Abstract
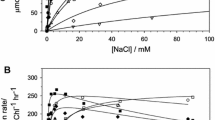
The anion azide, N3 -, has been previously found to be an inhibitor of oxygen evolution by Photosystem II (PS II) of higher plants. With respect to chloride activation, azide acts primarily as a competitive inhibitor but uncompetitive inhibition also occurs [Haddy A, Hatchell JA, Kimel RA and Thomas R (1999) Biochemistry 38: 6104–6110]. In this study, the effects of azide on PS II-enriched thylakoid membranes were characterized by electron paramagnetic resonance (EPR) spectroscopy. Azide showed two distinguishable effects on the S2 state EPR signals. In the presence of chloride, which prevented competitive binding, azide suppressed the formation of the multiline and g = 4.1 signals concurrently, indicating that the normal S2 state was not reached. Signal suppression showed an azide concentration dependence that correlated with the fraction of PS II centers calculated to bind azide at the uncompetitive site, based on the previously determined inhibition constant. No evidence was found for an effect of azide on the Fe(II)QA - signals at the concentrations used. This result is consistent with placement of the uncompetitive site on the donor side of PS II as suggested in the previous study. In chloride-depleted PS II-enriched membranes azide and fluoride showed similar effects on the S2 state EPR signals, including a notable increase and narrowing of the g = 4.1 signal. Comparable effects of other anions have been described previously and apparently take place through the chloride-competitive site. The two azide binding sites described here correlate with the results of other studies of Lewis base inhibitors.
Similar content being viewed by others
References
Berthold DA, Babcock GT and Yocum CF (1981) A highly resolved, oxygen-evolving Photosystem II preparation from spinach thylakoid membranes. FEBS Lett 134: 231–234
Britt RD (1996) Oxygen evolution. In: Ort DR and Yocum CF (eds) Oxygenic Photosynthesis: The Light Reactions, pp 137–164.Kluwer Academic Publishers, Dordrecht, The Netherlands
Cao J and Govindjee (1990) Anion effects on the electron acceptor side of Photosystem II in a transformable cyanobacterium Synechocystis 6803. In: Baltscheffsky M (ed) Current Research in Photosynthesis, Vol I, pp 515–518. Kluwer Academic Publishers, Dordrecht, The Netherlands
Critchley C (1985) The role of chloride in Photosystem II. Biochim Biophys Acta 811: 33–46
Coleman WJ (1990) Chloride binding proteins: mechanistic implications for the oxygen-evolving complex of Photosystem II. Photosynth Res 23: 1–27
Debus RJ (1992) Themanganese and calcium ions of photosynthetic oxygen evolution. Biochim Biophys Acta 1102: 269–352
DeRose V, Latimer MJ, Zimmermann J-L, Mukerji I, Yachandra V, Sauer K and Klein MP (1995) Fluoride substitution in the Mn cluster from Photosystem II: EPR and X-ray absorption spectroscopy studies. Chem Phys 194: 443–459
Diner BA and Petrouleas V (1990) Formation by NO of nitrosyl adducts of redox components of the Photosystem II reaction center. II. Evidence that HCO3¯/CO2 binds to the acceptor-side non-heme iron. Biochim Biophys Acta 1015: 141–149
Franzén L-G, Hansson Ö and Andréasson L-E (1985) The roles of the extrinsic subunits in Photosystem II as revealed by EPR. Biochim Biophys Acta 808: 171–179
Haddy A, Dunham WR, Sands RH and Aasa R (1992) Multifrequency EPR investigations into the origin of the S2-state signal at g = 4 of the O2-evolving complex. Biochim Biophys Acta 1099: 25–34
Haddy A, Hatchell JA, Kimel RA and Thomas R (1999) Azide as a competitor of chloride in oxygen evolution by Photosystem II. Biochemistry 38: 6104–6110.
