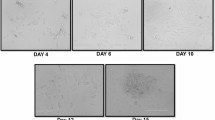
Abstract
Pheochromocytomas are rare tumours, with an incidence of 1–2 per million which arise from chromaffin cells of the adrenal medulla. They occur sporadically or as part of dominantly inherited cancer syndromes like multiple endocrine neoplasia 2 (MEN2A and 2B) and others. Continuous cell lines, not available so far, are essential tools for studies in these tumours. A continuous cell line (KNA) was established from a sporadic pheochromocytoma of the right adrenal gland of a 73-year-old woman. The KNA cells grow as suspensions of spheroids and show the morphological and immunocytochemical characteristics of neuronal chromaffin cells, such as neuroendocrine granules, and positive reactions to chromogranin- and related peptide-, neuron specific enolase and vasoactive intestinal peptide antibodies. Neurite-like processes are formed after addition of nerve growth factor. Chromosomal analyses revealed a diploid (46,XX, n=50) to hypodiploid (43–45,XX, n=15) karyotype. In hypodiploid metaphases most frequently #19, #17, #21 and #22 were missing. Chromosome arms 1p and 4q showed apparently consistent interstitial deletions: 6q, 8q, 13q and 22q showed clonal interstitial deletions. The cell line shows a heterozygous sequence variant TGC (cysteine) to TGG (tryptophan) in codon 611 in exon 10 of the RET proto-oncogene. So far, PC-12, a rat adrenal pheochromocytoma, has been the only continuous pheochromocytoma cell line available. KNA represents the first report on a human continuous pheochromocytoma cell line, the first report of structural chromosome aberrations in pheochromocytomas and the first report of a RET mutation TGC to TGG in exon 10 of the RET proto-oncogene in a sporadic pheochromocytoma. © 1998 Chapman and Hall
Similar content being viewed by others
References
Andersen, L. B., Fountain, J. W., Gutmann, D. H., Tarle, S. A., Glover, T. W., Dracopoli, N. C., Housman, D. E. & Collins, F. S. (1993) Mutations in the neurofibromatosis 1 gene in sporadic malignant melanomas. Nature Genetics 3, 118–21.
Arnold, A. (1996) RET mutation screening in sporadic pheochromocytoma. Journal of Clinical Endocrinology and Metabolism 81, 430 (letter).
Behmel, A., Reichl, E., Pfragner, R. & Niederle, B. (1997) High incidence of gonosomal aneuploidy in lymphocytes of patients with sporadic, MEN1 and MEN2 associated neuroendocrine tumors: agerelated X chromosome instability versus genuine mosaicism. Medizinische Genetik 1, 44, 1–3.
Benedum, U. M., Baeuerle, P. A., Koneckid, S., Frank, R., Rowell, J. & Mallet, J. (1986) The primary structure of bovine chromogranin A: a representative of a class of acidic secretory proteins common to a variety of peptidergic cells. EMBO Journal 5, 1495–502.
Benedum, U. M., Lamouroux, A., Konecki, D. S., Rosa, P., Hille, A., Baeuerle, P. A., Frank, R., Lottspicgs, S., Mallet, J. & Huttner, W. B. (1987) The primary structure of human secretogranin I (chromogranin B): comparison with chromogranin A reveals homologous terminal domains and a large intervening variable region. EMBO Journal 6, 1203–11.
Brower, M., Carney, D. N., Oie, H. K., Gazdar, A. F. & Minna, I. D. (1986) Growth of cell lines and clinical specimens of human non-small cell lung cancer in serum-free defined medium. Cancer Research 46, 798–806.
Caillard, T., Opstal, W. Y., Scarcerieux, V., Billardon, C. & Rostene, W. (1995) Treatment of PC12 cells by nerve growth factor, dexamethasone, and forskolin. Effects on cell morphology and expression of neurotensin and tyrosin hydroxylase. Molecular Neurobiology 10, 105–14.
Decker, H. J. H., Walter, T. A., Neumann, H. J. H. & Sandberg, G. A. (1988) Cytogenetics of familial pheochromocytoma: importance of trisomy 7 in tumor progression? Blut 57, 270.
