Abstract
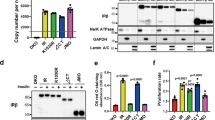
Insulin selectively induces mitogenesis in quiescent SV40 large T antigen-transformed murine 3T3T (CSV3-1) cells but not in quiescent nontransformed 3T3T cells. This mitogenic effect induced by insulin in CSV3-1 cells requires an induction of AP-1 activity associated with c-Jun and JunB. To further investigate the mechanisms that are involved in insulin-induced mitogenesis in CSV3-1 cells, the current experiments were performed. The results show that following insulin stimulation, the insulin receptor β-subunit and the insulin receptor substrate-1 undergo a much more significant tyrosine phosphorylation in CSV3-1 cells than in 3T3T cells. Insulin also induces tyrosine phosphorylation of a 73 kDa protein that is coprecipitated with the tyrosine-phosphorylated insulin receptor in CSV3-1 cells but not in 3T3T cells. The increased tyrosine phosphorylation in response to insulin stimulation in CSV3-1 cells does not appear to be due to an increase in the level of expression of the insulin receptor and does not appear to result from a significant change in tyrosine phosphatase activity compared to nontransformed cells. The results also show that the insulin effect in CSV3-1 cells is not mediated by insulin-like growth factor 1 receptor because insulin at the concentrations that induce mitogenesis does not increase the tyrosine phosphorylation of the insulin-like growth factor 1 receptor and the expression level of the receptor is not significantly changed in CSV3-1 cells compared to nontransformed cells. These data together indicate that the selective mitogenic effect of insulin on CSV3-1 cells involves increased tyrosine phosphorylation of the insulin receptor, the insulin receptor substrate-1 and the 73 kDa protein, although the underlying mechanisms need to be further elucidated.
Similar content being viewed by others
References
Sun H, Tonks NK: The coordinated action of protein tyrosine phosphatases and kinases in cell signaling. TIBS 19: 480–485, 1994
Stone RL, Dixon JE: Protein-tyrosine phosphatases. J Biol Chem 269: 31323–31326, 1994
Ullrich A, Schlessinger J: Signal transduction by receptors with tyrosine kinase activity. Cell 61: 203–212, 1990
Cadena DL, Gill GN: Receptor tyrosine kinases. FASEB J 6: 2332–2337, 1992
Streuli M: Protein tyrosine phosphatases in signaling. Curr Opin Cell Biol 8: 182–188, 1996
White MF, Kahn CR: The insulin signaling system. J Biol Chem 269: 1–4, 1994
Myers MG Jr, White MF: Insulin signal transduction and the IRS proteins. Ann Rev Pharmacol Toxicol 36: 615–658, 1996
Myers MG Jr, Sun XJ, White MF: The IRS-1 signaling system. TIBS 19: 289–293, 1994
Tavaré JM, Siddle K: Mutational analysis of insulin receptor function: Consensus and controversy. Biochim Biophys Acta 1178: 21–39, 1993
Czech MP: Signal transmission by the insulin-like growth factors. Cell 59: 235–238, 1989
Baserga R, Hongo A, Rubini M, Prisco M, Valentinis B: The IGF-1 receptor in cell growth, transformation and apoptosis. Biochim Biophys Acta 1332: F105–F126, 1997
Myers MG Jr, Sun XJ, Cheatham B, Jachna BR, Glasheen EM, Backer JM, White MF: IRS-1 is a common element in insulin and insulin-like growth factor-1 signaling to the phosphatidylinositol 3′-kinase. Endocrinology 132: 1421–1430, 1993
Wang H, Scott RE: Insulin-induced mitogenesis associated with transformation by the SV40 large T antigen. J Cell Physiol 147: 102–110, 1991
Wang H, Wang J-Y, Johnson LR, Scott RE: Selective induction of c-jun and jun-B but not c-fos or c-myc during mitogenesis in SV40–transformed cells at the predifferentiation growth arrest state. Cell Growth Differ 2: 645–652, 1991
Wang H, Scott RE: Induction of c-jun independent of PKC, pertussis toxin-sensitive G protein, and polyamines in quiescent SV40–transformed 3T3 T cells. Exp Cell Res 203: 47–55, 1992
Wang H, Scott RE: Unique and selective mitogenic effects of vanadate on SV40–transformed cells. Mol Cell Biochem 153: 59–67, 1995
Wang H, Xie Z, Scott RE: Induction of AP-1 activity associated with c-Jun and JunB is required for mitogenesis induced by insulin and vanadate in SV40–transformed 3T3T cells. Mol Cell Biochem 168: 21–30, 1997
Wang H, Scott RE: Distinct protein tyrosine phosphorylation during mitogenesis induced in quiescent SV40–transformed 3T3 T cells by insulin or vanadate. J Cell Physiol 158: 408–416, 1994
Wang H, Sturtevant DB, Scott RE: Nonterminal and terminal adipocyte differentiation of murine 3T3 T mesenchymal stem cells. In: JE Celis (ed). Cell Biology: A Laboratory Handbook. Academic Press, San Diego, 1994, pp 193–198