Katoh S (1972) Inhibitors of electron transport associated with Photosystem II in chloroplasts. Plant Cell Physiol 13: 273–286
Kawamoto K, Mano J and Asada K (1995) Photoproduction of the azidyl radical from the azide anion on the oxidizing side of Photosystem II and suppression of photooxidation of tyrosine Z by the azidyl radical. Plant Cell Physiol 36: 1121–1129
Koulougliotis D, Kostopoulos T, Petrouleas V and Diner BA (1993) Evidence for CN¯ binding at the PS II non-heme Fe2+. Effects on the EPR signal for QA-Fe2 + and on QA/QB electron transfer. Biochim Biophy Acta 1141: 275–282
Kühne H, Szalai VA, Brudvig GW (1999) Competitive binding of acetate and chloride in photosystem II. Biochemistry 38: 6604–6613
Lindberg K and Andréasson L-E (1996) A one-site, two-state model for the binding of anions in Photosystem II. Biochemistry 35: 14259–14267
Lindberg K, Vänngård T and Andréasson, LE (1993) Studies of the slowly exchanging chloride in Photosystem II of higher plants. Photosynth Res 38: 401–408
Ono T, Zimmermann JL, Inoue Y and Rutherford AW (1986) EPR evidence for a modified S-state transition in chloride-depleted Photosystem II. Biochim Biophys Acta 851: 193–201
Ono T, Nakayama H, Gleiter H, Inoue Y and Kawamori A (1987) Modification of the properties of S2 state in photosynthetic O2-evolving center by replacement of chloride with other anions. Arch Biochem Biophys 256: 618–624
Rutherford AW and Zimmermann JL (1984) A new EPR signal attributed to the primary plastosemiquinone acceptor in Photosystem II. Biochim Biophys Acta 767: 168–175
Rutherford AW, Zimmermann JL and Boussac A (1992) Oxygen evolution. In: Barber J (ed) The Photosystems: Structure, Function and Molecular Biology, pp 179–229. Elsevier Science Publishers, Amsterdam
Sanakis Y, Petrouleas V and Diner BA (1994) Cyanide binding at the non-heme Fe2+ of the iron-quinone complex of Photosystem II: At high concentration, cyanide converts the Fe2+ from high (S = 2) to low (S = 0) spin. Biochemistry 33: 9922–9928
Sandusky PO and Yocum CF (1984) The chloride requirement for photosynthetic oxygen evolution: analysis of the effects of chloride and other anions on amine inhibition of the oxygen-evolving complex. Biochim. Biophys. Acta 766: 603–611
Sandusky PO and Yocum CF (1986) The chloride requirement for photosynthetic oxygen evolution: Factors affecting nucleophilic displacement of chloride from the oxygen-evolving complex. Biochim Biophys Acta 849: 85–93
van Vliet P and Rutherford AW (1996) Properties of the chloridedepleted oxygen-evolving complex of Photosystem II studied by electron paramagnetic resonance. Biochemistry 35: 1829–1839
Vermaas WFJ and Rutherford AW(1984) EPRmeasurements on the effects of bicarbonate and triazine resistance on the acceptor side of Photosystem II. FEBS Lett 175: 243–248
Wincencjusz H, Yocum CF and van Gorkom HJ (1999) Activating anions that replace Cl¯ in the O2-evolving complex of Photosystem II slow the kinetics of the terminal step in water oxidation and destabilize the S2 and S3 states. Biochemistry 38: 3719–3725
Yachandra VK, Guiles RD, Sauer K and Klein MP (1986) The state of manganese in the photosynthetic apparatus. 5. The chloride effect in photosynthetic oxygen evolution. Biochim Biophys Acta 850: 333–342
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haddy, A., Allen Kimel, R. & Thomas, R. Effects of azide on the S2 state EPR signals from Photosystem II. Photosynthesis Research 63, 35–45 (2000). https://doi.org/10.1023/A:1006306819002
Issue Date:
DOI: https://doi.org/10.1023/A:1006306819002