Abstract
The relationship between coagulation cascade activation and glioma cell proliferation was examined. The human glioma cell lines T98G, TM-1 and normal human astrocyte cell strain (NHA) were examined. Using anti-tissue factor (TF) antibody, immunocytochemical detection of TF antigen was obtained in both cell lines and cell strain. TF antigen in cell lysates was also measured by enzyme linked immunosorbent assay (ELISA). In a one-stage clotting assay, T98G, TM-1 and NHA revealed procoagulant activity (PCA) in normal human plasma and factor VII deficient plasma. PCA in normal human plasma was significantly inhibited by both inhibitory anti-TF antibody and cysteine protease inhibitor HgCl2. This result indicates that T98G, TM-1 and NHA cells express not only TF but also cancer procoagulant (CP) at the same time.
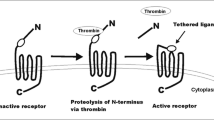
In a cell proliferation assay, thrombin induced proliferation in T98G and TM-1 cells in a dose-dependent fashion and in NHA cell in a bell-shaped fashion. This mitogenic stimulant was inhibited by the specific thrombin inhibitor hirudin. The combinations of coagulation factors II, V, and X with or without factor VII induced proliferation in T98G, TM-1, and NHA cells. The maximal mitogenic stimulatory effects were larger in glioma cells than in NHA. These mitogenic stimulatory effects were also inhibited by hirudin. Each coagulation factor on its own or in any other combination of coagulation factors had no proliferative effect. Thus, these mitogenic stimulatory effects were considered to be the effect of thrombin.
In conclusion, T98G and TM-1 human glioma cells express two different types of procoagulants TF and CP. In the presence of coagulation factors, these glioma cells can generate thrombin and this thrombin generation is capable of inducing glioma cell proliferation in vitro.
Similar content being viewed by others
References
Gordon SG: Cancer cell procoagulants and their role in malignant disease. Semin Thromb Hemostasis 18: 424-433, 1992
Murray JC: Coagulation and cancer. Br J Cancer 64: 422-424, 1991
Rao LVM: Tissue factors as a tumor procoagulant. Cancer Metastasis Res 11: 249-266, 1992
Dvorak HF: Tumors: wounds that do not heal: similarties between tumor stroma generation and wound healing. N Engl J Med 315: 1650-1659, 1986
Esumi N, Fan D, Filder IJ: Inhibition of murine melanoma experimental metastasis by recombinant desulfatohirudin, a highly specific thrombin inhibitor. Cancer Res 51: 4549-4556, 1991
Nierodzic ML, Plotkin A, Kajumo F, Karpatkin S: Thrombin stimulates tumor-platelet adhesion in vitro and metastasis in vivo. J Clin Invest 87: 229-236, 1991
Wojtukiewicz MZ, Tang DG, Ciarelli JJ, Nelson KK, Walz DA, Diglio CA, Mammen EF, Honn KV: Thrombin increases the metastatic potential of tumor cells. Int J Cancer 54: 793-806, 1993
Donati MB, Passerini CG, Casali B, Falanga A, Vannotti P, Fossati G, Semerano N, Gordon SG: Cancer procoagulant in human tumor cells: evidence from melanoma patients. Cancer Res 46: 6471-6474, 1986
Drake TA, Morrissey JH, Edgington TS: Selective cellular expression of tissue factor in human tissues. Am J Pathol 134: 1087-1097, 1989
Fleck C, Rao LVM, Rapaport SI, Varki N: Localization of human tissue factor antigen by immunostaining with monospecific, polyclonal anti-human tissue factor antibody. Tromb Res 59: 421-437, 1990
Eddleston M, Torre JC, Oldstone MBA, Loskutoff DJ, Edgington TS, Macknian N: Astrocytes are the primary source of tissue factor in the murine central nervous system. J Clin Invest 92: 349-358, 1993
Callander NS, Varki N, Rao LVM: Immunohistochemical identification of tissue factor in solid tumors. Cancer 70: 1194-1201, 1992
Hu T, Bach RR, Horton R, Konigsberg WH, Todd MB: Synthesis of tissue factor messenger RNA and procoagulant activity in breast cancer cells in response to serum stimulation. Tromb Res 72: 155-168, 1993
Idell S, Pueblitz S, Emri S, Gungen Y, Gray L, Kumar A, Holiday D, Koenig KB, Johnson AR: Regulation of fibrin deposition by malignant mesothelioma. Am J Pathol 147: 1318-1329, 1995
Saito T, Koyama T, Nagata K, Kamiyama R, Hirosawa S: Anticoagulant effects of retinoic acid on leukemia cells. Blood 87: 657-665, 1996
Wojtukiewic MZ, Zacharski LR, Memoli VA, Kisiel W, Kudryk BJ, Rousseau SM, Stump DC: Abnormal regulation of coagulation/fibrinolysis in small cell carcinoma of the lung. Cancer 65: 481-485, 1990
Falanga A, Alessio MG, Donati MB, Barbui T: A new procoagulant in acute leukemia. Blood 71: 870-875, 1988
Gordon SG: Cancer procoagulant. Methods Enzymol 244: 568-583, 1994
Pohjanpelto P: Stimulation of DNA synthesis in human fibroblasts by thrombin. J Cell Physiol 95: 189-194, 1978