Dinges, H. P., Wirnsberger, G. & HÖfler, H. (1989) Immunocytochemistry in cytology. Comparative evaluation of different techniques. Analytical and Quantitative Cytology and Histology 11, 22–32.
Eng, C., Mulligan, L. M., Smith, D. P., Healey, C. S., Frilling, A., Raue, F., Neumann, H. P., Ponder, M. A. & Ponder, B. A. J. (1995) Low frequency of germline mutations in the RET protooncogene in patients with apparently sporadic medullary thyroid carcinoma. Clinical Endocrinology 43, 123–7.
Eng, C., Clayton, D., Schuffenecker, I., Lenoir, G., Cote, G., Gagel, R. F., Ploos van Amstel, H. K., Lips, C. J. M., Nishisho, I., Takai, S. C., Marsh, D. J., Robinson, B. G., Frank-Raue, K., Raue, F., Xue, F., Noll, W. W., Romei, C., Pacini, F., Fink, M., Niederle, B., Zadenius, J., Nordenskjold, M., Komminoth, P., Handy, G. H., Gharib, H., Thibodeau, S. N., Lacroix, A., Frilling, A., Ponder, B. A. J. & Mulligan, L. M. (1996) The relationship between specific RET proto-oncogene mutations and disease phenotype in Multiple Endocrine Neoplasia Type 2. Journal of American Medical Association 276, 1575–9.
Eng, C., Mulligan, L. M., Healey, C. S., Houghton, C., Frilling, A., Raue, F., Thomas, G. A. & Ponder, B. A. J. (1996) Heterogeneous mutation of the RET proto-oncogene in subpopulations of medullary thyroid carcinoma. Cancer Research 56, 2167–70.
Eng, C. & Mulligan, L. M. (1997) Mutations of the RET proto-oncogene in the Multiple Endocrine Neoplasia Type 2 syndromes, related sporadic tumours, and Hirschsprung disease. Human Mutation 9, 97–109.
Evers, B. M., Rady, P. L., Stephen, K., Tyring, K., Sanchez, R. L., Rajamaran, S., Townsend, M., Jr. & Thompson, J. C. (1992) Amplification of the HER-2/neu proto-oncogene in human endocrine tumors. Surgery 112, 211–18.
Geldorf, A. A. (1995) Nerve-growth-factor-dependent neurite outgrowth assay; a ressearch model for chemotherapy-induced neuropathy. Journal of Cancer Research and Clinical Oncology 121, 657–69.
Greene, L. A. & Rein, G. (1977a) Release, storage and uptake of catecholamines by a clonal line of nerve growth factor responsive pheochromocytoma cells. Brain Research 129, 247–63.
Greene, L. A. & Rein, G. (1977b) Synthesis, storage and release of acetylcholine by a noradrenergic pheochromocytoma cell line. Nature 268, 349.
Greene, L. A. & Tischler, A. S. (1976) Establishment of a noradrenergic clonal line of rat pheochromocytoma cells which respond to nerve growth factor. Proceedings of the National Academy of Sciences USA 73, 2424–26.
Gutmann, D. H., Cole, J. L., Stone, W. J., Ponder, B. A. J. & Collins, F. S. (1994) Loss of neurofibromin in adrenal gland tumors from patients with neurofibromatosis type I. Genes, Chromosomes & Cancer 10, 55–8.
Gutmann, D. H., Geist, R. T., Kamala, R., Wallin, G. & Moley, J. F. (1993) Gene expression in pheochromocytomas from patients without NFI. Genes, Chromosomes & Cancer 13, 104–9.
Hoch-Ligeti, C. & Camp, J. L. (1959) Catecholamine production in tissue culture of human adrenal medullary tumors and adrenal medulla. Proceedings of the Society for Experimental Biology and Medicine 102, 692–3.
HÖfler, H., PÜtz, B., Ruhri, C., Wirnsberger, G., Klimpfinger, M. & Smolle, J. (1987) Simultaneous localization of calcitonin mRNA and peptide in a medullary thyroid carcinoma. Virchows Archiv 54, 144–51.
Iyengar, S., Tallini, G., Sirugo, G., Bale, A. E. & Kidd, K. K. (1995) Mutation of the vHL Gene in individuals with sporadic and familial pheochromocytoma.American Journal of Human Genetics 57, Supplement A67,357.
