Abstract
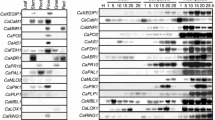
Soybean cell cultures (cv. Williams 82) respond to Pseudomonas syringae bacteria expressing the avirulence gene AvrA with a hypersensitive reaction, a programmed cell death (PCD) of plant cells to pathogen attack. This PCD is under control of salicylic acid (SA) via an unknown mechanism. In the presence of low concentrations of SA, the cells undergo a very rapid cell death, which needs only half of the time required for the normal hypersensitive reaction (HR). Northern blot studies for defence-related genes show that the expression of many of these genes is tightly linked to the status of the cell death program rather than to pathogen-derived elicitors. Thus the expression is much faster in the SA-accelerated PCD than in the normal hypersensitive reaction. In contrast, other pathogen-responsive genes are induced independently of the speed of PCD, indicating a divergent signalling mechanism. The production of reactive oxygen species during the oxidative burst of bacteria-inoculated soybean cells is slightly enhanced in the presence of SA but occurs at the same time as in untreated cells, suggesting that SA exhibits the control of the PCD downstream of the oxidative burst. Consistent with these findings a HR-specific marker gene is neither directly induced by H2O2 or SA. However, this gene shows a high expression in the regular HR and is induced much faster in the SA-accelerated PCD.
Similar content being viewed by others
References
Altschul, S.F., Gish, W., Miller, W., Myers, E.W. and Lipman, D.J. 1990. Basic local alignment search tool. J. Mol. Biol. 215: 403–410.
Alvarez, M.E., Pennell, R.I., Meijer, P.J., Ishikawa, A., Dixon, R.A. and Lamb, C. 1998. Reactive oxygen intermediates mediate a systemic signal network in the establishment of plant immunity. Cell 92: 773–784.
Anderson, M.D., Chen, Z. and Klessig, D.F. 1998. Possible involvement of lipid peroxidation in salicylic acid-mediated induction of PR-1 gene expression. Phytochemistry 47: 555–566.
Bennett, M., Gallagher, M., Fagg, J., Bestwick, C., Paul, T., Beale, M. and Mansfield, J. 1996. The hypersensitive reaction, membrane damage and accumulation of autofluorescent phenolics in lettuce cells challenged by Bremia lactucae. Plant J. 9: 851–865.
Bowles, D.J. 1990. Defense-related proteins in higher plants. Annu. Rev. Biochem. 59: 873–907.
Brisson, L.F., Tenhaken, R. and Lamb, C.J. 1994. Function of oxidative cross-linking of cell wall structural proteins in plant disease resistance. Plant Cell 6: 1703–1712.
Chamnongpol, S., Willekens, H., Moeder, W., Langebartels, C., Sandermann, H. Jr., Van Montagu, M., Inzé, D. and Van Camp, W. 1998. Defense activation and enhanced pathogen tolerance induced by H2O2 in transgenic tobacco. Proc. Natl. Acad. Sci. USA 95: 5818–5823.
Chomczynski, P. and Sacchi, N. 1987. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162: 156–159.
Church, G.M. and Gilbert, W. 1984. Genomic sequencing. Proc. Natl. Acad. Sci. USA 81: 1991–1995.
Crowell, D.N. and Amasino, R.M. 1991. Induction of specific mRNAs in cultured soybean cells during cytokinin or auxion starvation. Plant Physiol. 95: 711–715.
Dangl, J.L., Dietrich, R.A. and Richberg, M.H. 1996. Death don't have no mercy: cell death programs in plant-microbe interactions. Plant Cell 8: 1793–1807.
Delaney, T.P., Uknes, S., Vernooij, B., Friedrich, L., Weymann, K., Negrotto, D., Gaffney, T., Gut-Rella, M., Kessmann, H., Ward, E. and Ryals, J.A. 1994. A central role of salicylic acid in plant disease resistance. Science 266: 1247–1250.
Delledonne, M., Xia, Y., Dixon, R.A. and Lamb, C. 1998. Nitric oxide functions as a signal in plant disease resistance. Nature 394: 585–588.
Draper, J. 1997. Salicylate, superoxide synthesis and cell suicide in plant defence. Trends Plant Sci. 2: 162–165.
Droog, F., Spek, A., van der Kooy, A., de Ruyter, A., Hoge, H., Libbenga, K., Hooykaas, P. and van der Zaal, B. 1995. Promotor analysis of the auxin-regulated tobacco glutathione S-transferase genes Nt103-1 and Nt103-35. Plant Mol. Biol. 29: 413–429.
