Abstract
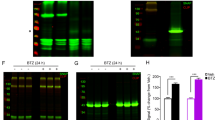
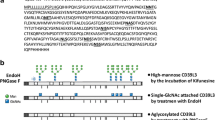
UV cross-linking studies of the natriuretic pepti de receptor- B (NPR-B )using radio labeled C-type natriuretic peptide (CNP) indicate that onlyfully glycosylated receptors are capable of binding ligand. We thereforeused site-directed mutagenesis to determine which potential glycosylationsites are occupied by carbohydrate, and the relevant mutants werecharacterized in order to understand the function of carbohydrate additionat those sites. Our results suggest that five of seven potential N-linkedglycosylation sites are modified. In addition, mutation of asparagine 24results in a loss of ~90% of receptor activity. This mutant isexpressed at levels comparable to the wild-type receptor, and its activityis not significantly different from that of wild-type NPR-B in terms of EC50for CNP. Ligand binding studies on this mutant further show that althoughthere is no change in affinity for ligand, ~90% of receptor bindingis lost. These data suggest that many of the mutant receptors are simply notproperly folded. Our results indicate that glycosylation of asparagine 24 ofNPR-B receptors may be critical for the formation of a competent ligandbinding domain.
Similar content being viewed by others
References
Jamison R, Canaan-Kuhl, S, Pratt R: The natriuretic peptides and their receptors. Am J Kid Dis 20: 519–530, 1992
Drewett JG, Garbers DL: The family of guanylyl cyclase receptors and their ligands. Endo Rev 15(2): 135–162, 1994
Brenner BM, Ballerman BJ, Gunning ME, Zeidel ML: Diverse biological actions of atrial natriuretic peptides. Physiol Rev 70: 665–669, 1990
Needleman P, Blaine EH, Greenwald JE, Michener ML, Saper CB, Stockman PT, Tolunay HE: The biochemical pharmacology of atrial peptides. Ann Rev Pharmacol Toxicol 29: 23–54, 1989
Babinski K, Haddad P, Vallerand D, McNicoll N, De Léan A, Ong H: Natriuretic peptides inhibit cicotine-induced whole-cell currents and catecholamine secretion in bovine chromaffin cells: Evidence for the involvement of the atrial natriuretic factor R2 receptors. J Neurochem 64: 1080–1087, 1995
Samson WK, Huang F-LS, Fulton RG: C-type natriuretic peptide mediates the hypothalamic actions of the natriuretic peptides inhibit luteinizing hormone secretion. Endocrin 132: 504–509, 1993
Shimekaki W, Ohta S, Nagata K: C-type natriuretic peptide stimulates secretion of growth hormone from rat pituitary-derived GH3 cells via a cyclic-GMP-mediated pathway. Eur J Biochem 222: 645–650, 1994
Trachte GJ, Kanwal S, Elmquist BJ, Zeigler RJ: C-type natriuretic peptide neuromodulates via ‘clearance’ receptors. Am J Physiol 268: C978–C984, 1995
Tsutsui M, Yanagihara N, Minami K, Kobayashi H, Nakashima Y, Kuroiwa A, Izumi F: C-type natriuretic peptide stimulates catecholamine synthesis through the accumulation of cyclic GMP in cultured bovine adrenal medullary cells. J Pharmacol Exp Ther 268(2): 584–589, 1994
Furuya M, Yoshida M, Hayashi Y, Ohnuma N, Minamino N, Kangawa K, Matsuo H: C-type natriuretic peptide is a growth inhibitor of rat vascular smooth muscle cells. Biochem Biophys Res Comm 177: 927–931, 1991
Kariya K, Kawahara Y, Araki S, Fukuzaki H, Takai Y: Antiproliferative action of cyclic GMP-elevating vasodilators in cultured rabbit aortic smooth muscle cells. Atherosclerosis 80: 143–147, 1989
Porter JG, Catalano R, McEnroe G, Lewicki JA, Protter AA: C-type natriuretic peptide inhibits growth factor-dependent DNA synthesis in smooth muscle cells. Am J Physiol 263: C1001–C1006, 1992
Stingo A, Clavell A, Aarhus L, Burnett J: Cardiovascular and renal action of C-type natriuretic peptide. Am J Physiol 262: H308–H312, 1992