Johnson, M. & Johnson, B. (1984) Efficient synthesis of high specific 35S-labeled human b-globin pre-mRNA. Biotechniques 2, 156–162.
Jordan, D. K., Patil, S. R., Divelbiss, J. E., Vemuganti, S., Headley, C., Waziri, H. & Gurll, J. (1989) Cytogenetic abnormalities in tumors of patients with von Hippel-Lindau disease. Cancer Genetics and Cytogenetics 42, 227–41.
Khosla, S., Patel, V. M., Hay, I. D., Schaid, D. J., Grant, S., van Heerden, J. A. & Thibodeau, S. N. (1991) Loss of heterozygosity suggests multiple genetic alterations in pheochromocytomas and medullary thyroid carcinomas. Journal of Clinical Investigation 87, 1691–9.
Lasfargues, E. Y. & Moore, D. H. (1971) A method for the continuous cultivation of mammary epithelium. In Vitro Cellular & Developmental Biology-Animal 7, 21–25.
Luft, J. H. (1961) Improvements in epoxy resin embedding methods. Journal of Biophysical & Biochemical Cytology 9, 409–14.
Mathew, C. G. P., Smith, B. A., Thorpe, K., Wong, Z., Royle, N. J., Jeffries, A. J. & Ponder, B. A. J. (1987) Deletion of genes on chromosome 1 in endocrine neoplasia. Nature 328, 524–26.
Modigliani, E., Vasen, H. M., Raue, K., Dralle, H., Frilling, A., Gheri, G., Brandi, M. L., Limberg, E., Niederle, B., Forgas, L., Rosenberg-Bourgin, M., Calmettes, C. & The European Study Group (1995) Pheochromocytoma in multiple endocrine neoplasia type 2: European study. Journal of Internal Medicine 238, 363–7.
Moley, J. F., Brother, M. B., Fong, C. T., White, P. S., Baylin, S. B., Nelkin, B., Wells, S. A. & Brodeur, G. M. (1992) Consistent association of 1p loss of heterozygosity with pheochromocytomas from patients with multiple endocrine neoplasia type 2 syndromes. Cancer Research 52, 770–4.
Mulligan, L. M., Gardner, E., Smith, B. A., Mathew, C. G. & Ponder, B. A. J. (1993) Genetic events in tumour initiation and progression in multiple endocrine neoplasia type 2. Genes Chromosomes & Cancer 6, 166–77.
Nagatsu, T. & Udenfriend, S. (1972) Photometric assay of dopamine β-hydroxylase. Clinical Chemistry 18, 980–983.
Neumann, H. J. H., Schempp, W. & Wienker, T. F. (1988) High-resolution chromosome banding and fragile site studies in von Hippel-Lindau syndrome. Cancer Genetics Cytogenetics 31, 41–46.
Pfragner, R., HÖfler, H., Behmel, A., Ingolic, E. & Walser, V. (1990) Establishment and characterization of continuous cell line MTC-SK derived from a human medullary thyroid carcinoma. Cancer Research 50, 4160–66.
Pfragner, R., Sadjak, A. & Walser, V. (1984) The effect of nerve growth factor (NGF) on the catecholamine contents of two human pheochromocytomas in tisue culture. Experimental Pathology 26, 21–31.
Pfragner, R. & Walser, V. (1980) Long-term tissue culture of human pheochromocytomas. Experimental Pathology 18, 423–9.
Pfragner, R. Wirnsberger, G., Behmel, A., Wolf, G., Passath, A., Ingolic, E., Adamiker, D. & Schauenstein, K. (1993) New continuous cell line from human medullary thyroid carcinoma: SINJ. Phenotypic analysis and in vivo carcinogenesis. International Journal of Oncology 2, 831–36.
Pfragner, R., Wirnsberger, G., Niederle, B., Behmel, A., Rinner, I., Mandl, A., Wawrina, F., Luo, J. S., Adamiker, D., HÖger, H., Ingolic, E. & Schauenstein, K. (1996) Establishment of a continuous cell line from a human carcinoid of the small intestine (KRJ-I): Characterization and effects of 5-azacytidine on proliferation. International Journal of Oncology 8, 513–520.