Du, H. and Klessig, D.F. 1997. Role for salicylic acid in the activation of defense responses in catalase-deficient transgenic tobacco. Mol. Plant-Microbe Interact. 10: 922–925.
Durner, J., Shah, J. and Klessig, D.F. 1997. Salicylic acid and disease resistance in plants. Trends Plant Sci. 2: 266–274.
Ebel, J. and Scheel, D. 1992. Elicitor recognition and signal transduction. In: T. Troller and F. Meins Jr. (Eds.) Genes Involved in Plant Defense, Springer-Verlag, Vienna, pp. 183–205.
Esterbauer, H. and Cheeseman, K.H. 1990. Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. Meth. Enzymol. 186: 407–421.
Friedrich, L., Vernooij, B., Gaffney, T., Morse, A. and Ryals, J. 1995. Characterization of tobacco plants expressing a bacterial salicylate hydroxylase gene. Plant Mol. Biol. 29: 959–968.
Glazener, J.A., Orlandi, E.W. and Baker, C.J. 1996. The active oxygen response of cell suspensions to incompatible bacteria is not sufficient to cause hypersensitive cell death. Plant Physiol. 110: 759–763.
Goodman, R.N. and Novacky, A.J. 1994. The Hypersensitive Reaction in Plants to Pathogens. A Resistance Phenomenon. APS Press, St. Paul, MN.
Habereder, H., Schröder, G. and Ebel, J. 1989. Rapid induction of phenylalanine ammonia-lyase and chalcone synthase mRNAs during fungus infection of soybean (Glycine max L.) roots or elicitor treatment of soybean cell cultures at the onset of phytoalexin synthesis. Planta 177: 58–65.
Hammond-Kosack, K.E., Tang, S., Harrison, K., Jones, J.D.G. 1998. The tomato Cf-9 disease resistance gene functions in tobacco and potato to confer responsiveness to the fungal avirulence gene product Avr9. Plant Cell 10: 1251–1266.
Jabs, T., Dietrich, R.A. and Dangl, J.L. 1996. Initiation of runaway cell death in an Arabidopsis mutant by extracellular superoxide. Science 273: 1853–1856.
Kauss, H., Theisinger-Hinkel, E., Mindermann, R. and Conrath, U. 1992. Dichlorisonicotinic and salicylic acid, inducers of systemic acquired resistance, enhance fungal elicitor responses in parsley cells. Plant J. 2: 655–660.
Kauss, H. and Jeblick, W. 1995. Pretreatment of parsley suspension cultures with salicylic acid enhances spontaneous and elicited production of H2O2. Plant Physiol. 108: 1171–1178.
Keen, N.T. and Buzzell, R.I. 1991. New disease resistance genes in soybean against Pseudomonas syringae pv. glycinea: evidence that one of them interacts with a bacterial elicitor. Theor. Appl. Genet. 81: 133–138.
Kiedrowski, S., Kawalleck, P., Hahlbrock, K., Somssich, I.E. and Dangl, J.L. 1992. Rapid activation of a novel plant defense gene is strictly dependent on the Arabidopsis RPM1 disease resistance locus. EMBO J. 11: 4677–4684.
Lamb, C. and Dixon, R.A. 1997. The oxidative burst in plant disease resistance. Annu. Rev. Plant Physiol. Plant Mol. Biol. 48: 251–275.
Levine, A., Tenhaken, R., Dixon, R.A. and Lamb, C.J. 1994. H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79: 583–593.
Levine, A., Pennell, R.I., Alvarez, M.E., Palmer, R. and Lamb, C. 1996. Calcium-mediated apoptosis in a plant hypersensitive disease resistance response. Curr. Biol. 6: 427–437.
Mauch-Mani, B. and Slusarenko, A.J. 1996. Production of salicylic acid precursors is a major function of phenylalanine ammonialyase in the resistance of Arabidopsis to Peronospora parasitica. Plant Cell 8: 203–212.
Meier, B.M., Shaw, N. and Slusarenko, A.J. 1993. Spatial and temporal accumulation of defense gene transcripts in bean (Phaseolus vulgaris) leaves in relation to bacteria-induced hypersensitive cell death. Mol. Plant-Microbe Interact. 6: 453–466.
