Abstract
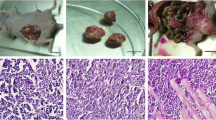
The actual mechanisms responsible for lymph node metastasis in gastric cancer are still unclear. To investigate the mechanisms of lymph node metastasis in gastric cancer, we established a lymph node metastatic model for human scirrhous gastric carcinoma. Lymph node metastasis had frequently developed after orthotopic implantation of OCUM-2M LN derived from a scirrhous gastric cancer cell line, OCUM-2M, which had low capacity for lymph node metastasis. We elucidated the different characteristics including binding ability, migratory capacity and immunoresponses induced by the cell surface molecules of these two cell lines. The binding ability to Matrigel and migratory capacity of OCUM-2M LN cells were significantly greater than those of OCUM-2M cells. On flow cytometric analysis, both OCUM-2M and OCUM-2M LN cells strongly expressed HLA-I (99.5 and 97.1%) and LFA-3 (76.6 and 99.2%) in level of expression between the two cell lines, but neither cell line expressed HLA-II (0.0 and 0 .0%), B7-1 (0.0 and 0.0%) or B7-2 (0.4 and 0.3%). ICAM-1 expression in OCUM-2M LN cells was weaker (0.7%) than that in OCUM-2M cells (36.8%). Strong adhesiveness and cytotoxicity of mononuclear lymphocytes for OCUM-2M cells were observed in adhesion and cytotoxic assays, both of which were significantly decreased by the addition of anti-ICAM-1 antibodies. On the other hand, the adhesiveness and cytotoxicity of OCUM-2M LN cells were significantly less than those of OCUM-2M cells, and were not affected by the addition of anti-ICAM-1 antibodies. These findings suggest that decreased ICAM-1 expression in a new gastric cancer cell line with a high rate of lymph node metasta-sis may in turn decrease immune responses mediated through LFA-1-dependent effector cell adhesion, and that this escape from the immunosurveillance system may be one of the factors inducing lymph node metas-tasis. In conclusion, we established a gastric cancer cell line, OCUM-2M LN, with a high rate of lymph node metasta sis. An in vivo lymph node-metastatic model with this cell line should be useful for analysing the mech-anism and therapeutic approach of lymph node metastasis. © Rapid Science Ltd.
Similar content being viewed by others
References
Ishii H, Gouchi A and Orita K, 1994, The enhancement of cell surface ICAM-1 and HLA Class I antigens in human gastric cancer cell lines by IFN-gamma. Acta Med Okayama, 48, 73-9.
Makgoba MW, Sanders ME, Luce GEG, et al. 1988, ICAM-1 a ligand for LFA-1 dependent adhesion of B, T and myeloid cells. Nature, 331, 86-8.
Marlin SD and Springer TA, 1987, Purified intercellular adhesion molecule (ICAM-1) is a ligand for lymphocyte function-associated antigen (LGFA-1). Cell, 51, 813-19.
Rothelin R, Dustin ML, Marlin SD and Springer TA, 1986, A human intercellular adhesion molecule (ICAM-1) distinct from LFA-1. J Immunol, 137, 1270-4.
Yashiro M, Chung YS, Nishimura S, Inoue T and Sowa M, 1995, Establishment of two new scirrhous gastric carcinoma. Br J Cancer, 72, 1200-10.
Cabanas C and Hogg N, 1991, Lymphocyte-fibroblast adhesion. A useful model for analysis of the interaction of the leucocyte integrin LFA-1 with ICAM-1. FEBS, 292, 284-8.
Kumegawa H, 1994, The relationship between expression of ICAM-1 on gastric carcinoma and TIL. J Kurume Med Assoc, 57, 233-44.
Ishihara S, Nakajima T, Ota K, et al. 1994, The analysis of prognosis of patients with gastric cancer with the nodal (N, n) and the number of involved lymph nodes. Jpn J Cancer Chemother, 21, 1758-61.