Polinger, I. S. (1970) Separation of cell types in embryonic heart cell cultures. Experimental Cell Research 63, 78–82.
porta, S., Epple, A., Kvetnansky, R., Leitner, G., Emsenhuber, W. & Radkohl, W. (1995) Temporary increase of plasma epinephrine affects stress response 24 hours later. Physiology & Behaviour 88, 223–8.
REUBI, C. Waser, B., Khosla, S., Kvols, L., Geollner, J. R., Krenning, E. & Lamberts, S. (1992) In vitro and in vivo detection of somatostatin receptors in pheochromocytomas and paragangliomas. Journal of Clinical Endocrinology and Metabolism 74, 1082–89
Reynolds, E. S. (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. Journal of Cell Biology 17, 208–12.
Rodien, P., Jeunemaitre, X., Dumont, C., Beldjord, C. & Plouin, P. F. (1997) Genetic alterations of the RET proto-onogene in familial and sporadic pheochromocytomas. Hormone Research 47, 263–268.
Ryan, U. S. & Hart, M. A. (1986) Electron microscopy of endothelial cells in culture I. Transmission microscopy. Journal of Tissue Culture Methods 10, 31–33.
Ryan, U. S. & Hart, M. A. (1986) Electron microscopy II. Scanning electron microscopy and OTOTO impregnation method. Journal of Tissue Culture Methods 10, 35–36.
Shine, E., Fujita, S., Takami, K., Kurahashi, H., Kurita, T., Kobayashi, T., Mon, T., Nishisho, I. & Takai, S. (1993) Deletion mapping of chromosome 1p and 22q in pheochromocytoma. Japanese Journal of Cancer Research 84, 402–8.
Takai, S., Tataishi, H., Nishisho, I., Miki, T., Motomura, K., Miyauchi, A., Kato, M. & Ikeuchi, T. (1987) Loss of genes on chromosome 22 in medullary thyroid carcinoma and pheochromocytoma. Japanese Journal of Cancer Research 78, 894–98.
Tanaka, N., Nishisho, I., Yamamoto, M., Miya, A., Shin, E., Karakawa, K., Fujita, S., Kobayashi, T., Rouleau, G. A., Mori, T. & Takai, S. (1992) Loss of heterozygosity on the long arm of chromosome 22 in pheochromocytoma. Genes, Chromosomes & Cancer 5, 399–403.
The, I., Murthy, A. E., Hannigan, G. E., Jacoby, L. B., Menon, A. G., Gusella, J. F. & Bednards, A. (1993) Neurofibromatosis type 1 gene mutations in neuroblastoma. Nature Genetics 3, 62–66.
Tischler, A. S., Dichter, M. A., Biales, B., Delellis, R. A. Wolfe, H. (1976) Neuronal properties of cultured human endocrine tumor cells of proposed neural crest origin. Science 192, 902–4.
Tischler, A. S. & Greene, L. A. (1975) Nerve growth factor-induced process formation by cultured rat pheochromocytoma cells. Nature 258, 341–2.
Tischler, A. S. & Greene, A. (1978) Morphologic and cytochemical properties of a clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Laboratory Investigation 39, 77–89.
Wirnsberger, G. H., Becker, H., Ziervogel, F. & HÖfler, H. (1992) Diagnostic immunohistochemistry of neuroblastic tumors. American Journal of Surgical Pathology 16, 49–57.
Zeiger, M. A., Berton, Z., Keiser, H., Linehan, W. M. & Gnarra, J. R. (1993) Loss of heterozygosity on the short arm of chromosome 3 in sporadic, von Hieppel-Lindau disease-associated, and familial pheochromocytoma. Genes, Chromosomes & Cancer 13, 151–6.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pfragner, R., Behmel, A., Smith, D.P. et al. First continuous human pheochromocytoma cell line: KNA Biological, cytogenetic and molecular characterization of KNA cells. J Neurocytol 27, 175–186 (1998). https://doi.org/10.1023/A:1006959625068
Issue Date:
DOI: https://doi.org/10.1023/A:1006959625068