Mittler, R., Shulaev, V., Seskar, M. and Lam, E. 1996. Inhibition of programmed cell death in tobacco plants during a pathogeninduced hypersensitive response at low oxygen pressure. Plant Cell 8: 1991–2001.
Orlandi, E.W., Hutcheson, S.W. and Baker, C.J. 1992. Early physiological responses associated with race-specific recognition in soybean leaf tissue and cell suspensions treated with Pseudomonas syringae pv. glycinea. Physiol. Mol. Plant Path. 40: 173–180.
Pennell, R.I. and Lamb, C. 1997. Programmed cell death in plants. Plant Cell 9: 1157–1168.
Rao, M.V., Paliyath, G., Ormrod, D.P., Murr, D.P. and Watkins, C.B. 1997. Influence of salicylic acid on H2O2 production, oxidative burst, and H2O2-metabolizing enzymes. Plant Physiol. 115: 137–149.
Rate, D.N., Cuenca, J.V., Bowman, G.R., Guttman, D.S. and Greenberg, J.T. 1999. The gain-of-function Arabidopsis acd6 mutant reveals novel regulation and function of the salicylic acid signaling pathway in controlling cell death, defenses and cell growth. Plant Cell 11: 1695–1708.
Sánchez-Casas, P. and Klessig, D.F. 1994. a salicylic acid-binding activity and a salicylic acid-inhibitable catalase activity are present in a variety of plant species. Plant Physiol 106: 1675–1679.
Seehaus, K. and Tenhaken, R. 1998. Cloning of genes by mRNA differential display induced during the hypersensitive reaction of soybean after inoculation with Pseudomonas syringae pv. glycinea. Plant Mol. Biol. 38: 1225–1234.
Shirasu, K., Nakajima, H., Krishnamachari-Rajasekhar, V.K., Dixon, R.A. and Lamb, C. 1997. Salicylic acid potentiates an agonist-dependent gain control that amplifies pathogen signals in the activation of defense mechanisms. Plant Cell 9: 261–270.
Tenhaken, R., Levine, A., Brisson, L.F., Dixon, R.A. and Lamb, C. 1995. Function of the oxidative burst in hypersensitive disease resistance. Proc. Natl. Acad. Sci. USA 92: 4158–4163.
Tenhaken, R. and Rübel, C. 1997. Salicylic acid is needed in hypersensitive cell death in soybean but does not act as a catalase inhibitor. Plant Physiol. 115: 291–298.
Thomma, B.P.H.J., Eggermont, K., Penninckx, I.A.M.A., Mauch-Mani, B., Vogelsang, R., Cammue, R.P.A. and Broekaert, W.F. 1998. Separate jasmonate-dependent and salicylate-dependent defense-response pathways in Arabidopsis are essential for resistance to distinct microbial pathogens. Proc. Natl. Acad. Sci. USA 95: 15107–15111.
Thulke, O. and Conrath, U. 1998. Salicylic acid has a dual role in the activation of defence-related genes in parsley. Plant J. 14: 35–42.
Walter, M.H., Liu, J.W., Grand, C., Lamb, C.J. and Hess, D. 1990. Bean pathogenesis-related (PR) proteins deduced from elicitorinduced transcripts are members of a ubiquitous new class of conserved PR proteins including pollen allergens. Mol. Gen. Genet. 222: 353–360.
Walter, M.H., Liu, J.W., Wuenn, J. and Hess, D. 1996. Bean ribonuclease-like pathogenesis-related protein genes (Ypr10) display complex patterns of developmental, dark-induced and exogenous-stimulus-dependent expression. Eur. J. Biochem. 239: 281–293.
Whalen, M.C., Innes, R.W., Bent, A.F. and Staskawicz, B.J. 1991. Identification of Pseudomonas syringae pathogens of Arabidopsis and a bacterial locus determining avirulence on both Arabidopsis and soybean. Plant Cell 3: 49–59.
Weymann, K., Hunt, M., Uknes, S., Neuenschwander, U., Lawton, K., Steiner, H.Y. and Ryals, J. 1995. Suppression and restoration of lesion formation in Arabidopsis lsd mutants. Plant Cell 7: 2013–2022.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ludwig, A., Tenhaken, R. Defence gene expression in soybean is linked to the status of the cell death program. Plant Mol Biol 44, 209–218 (2000). https://doi.org/10.1023/A:1006439504748
Issue Date:
DOI: https://doi.org/10.1023/A:1006439504748