Abstract
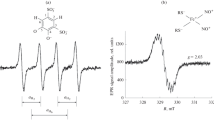
Spin-trapping techniques combined with electron paramagnetic resonance (EPR) spectroscopy to measure nitric oxide (·NO) production were compared in the ischemic-reperfused myocardium for the first time, using both aqueous-soluble and lipophilic complexes of reduced iron (Fe) with dithiocarbamate derivatives. The aqueous-soluble complex of Fe and N-methyl-D-glucamine dithiocarbamate (MGD) formed MGD2-Fe-NO complex with a characteristic triplet EPR signal (aN12.5 G and giso = 2.04) at room temperature, in native isolated rat hearts following 40 min global ischemia and 15 min reperfusion. Diethyldithiocarbamate (DETC) and Fe formed in ischemic-reperfused myocardium the lipophilic DETC2-Fe-NO complex exhibiting an EPR signal (g⊥ = 2.04 and g∥ = 2.02 at 77K) with a triplet hyperfine structure at g⊥. Dithiocarbamate-Fe-NO complexes detected by both trapping agents were abolished by the ·NO synthase inhibitor, NG-nitro-L-arginine methyl ester. Quantitatively, both trapping procedures provi ded similar values for tissue ·NO production, which were observed primarily during ischemia. Postischemic hemodynamic recovery of the heart was not affected by the trapping procedure. (Mol Cell Biochem 175: 91–97, 1997)
Similar content being viewed by others
References
Moncada S, Palmer RM Jr, Higgs EA: Nitric Oxide: Physiology, pathophysiology, and pharmacology. Pharmacol Rev 43:109–142, 1991
Darley-Usmar V, Wiserman H, Halliwell B: Nitric oxide and oxygen radicals: a question of balance. FEBS Lett 369: 131–135, 1995
Zweier JL, Wang P, Kuppusamy P: Direct measurement of nitric oxide generation in the ischemic heart using electron paramagnetic resonance spectroscopy. J Biol Chem 270: 304–307, 1995
Komarov AM, Kramer JH, Gibson B, Mak IT, Weglicki WB: Increased nitric oxide complex formation in native postischemic rat heart tissue. Circulation 92(Suppl. 1): 0210, 1995
Maulik N, Engelman DT, Watanabe M, Engelman R, Maulik G, Gordis GA, Das DK: Nitric oxide signaling in ischemic heart. Cardiovasc Res 30: 593–601, 1995
Lefer AM, Tsao PS, Lefer DJ, Ma XL: Role of endothelial dysfunction in the pathogenesis of reperfusion injury after myocardial ischemia. FASEB J 5: 2029–2034; 1991
Matheis G, Sherman MP, Buckberg GD, Haybron DM, Yong HH, Ignarro LJ: Role of L-arginine-nitric oxide pathway in myocardial reoxygenation injury. Am J Physiol 262: H616–620, 1992
Komarov A, Mattson D, Jones MM, Singh PK, Lai C-S: In vivo spin trapping of nitric oxide in mice. Biochem Biophys Res Commun 195: 1191–1198, 1993
Lai C-S, Komarov AM: Spin trapping of nitric oxide produced in vivo in septic shock mice. FEBS Lett 345: 120–124, 1994
Lai C-S, Komarov AM: Dithiocarbamate spin traps for in vivo detection of nitric oxide produced in mice. In: H Ohya-Nishiguchi, L Packer (eds). Bioradicals Detected by ESR Spectroscopy. Birkhauser Verlag, Bazel, 1995, pp 163–171
Komarov AM, Lai C-S: Detection of nitric oxide production in mice by spin-trapping electron paramagnetic resonance spectroscopy. Biochim Biophys Acta 1272: 29–36, 1995
Mordvintcev P, Mulsch A, Busse R, Vanin A: On-line detection of nitric oxide formation in liquid aqueous phase by electron paramagnetic resonance spectroscopy. Anal Biochem 199: 142–146, 1991
Mulsch A, Mordvintcev P, Bassenge E, Jung F, Clement B, Busse R: In vivo spin trapping of glyceryl trinitrate-derived nitric oxide in rabbit blood vessels and organs. Circulation 92: 1876–1882, 1995
Shinobu LA, Jones SG, Jones MM: Sodium N-methyl-D-glucamine dithiocarbamate and cadmium in toxication. Acta Pharmacol Toxicol 54: 189–194, 1984
Giboreau P, Morin C: Procedure for the preparation of pure dithiocarbamates. J Org Chem 59: 1205–1207, 1994.
Kramer JH, Misik V, Weglicki WB: Magnesium-deficiency potentiates free radical production associated with postischemic injury to rat hearts: vitamin E affords protection. Free Radic Biol Med 16: 713–723, 1994
Kojima S, Kiyozumi M, Honda T, Senba Y, Kaminaka K, Ohnishi M: Studies on poisonous metals. XVIII. Effects of several dithiocarbamates on tissue distribution and excretion of cadmium in rats. Chem Pharm Bull 35: 3838–3844, 1987
Wang P, Zweier JL: Measurement of nitric oxide and peroxynitrite generation in the postischemic heart. Evidence for peroxynitrite-mediated reperfusion injury. J Biol Chem 271: 29223–29230, 1996
Lancaster JR Jr: Simulation of the diffusion and reaction of endogenously produced nitric oxide. Proc Natl Acad Sci USA 91: 8137–8141, 1994
Konorev EA, Joseph J, Tarpey M, Kalyanaraman B: The mechanism of cardioprotection by S-nitrosoglutathione monoethyl ester in rat isolated heart during cardioplegic ischaemic arrest. Brit J Pharmacol 119: 511–518, 1996
Beckman JS, Beckman TW, Chen J, Marshall PA, Freeman BA: Apparent hydroxyl radical production from peroxynitrite: implications for endothelial cell injury from nitric oxide and superoxide. Proc Natl Acad Sci USA 87: 1620–1624, 1990
Masini E, Lupini M, Mugnai L, Raspanti S, Mannaioni PF: Polydeoxyribonucleotides and nitric oxide release from guinea-pig hearts during ischaemia and reperfusion. Brit J Pharmacol 115: 629–635, 1995
Node K, Kitakaze M, Kosaka H, Komamura K, Minamino T, Tada M, Inoe M, Hori M, Kamada T: Plasma nitric oxide end products are increased in the ischemic canine heart. Biochem Biophys Res Commun 211: 370–374, 1995
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Komarov, A.M., Kramer, J.H., Mak, I.T. et al. Epr detection of endogenous nitric oxide in postischemic heart using lipid and aqueous-soluble dithiocarbamate-iron complexes. Mol Cell Biochem 175, 91–97 (1997). https://doi.org/10.1023/A:1006897530800
Issue Date:
DOI: https://doi.org/10.1023/A:1006897530800