. J. & Gilmore, S. A. (1983) Interactions between intraspinal Schwann cells and the cellular constituents normally occurring in the spinal cord: an ultrastructural study in the irradiated rat. Brain Res. 276, 17–30.
Stemple, D. L. & Mahanthappa, N. K. (1997) Neural stem cells are blasting off. Neuron 18, 1–4.
Suzuki, M. & Raisman, G. (1992) The glial framework of central white matter tracts: Segmented rows of contiguous interfascicular oligodendrocytes and solitary astrocytes give rise to a continuous meshwork of transverse and longitudinal processes in the adult rat fimbria. Glia 6, 222–235.
Suzuki, M. & Raisman, G. (1994) Multifocal pattern of postnatal development of the macroglial framework of the rat fimbria. Glia 12, 294–308.
Taniuchi, M., Clark, B. H., Schweitzer, J. B. & Johnson, E. M., JR. (1988) Expression of nerve growth factor receptors by Schwann cells of axotomised peripheral nerves: ultrastructural location, suppression by axonal contact, and binding properties. J. Neurosci. 8, 664–681.
Tuszynski, M. H., Peterson, D. A., Ray, J., Baird, A., Nakahara, Y. & Gage, F. H. (1994) Fibroblasts genetically modified to produce nerve growth factor induce robust neuritic ingrowth after grafting to the spinal cord. Exp. Neurol. 126, 1–14.
Vela, J. M., Dalmau, I., Gonz ´ alez, B. & Castellano, B. (1996) The microglial reaction in spinal cords of jimpy mice is related to apoptotic oligodendrocytes. Brain Res. 712, 134–142.
Warrington, A. E., Barbarese, E. & Pfeiffer, S. E. (1993) Differential myelinogenic capacity of specific developmental stages of the oligodendrocyte lineage upon transplantation into hypomyelinating hosts. J. Neurosci. Res. 34, 1–13.
Webster, H. D. F., Martin, J. R. & O'connell, M. F. (1973) The relationships between interphase Schwann cells and axons before myelination: a quantitative electron microscopic study. Dev. Biol. 32, 401–416.
Wren, D. R. & Noble, M. (1989) Oligodendrocytes and oligodendrocyte/type-2-astrocyte progenitor cells of adult rats are specifically susceptible to the lytic effects of complement in absence of antibody. Proc. Natl. Acad. Sci. USA 86, 9025–9029.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, Y., Field, P.M. & Raisman, G. Death of oligodendrocytes and microglial phagocytosis of myelin precede immigration of Schwann cells into the spinal cord. J Neurocytol 28, 417–427 (1999). https://doi.org/10.1023/A:1007026001189
Issue Date:
DOI: https://doi.org/10.1023/A:1007026001189