Schultz S, Singh S, Bellet RA, Singh G, Tubb J, Chin H, Garbers D: The primary structure of a plasma membrane guanylate cyclase demonstrates diversity within this new receptor family. Cell 58: 1155–1162, 1989
Chang M-S, Lowe D, Lewis M, Hellmiss R, Chen E, Goeddel DV: Differential activation by atrial and brain natriuretic peptides of two different receptor guanylyl cyclases. Nature 341: 68–72, 1989
Duda T, Goraczniak RM, Sitaramayya A, Sharma RK: Cloning and expression of an ATP-regulated human retina C-type natriuretic factor receptor guanylyl cyclase. Biochem 32: 1391–1395, 1993
Fenrick R, Babinski K, McNicoll N, Therrien M, Drouin J, De Léan A: Cloning and functional expression of the bovine natriuretic peptide receptor-B (natriuretic factor R1C subtype). Mol Cell Biochem 137: 173–182, 1994
Lowe D, Chang M-S, Hellmiss R, Chen E, Singh S, Garbers DL, Goeddel DV: Human atrial natriuretic peptide receptor defines a new paradigm for second messenger signal transduction. EMBO J 8: 1377–1384, 1989
Chinkers M, Garbers DL, Chang M-S, Lowe D, Chin H, Goeddel DV, Schulz S: A membrane form of guanylyl cyclase is an atrial natriuretic peptide receptor. Nature 38: 78–83, 1989
Pandey KN, Singh S: Molecular cloning and expression of murine guanylyl cyclase/atrial natriuretic factor receptor cDNA. J Biol Chem 265: 12342–12348, 1990
Schoenfeld JR, Sehl P, Quan C, Burnier JP, Lowe DG: Agonist selectivity for three species of natriuretic peptide receptor-A. Mol Pharm 47: 172–180, 1995
Fenrick R, McNicoll N, De Léan A: N-linked glycosylation is critical for natriuretic peptide receptor-B function. Mol Cell Biochem 165: 103–109, 1996
Kornfeld R, Kornfeld S: Assembly of asparagine-linked oligosaccarides. Ann Rev Biochem 54: 631–664, 1985
Cullen BR: Use of eukaryotic expression technology in the functional analysis of cloned genes. Methods Enzymol 152: 684–703, 1987
Fethiere J, Meloche S, Nguyen TT, Ong H, De Léan A: Distinct properties of atrial natriuretic factor receptor subpopulations in epithelial and fibroblast cell lines. Mol Pharm 35: 584–592, 1989
Rondeau J-J, McNicoll N, Lord C, Larose L, Meloche S, Gagnon J, Ong H, De Léan A: Production of polyclonal antibody to the bovine adrenal atrial natriuretic factor R1 receptor. J Receptor Research 12(4): 485–505, 1992
Larose L, McNicoll N, Rondeau J-J, Escher E, De Léan A: Photoaffinity labelling of atrial natriuretic factor (ANF)-R1 receptor by underivatized 125I-ANF. Biochem 267: 379–384, 1990
De Léan A, Munson PJ, Rodbard D: Simultaneous analysis of families of sigmoidal curves: Application to bioassay, radioligand assay, physiological dose-response curves. Am J Physiol 235: E97–El02, 1978
De Léan A, Hancock AA, Lefkowitz RJ: Validation and statistical analysis of a computer modeling method for quantitative analysis of radioligand binding data for mixtures of pharmacological receptor subtypes. Mol Pharm 21: 5–16, 1981
Bennett B, Bennett G, Vitangcol R, Jewett J, Burnier J, Henzel W, Lowe D: Extracellular domain-IgG fusion Proteins for three human natriuretic peptide receptors. J Biol Chem 266: 23060–23067, 1991
Bordo D, Argos P: Suggestions for ‘safe’ residue substitutions in sitedirected mutagenesis. Journal of Molecular Biology 217: 21–29, 1991
Stults JT, O'Connell KL, Garcia C, Wong S, Engel AM, Garbers DL, Lowe DG: The disulfide linkages and glycosylation sites of the human natriuretic peptide receptor-C. Biochem 33: 11372–11381, 1994
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fenrick, R., Bouchard, N., McNicoll, N. et al. Glycosylation of asparagine 24 of the natriuretic peptide receptor-B is crucial for the formation of a competent ligand binding domain. Mol Cell Biochem 173, 25–32 (1997). https://doi.org/10.1023/A:1006855522272
Issue Date:
DOI: https://doi.org/10.1023/A:1006855522272