Abstract
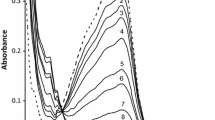
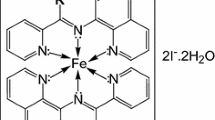
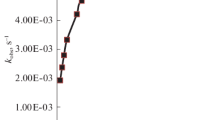
The kinetics of substitution of Fe(tptz)2+2 by 2,2′-bipyridine and 1,10-phenanthroline have been investigated in acetate buffers in the 3.6–5.6 pH range employing stopped-flow spectrophotometry. These reactions are very fast and complete within 5 s. The rate of substitution is linearly dependent on [phen] and [bpy]2, and increases with the increase in pH. Suitable mechanisms have been proposed involving the unprotonated form of the entering ligand, viz. bipyridine/phenanthroline as the reactive species. The pKa values of bipyridine and phenanthroline, determined from the kinetic data, are in agreement with the literature values. It is concluded that the substitutions of iron(II)-diimine complexes also occur by an associative mechanism.
Similar content being viewed by others
References
A.A. Schilt, Analytical applications of 1,10-phenanthroline and related compounds, Internat ser, of monographs in analytical chemistry, vol. 32, Pergman Press, New York, 1969.
M.L. Moss and M. G. Melon, Ind. Eng. Chem., 14, 862 (1942).
P.F. Collins, H. Diehl and G. F. Smith, Anal. Chem., 31, 1862 (1959).
D.W. Margerum and L. P. Margenthaler, J. Am. Chem. Soc., 84, 706 (1961).
J. Burgess, J. Chem. Soc. Dalton Trans., 1061 (1972).
J. Burgess, J. Chem. Soc. Dalton Trans., 203 (1972).
G.K. Pagenkopf and D.W. Margerum, Inorg. Chem., 7, 2514 (1968).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Visweswara Rao, G., Sridhar, Y., Hela, P.G. et al. Kinetics and mechanism of substitution of bis(tptz)iron(II) by 2,2′-bipyridine and 1,10-phenanthroline. Transition Metal Chemistry 24, 566–570 (1999). https://doi.org/10.1023/A:1006920728571
Issue Date:
DOI: https://doi.org/10.1023/A:1006920728571