Abstract
Cisplatin-induced neuropathy is a major dose-limiting toxicity. Counteraction by amifostine and its metabolite WR1065 may reduce peripheral neurotoxicity in a number of patients. Using the nerve growth factor (NGF)-dependent neurite outgrowth from the PC12 pheochromocytoma cell line as an in vitro assay for neurotoxicity, the protective effects of amifostine and WR1065 against cisplatin action on neurite formation by PC12 cells were studied.
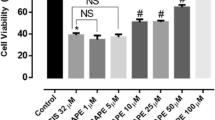
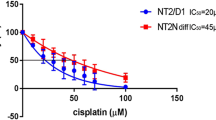
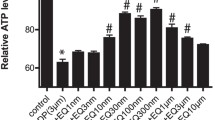
Cisplatin in a concentration of 10 µg/ml significantly decreased the percentage of neurite forming cells from 84% to 40%. Amifostine in doses of 0.4 and 0.8 mM proved to protect significantly against the cisplatin-induced decrease in neurite formation, when co-incubated with cisplatin. Also the metabolite WR1065 protected significantly against cisplatin neurotoxicity in a dose of 0.12 mM. Our results show a significant protection by amifostine and its main metabolite WR1065 against cisplatin-induced neurotoxicity using an in vitro model.
Similar content being viewed by others
References
Roelofs RI, HrusheskyW, Rogin J, Rosenberg L: Peripheral sensory neuropathy and cisplatin chemotherapy. Neurology 34: 934–938, 1984
Gerritsen van der Hoop RG, van der Burg MEL, ten Bokkel Huinink WW, van Houwelingen C, Neijt JP: Incidence of neuropathy in 395 patients with ovarian cancer treated with or without cisplatin. Cancer 66: 1697–1702, 1990
Riggs JE, Ashraf M, Snyder RD, Gutmann L: Prospective nerve conduction studies in cisplatin therapy. Ann Neurol 23: 92–94, 1988
Walsh TJ, Clark AW, Pahrad IM, Green WR: Neurotoxic effects of cisplatin therapy. Arch Neurol 39: 719–720, 1982
Daugaard GK, Petrera J, Trojaborg W: Electrophysiological study of the peripheral and central neurotoxic effects of cisplatin. Acta Neurol Scand 76: 86–93, 1987
Gregg RW, Molepo JM, Monpetit VJA, Mikael NZ, Redmond D, Gadia M, Stewart DJ: Cisplatin neurotoxicity: the relationship between dosage, time, and platinum concentration in neurologic tissues, and morphologic evidence of toxicity. J Clin Oncol 10: 795–803, 1992
Kemp G, Rose P, Lurain J, Berman M, Manetta A, Roullet B, Homesley H, Belpomme D, Glick J: Amifostine pretreatment for protection against cyclophosphamide-induced and 5 cisplatin-induced toxicities: results of a randomized control trial in patients with advanced ovarian cancer. J Clin Oncol 14: 2101–2112, 1996
Spencer CM, Goa KL: Amifostine, a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential as a radioprotector and cytotoxic chemoprotector (Review). Drugs 50: 1001–1031, 1995
Treskes M, Nijtmans L, Fichtinger Schepman AM, van der Vijgh WJF: Cytostatic activity of cisplatin in the presence of WR2721 and its thiol metabolite WR1065 in OVCAR-3 human ovarian cancer cells as compared to V79 fibroblasts. Anticancer Res 12: 2261–2266, 1992
Nakamura J, Shaw LM, Brown DQ: Hydrolysis of WR2721 by mouse liver cell fractions. Radiat Res 109: 143–152, 1987
Yuhas JM, Spellman JM, Jordan SW, Pardini MC, Afzal SMJ, Culo F: Treatment of tumours with the combination of WR2721 and cis-diamminedichloroplatinum-(II) or cyclophosphamide. Br J Cancer 42: 547–585, 1980
Myers CE, McGuire WP, Liss RH, Ifrim I, Grotzinger K, Young RC: Adriamycin: the role of lipid peroxidation in cardiac toxicity and tumor response. Science 197: 165–167, 1977
Treskes M, Nijtmans LG, Fichtinger Schepman AMJ, van der Vijgh WJ: Effects of the modulating agent WR2721 and its main metabolites on the formation and stability of cisplatin-DNA adducts in vitro in comparison to the effects of thiosulphate and diethyldithiocarbamate. Biochem Pharmacol 43: 1013–1019, 1992
DeNeve WJ, Everett CK, Suminski JE, Valeriote FA: Influence of WR2721 onDNAcross-linking by nitrogen mustard in normal mouse bone marrow and leukemia cells in vivo. Cancer Res 48: 6002–6005, 1988
Geldof AA: Nerve-growth-factor-dependent neurite outgrowth assay; a research model for chemotherapy-induced neuropathy. J Cancer Res Clin Oncol 121: 657–660, 1995
Geldof AA, Minneboo A, Heimans JJ: Vinca-alkaloid neurotoxicity measured using an in vitro model. J Neurooncol 37: 109–113, 1998
Brown DW, Graham WJ, McKenzie LJ, Pittock JW, Shaw LM: Can WR2721 be improved upon? Pharmacol Ther 39: 157–168, 1988
Greene LA, Tischler AS: Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which responds to nerve growth factor. Proc Natl Acad Sci USA 73: 2424–2428, 1976
Shpall EJ, Stemmer SM, Hami L, Franklin WA, Shaw L, Bonner HS, Bearman SI, Peters WP et al.: Amifostine (WR2721) shortens the engraftment period of 4–hydroperoxycyclophosphamide-purged bone marrow in breast cancer patients receiving high-dose chemotherapy with autologous bone marrow support. Blood 83: 3132–3137, 1994
Douay L, Hu C, Giarratana MC, Bouchet S, Conlon J, Capizzi RI, Gorin NC: Amifostine improves the antileukemic therapeutic index of mafosfamide: implications for bone marrow purging. Blood 86: 2849–2855, 1995
Meier T, Issels RD: Degradation of 2–(3–aminopropylamino)-ethanethiol (WR1065) by Cu-dependent amine oxidases and influence on glutathione status of Chinese hamster ovary cells. Biochem Pharmacol 50: 489–496, 1995
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Verstappen, C., Geldof, A., Postma, T. et al. In Vitro Protection from Cisplatin-induced Neurotoxicity by Amifostine and its Metabolite WR1065. J Neurooncol 44, 1–5 (1999). https://doi.org/10.1023/A:1006241622639
Issue Date:
DOI: https://doi.org/10.1023/A:1006241622639