Makino M, Moriwaki S and Yonekawa M, 1991, Prognostic significance of the metastatic lymph nodes in patients with gastric cancer. J Surg Oncol, 47, 12-16.
Takeichi M, 1993, Adhesion and metastasis. Oncologia, 26, 149-62.
Ogoshi K, Tajima T, Mitomi T and Tsuji K, 1994, HLADR4 antigen and lymph node metastases in poorly differentiated adenocarcinoma of the stomach. Cancer, 732250-2.
Black M, Opler S and Speer F, 1956, Structural representation of tumor-host relationships in gastric carcinoma. Surg Gynecol Obstet, 102, 599-603.
Good RA, 1991, Experiments of nature in the development of modern immunology. Immunol Today, 12, 283-6.
Herberman RB and Oltaldo JR, 1981, Natural killer cells; their role defense against disease. Science, 214, 24-30.
Springer TA, 1990, Adhesion receptor in immune system. Nature, 346, 425-34.
Springer TA, Dustin ML, Kishimoto TK and Marlin SD, 1987, The molecules. Cell adhesion receptors of the immune system. Ann Rev Immunol, 5, 223-52.
Damale NK, 1992, Differential costimulatory effects of adhesion molecules B7, ICAM-1, LFA-3, and VCAM-on resting and antigen-primed CD4+ T lymphocytes. J Immunol, 148.
Hatchcock KS, Laszlo G, Pucillo C, Linsley P and Hodes RJ, 1994, Comparative analysis of B7-1 and B7-2 costimulatory ligands: expression and function. J Exp Med, 180, 631-40.
Hummphries MJ, Olden K and Yamda K, 1981, A synthetic peptide from fibronectin inhibits experimental metastases of murine melanoma cells. Science, 233, 467-70.
Hynes RO, 1987, Integrins: a family of cell surface receptors. Cell, 48, 549-54.
Iwamoto Y, Robey FA, Graf J, et al. 1987, YIGSR, a synthetic laminin pentapeptide, inhibits experimental metastases formation. Science, 238, 1132-4.
Kishimoto TK, Larson RS, Corbi AL, et al. 1989, Leucocyte integrins. In: Springer TA, Anderson DC, Rosenthal AS, Rothline R, eds. Leucocyte Adhesion Molecules. New York: Springer-Verlag, pp. 7-43.
Davignon D, Martz E, Renolds T, Kurzinger K and Springer TA, 1981, Monoclonal antibody to a novel lymphocyte function-associated antigen (LFA-1): Mechanism of blocking of T lymphocyte mediated killing and effects on other T and B lymphocytes functions. J Immunol, 127, 590-5.
Krensky AM, Sanchez-Madrid F, Robbins E, et al. 1983, The functional significance, distribution and structure of LFA-1, LFA-2 and LFA-3: Cell surface antigens associated with CTL-target interactions. J Immunol, 131, 611-16.
Sanchez-Madrid F, Nagy JA, Robbins E, Simon P and Springer TA, 1983, A human leucocytes differentiation family with distinct alpha subunits and a common beta subunit: The lymphocyte function-associated antigen (LFA-1), the C3bi complement receptor (OKM1/ Mac1) and the p150.95 molecule. J Exp Med, 158, 1785-803.
Dustin ML, Rothlein R, Bhan AK, Dinarello CA and Springer TA, 1986, Induction by IL-1 and interferongamma. Tissue distribution, biochemistry, and function of a natural adherence molecule (ICAM-1). J Immunol, 137, 245-54.
Siu G, Hendric SM and Brian AA, 1989, Isolation of the murine intercellular adhesion molecule 1 (ICAM-1) gene. J Immunol, 143, 3813-20.
Hildreth JEK, Gotch FM, Hildreth PDK and McMichael AJ, 1983, A human lymphocytes-associated antigen involved in cell-mediated lympholysis. Eur J Immunol, 13, 202-8.