Wang H, Scott RE: Adipocyte differentiation selectively represses the serum inducibility of c-jun and junB by reversible transcription-dependent mechanisms. Proc Natl Acad Sci USA 91: 4649–4653, 1994
Wang H, Xie Z, Scott RE: Differentiation modulates the balance of positive and negative Jun/AP-1 DNA binding activities to regulate cellular proliferative potential: different effects in nontransformed and transformed cells. J Cell Biol 135: 1151–1162, 1996
Wang H, Xie Z, Scott RE: Adipocyte differentiation progressively represses growth factor responsiveness without modifying the expression or phosphorylation of multiple growth factor receptors. Mol Cell Differ 4: 211–225, 1996
Estervig DN, Minoo P, Tzen C-Y, Scott RE: Three distinct effects of SV40 T-antigen gene transfection on cellular differentiation. J Cell Physiol 142: 552–558, 1990
Wang H, Xie Z, Scott RE: JunD phosphorylation, and expression of AP-1 DNA binding activity modulated by serum growth factors in quiescent murine 3T3T cells. Oncogene 13: 2639–2647, 1996
Cullen KJ, Yee D, Sly WS, Perdue J, Hampton B, Lippman ME, Rosen N: Insulin-like growth factor receptor expression and function in human breast cancer. Cancer Res 50: 48–53, 1990
Olefsky JM: The insulin receptor: A multifunctional protein. Diabetes 39: 1009–1016, 1990
Harada S, Smith RM, Smith JA, White MF, Jarett L: Insulin-induced egr-1 and c-fos expression in 32D cells requires insulin receptor, Shc, and mitogen-activated protein kinase, but not insulin receptor substrate-1 and phosphatidylinositol 3–kinase activation. J Biol Chem 271: 30222–30226, 1996
Lavan BE, Lane WS, Lienhard GE: The 60–kDa phosphotyrosine protein in insulin-treated adipocytes is a new member of the insulin receptor substrate family. J Biol Chem 272: 11439–11443, 1997
Lavan BE, Fantin VR, Chang ET, Lane WS, Keller SR, Lienhard GE: A novel 160–kDa phosphotyrosine protein in insulin-treated embryonic kidney cells is a new member of the insulin receptor substrate family. J Biol Chem 272: 21403–21407, 1997
Smith-Hall J, Pons S, Patti ME, Burks DJ, Yenush L, Sun XJ, Kahn CR, White MF: The 60 kDa insulin receptor substrate functions like an IRS protein (pp60IRS3) in adipose cells. Biochemistry 36: 8304–8310, 1997
Chen J, Sadowski HB, Kohanski RA, Wang LH: Stat5 is a physiological substrate of the insulin receptor. Proc Natl Acad Sci USA 94: 2295–2300, 1997
Rondinone CM, Wang LM, Lonnroth P, Wesslau C, Pierce JH, Smith U: Insulin receptor substrate (IRS) 1 is reduced and IRS-2 is the main docking protein for phosphatidylinositol 3–kinase in adipocytes from subjects with non-insulin-dependent diabetes mellitus. Proc Natl Acad Sci USA 94: 4171–4175, 1997
Yenush L, White MF: The IRS-signaling system during insulin and cytokine action. Bioessays 19: 491–500, 1997
Kosaki A, Yamada K, Suga J, Otaka A, Kuzuya H: 14–3–3β-protein associates with insulin receptor substrate 1 and decreases insulin-stimulated phosphatidylinositol 3′-kinase activity in 3T3L1 adipocytes. J Biol Chem 273: 940–944, 1998
White MF, Shoelson SE, Keutmann H, Kahn CR: A cascade of tyrosine autophosphorylation in the β-subunit activates the phosphotransferase of the insulin receptor. J Biol Chem 263: 2969–2980, 1988
Wilden PA, Kahn CR, Siddle K, White MF: Insulin receptor kinase domain autophosphorylation regulates receptor enzymatic function. J Biol Chem 267: 16660–16668, 1992
Wilden PA, Siddle K, Haring E, Backer JM, White MF, Kahn CR: The role of insulin receptor kinase domain autophosphorylation in receptor-mediated activities: Analysis with insulin and anti-receptor antibodies. J Biol Chem 267: 13719–13727, 1992
Formisano P, Condorelli G, Condorelli G, Beguinot F: Antiphosphotyrosine immunoprecipitation of an insulin-stimulated receptor phosphatase activity from FRTL5 cells. Endocrinology 128: 2949–2957, 1991
Mooney RA, Bordwell KL: Differential dephosphorylation of the insulin receptor and its 160–kDa substrate (pp 160) in rat adipocytes. J Biol Chem 267: 14054–14060, 1992
Swarup G, Cohen S, Garber DL: Inhibition of membrane phosphotyrosyl-protein phosphatase activity by vanadate. Biochem Biophys Res Commun 107: 1104–1109, 1982
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wang, H. Increased tyrosine phosphorylation of the insulin receptor, the insulin receptor substrate-1 and a 73 kDa protein associated with insulin-induced mitogenesis in SV40-transformed 3T3T cells. Mol Cell Biochem 197, 61–70 (1999). https://doi.org/10.1023/A:1006937720559
Issue Date:
DOI: https://doi.org/10.1023/A:1006937720559