Bachhuber BG, Sarembock IJ, Gimple LW, Mcnamara CA, Owens GK: Thrombin-induced mitogenesis in cultured aortic smooth muscle cells requires prolonged thrombin exposure. Am J Physiol 268: C1141-C1147, 1995
Cavanaugh KP, Gurwitz D, Cunningham DD, Bradshaw RA: Reciprocal modulation of astrocyte stellation by thrombin and protease nexin-1. J Neurochem 54: 1735-1743, 1990
Gurwitz D, Cunningham DD: Thrombin modulates and reverses neuroblastoma neurite outgrowth. Proc Natl Acad Sci USA 85: 3440-3444, 1988
Grabham P, Cunningham DD: Thrombin receptor activation stimulates astrocyte proliferation and reversal of stellation by distinct pathways: involvement of tyrosine phosphorylation. J Neurochem 64: 583-591, 1995
Nelson RB, Siman R: Thrombin and its inhibitors regulate morphological and biochemical differentiation of astrocytes in vitro. Dev Brain Res 54: 93-104, 1990
Rasmussen UB, Vouret-Craviari V, Jallat S, Schlesinger Y, Pages G, Pavirani A, Lecocq JP, Pouyssegur J, Van Obberghen-Schilling E: cDNA cloning and expression of a hamster α-thrombin receptor coupled to Ca2+ mobilization. FEBS Lett 288: 123-128, 1991
Vu TH, Hung DT, Wheaton VI, Coughlin SR: Molecular cloning of a functional thrombin receptor reveals a novel proteolytic mechanism of receptor activation. Cell 64: 1057-1068, 1991
Nakamura S, Goto A, Takemoto S, Mizuguchi J, Funatsu A: Monoclonal antibodies to human tissue factor. 'Abstract' Thromb Haemost 65: 1216, 1991
Asakura H, Kamikubo Y, Goto A, Shiratori H, Jokaji H, Saito M, Uotani C, Nakamura S: Role of tissue factor in disseminated intravascular coagulation. Thromb Res 80: 217-224, 1995
Suefuji H, Ogawa H, Yasue H, Kaikita K, Soejima H, Motoyama T, Mizuno Y, Oshima S, Saito T, Tsuji I, Kumeda K, Kamikubo Y, Nakamura S: Increased plasma tissue factor levels in acute myocardinal infarction. Am Heart J 134: 253-259, 1997
Imamura T, Iyama K, Takeya M, Kanbara T, Nakamura S: Role of macrophage tissue factor in development of delayed hyper-sensitivity reaction in monkey skin. Cell Immunol 152: 614-622, 1993
Higure A, Okamoto M, Hirata K, Todoroki T, Nagafuchi Y, Takeda S, Itoh H, Osato K, Nakamura S: Macrophages and neutrophils infiltrating into the liver are responsible for tissue factor expression in a rabbit model of acute obstructive Cholangitis. Thromb Haemost 75: 791-795, 1996
Arai K, Horie U, Kurimoto M, Endo S, Hiraga K, Takaku A: A cell line of human malignant astrocytoma producing autocrine growth factor. In Vitro Cell Dev Biol 27A: 606-614, 1991
Kurimoto M, Endo S, Arai K, Horie Y, Nogami K, Takaku A: TM-1 cells from an established human malignant glioma cell line produce PDGF, TGF-α and TGF-β which cooperatively play a stimulatory role for an autocrine growth promotion. J Neuro-Oncol 22: 33-44, 1994
Mosmann T: Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65: 55-63, 1983
Hetland O, Brovold AB, Holme R, Gaudernack G, Prydz H: Thromboplastin (tissue factor) in plasma membranes of human monocytes. Biochem J 228: 735-743, 1985
Maynard JR, Heckman CA, Pitlic FA, Nemerson Y: Association of tissue factor activity with the surface of cultured cells. J Clin Invest 55: 814-824, 1975
Muller M, Flossel C, Haase M, Luther T, Albrecht S, Nawroth PP, Zhang Y: Cellular localization of tissue factor in human breast cancer cell lines. Virchows Arch 64: 265-269, 1993
Donati MB, Falanga A, Consonni R, Alessio MG, Bassan R, Buelli M, Catani L, Pogliani F, Gugliotta L, Masera G, Barbui T: Cancer procoagulant in acute nonlymphoid leukemia: relationship of enzyme detection to disease activity. Thromb Haemost 64: 11-16, 1990
Falanga A, Shaw F, Donati MB, Conssoni R, Barbui T, Gordon S: Inhibition of cancer procoagulant by peptidyl diazomethyl ketones and peptidyl sulfonium salts. Tromb Res 54: 389-398, 1989
Dvorak HF, Senger DR, Dvorak AM, Harvey VS, Mcdonagh J: Regulation of extravascular coagulation by microvascular permeability. Science 227: 1059-1061, 1985
Keck PJ, Hauser SD, Krivi G, Sanzo K, Warren T, Reder J, Connolly DT: Vascular permeability factor, an endothelial cell mitogen related to PDGF. Science 246: 1309-1312, 1989
Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N: Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246: 1306-1309, 1989
Tsai J-C, Goldman CK, Gillespie GY: Vascular endothelial growth factor in human glioma cell lines: induced secretion by EGF, PDGF-BB, and bFGF. J Neurosurg 82: 864-873, 1995
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ogiichi, T., Hirashima, Y., Nakamura, S. et al. Tissue Factor and Cancer Procoagulant Expressed by Glioma Cells Participate in their Thrombin-mediated Proliferation. J Neurooncol 46, 1–9 (2000). https://doi.org/10.1023/A:1006323200001
Issue Date:
DOI: https://doi.org/10.1023/A:1006323200001