Springer TA, Dustin ML, Kishimoto TK and Marlin SD, 1987, The molecule. Cell adhesion receptors of the immune system. Ann Rev Immunol, 5, 223-52.
Marz E, 1987, LFA-1 and other accessory molecules functioning in adhesion of T and B lymphocytes. Hum Immunol, 18, 3-37.
Nishimura T and Itoh T, 1988, Higher level expression of lymphocyte function associated antigen-1 (LFA-1) in vivonatural killer cells. Eur J Immunol, 182077-80.
Staunton DE, Merluzzi VJ, Rothlein R, Barton R, Marlil SD and Springer TA, 1989, A cell adhesion molecule, ICAM-1, is the major surface receptor for rhinoviruses. Cell, 56, 849-53.
Teshigawara K, Okubo Y, Sugie K and Uchida A, 1994, The regulation of NK cell activity through LFA-1 and p58. Clin Immunol, 26, 629-35.
Billaud M, Alain C, Seigneurin JM and Lenoir GM, 1987, LFA-1, LFA-3 and ICAM-1 expression in Burkitt's lymphoma. Lancet, 2, 1327-8.
Gregory CD, Murray RJ, Edwardis CF and Rickinson AB, 1988, Down-regulation of cell adhesion molecules LFA-3 and ICAM-1 in Epstein-Barr virus-positive Burkitt's lymphoma underlies tumor cell escape from virus-specific T cell surveillance. J Exp Med, 167, 1811-24.
Johnson JP, Stade BG, Holzmann B, Schwable W and Riethmuller G, 1988, De novoexpression of intercellular adhesion molecule 1 in melanoma correlates with increased risk of metastasis. Proc Natl Acad Sci, 86, 641-4.
Maio M, Gulwani B, Langer JA, et al. 1989, Modulation by interferons of HLA antigen, high-molecular-weight melanoma-associated antigen, and intercellular adhesion molecule 1 expression by cultured melanoma cell with different metastatic potential. Cancer Res, 49, 2980-7.
Tomita Y, Nishiyama T, Watanabe H, Fujiwara M and Sato S, 1989, Expression of intercellular adhesion molecule 1 (ICAM-1) on renal-cell cancer: possible significance in host immune response. Int J Cancer, 43, 768-73.
Minervini R, Panichi V, Vigano L, et al. 1994, Cellular expression of lymphocyte function-associated antigen-1 and intercellular adhesion molecule-1 in normal kidney structures and in renal cancer. Eur Urol, 26, 103-6.
Shibata M, Ando K, Amano S and Kurosu Y, 1996, Local expression and circulating form of ICAM-1 in colorectal cancer. Ann Cancer Res Ther, 5, 29-33.
Barton RW, Rothlein R, Ksiazek J and Kennedy C, 1989, The effect of antiintercellular adhesion molecule-1 on phorbol-ester-induced rabbit lung inflammation. J Immunol, 143, 1278-82.
Trejdosiewics LK, 1992, Intestinal intraepithelial lymphocytes and lymphoepithelial interactions in the human gastrointestinal mucosa. Immunol Left, 32, 13-20.
Malzia G, Calabrese A, Cottone M, et al. 1991, Expression of leukocyte adhesion molecules by mucosal mononuclear phagocytes in inflammatory bowel disease. Gastroenterology, 100, 150-9.
Tsujisaki M, Imai K, Hirata H, et al. 1991, Detection of circulating intercellular adhesion molecule-1 antigen in malignant diseases. Clin Exp Immunol, 85, 3-8.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fujihara, T., Sawada, T., Hirakawa-YS Chung, K. et al. Establishment of lymph node metastatic model for human gastric cancer in nude mice and analysis of factors associated with metastasis. Clin Exp Metastasis 16, 389–398 (1998). https://doi.org/10.1023/A:1006550704555
Issue Date:
DOI: https://doi.org/10.1023/A:1006550704555