Abstract
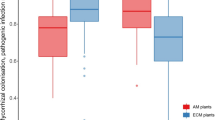
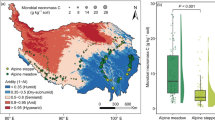
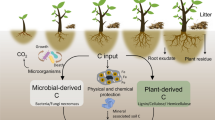
Measurements of root and nutrient distributions in agroforestry systems have often been obtained from unreplicated plots. We, therefore, measured the vertical and horizontal distribution of root length and soil nitrate in replicated plots of sesbania [Sesbania sesban (L.) Merr.] to assess plot-to-plot variation and identify appropriate sampling schemes. Sampling was on profile walls exposed from pits at two on-farm sites (Ochinga and Muange) in Kenya. At Ochinga, soil was sampled at 27 0.15-m depth intervals and eight 0.15-m-wide locations perpendicular to 2.25-m-wide rows of 19-month-old sesbania. At Muange, sampling was 18 months after establishment of sesbania from seedlings with a 1 m by 1 m spacing. Soil was sampled at 17 0.15-m depth intervals and nine 0.15-m-wide sampling locations – one exactly below the tree and four each on opposite sides of the tree. Root length density generally decreased with distance from sesbania and with soil depth. Soil nitrate, however, followed different trends in the four replications at Ochinga and the three replications at Muange. At Ochinga, nitrate above 2.5-m depth clearly increased with distance from the tree in two replications but followed no distinct trend in another replication. At Muange, nitrate above 1.2-m depth decreased with distance from the tree in one replication, increased with distance from the tree in another replication and followed no clear trend in another replication. The suitability of various sampling schemes for measuring root length and nitrate was accessed from percent bias between measured values at sampling locations in the scheme and actual values for an entire transect of sampling locations. Bias associated with selection of sampling locations tended to be higher within the 2.25-m-wide sesbania rows than the 1 m by 1 m sesbania spacing. Soil sampling in systems with rows of young trees should be at several locations along a transect perpendicular to tree rows in order to minimize bias.
Similar content being viewed by others
References
Buresh RJ and Tian G (1997) Soil improvement by trees in sub-Saharan Africa. Agrofor Syst 38: 51–76
Dorich RA and Nelson DW (1984) Evaluation of manual cadmium reduction methods for determination of nitrate in potassium chloride extract of soil. Soil Sci Soc Am J 48: 72–75
Eastham J and Rose CW (1990) Tree/pasture interactions at a range of tree densities in an agroforestry experiment: I. Rooting patterns. Aust J Agric Res 41: 683–695
Gomez KA and Gomez AA (1984) Statistical Procedures for Agricultural Research. Second edition. John Wiley and Sons, New York, 680 p
Hartemink AE, Buresh RJ, Jama B and Janssen BH (1996) Soil nitrate and water dynamics in sesbania fallow, weed fallows, and maize. Soil Sci Soc Am J 60: 568–574
Hilsheimer R and Harwig J (1976) Colorimetric determination of nitrate from meat and other foods: An alternative colour reagent for the carcinogenic 1-naphtylamine and an improved extraction method. Can Inst Food Sci Technol J 9: 225–227
Jama B, Buresh RJ, Ndufa JK and Shepherd KD (1998) Vertical distribution of roots and soil nitrate: Tree species and phosphorus effects. Soil Sci Soc Am J 62: 280–286
Jonsson K, Fidjeland L, Maghembe JA and Högberg P (1988) The vertical distribution of fine roots of five tree species and maize in Morogoro, Tanzania. Agrofor Syst 6: 63–69
Kirchhof G (1992) Measurement of root length and thickness using a hand-held computer scanner. Field Crops Res 29: 79–88
Kumar K, Prihar SS and Gajri PR (1993) Determination of root distribution of wheat by auger sampling. Plant and Soil 149: 245–253
Kwesiga F and Coe R (1994) The effect of short rotation Sesbania sesbanplanted fallows on maize yield. For Ecol Manage 64: 199–208
Mekonnen K, Buresh RJ and Jama B (1997) Root and inorganic nitrogen distributions in sesbania fallow, natural fallow and maize fields. Plant and Soil 188: 319–327
Newman EI (1966) A method for estimating the total length of root in a sample. J Appl Ecol 3: 139–145
Onim, JFM, Otieno K and Dzowela B (1990) The role of sesbania as multipurpose trees in small-scale farms in western Kenya. In: Macklin B and Evans DO (eds) Perennial SesbaniaSpecies in Agroforestry Systems. Proceedings of Workshop held in Nairobi, 27–31 March 1989, pp 167–179. Spec. Publ. 90-01. Nitrogen Fixing Tree Association, Waimanalo, Hawaii, USA
Ruark, GA and Zarnoch SJ (1992) Soil carbon, nitrogen, and fine root biomass sampling in a pine stand. Soil Sci Soc Am J 56: 1945–1950
Ruhigwa BA, Gichuru MP, Mambani B and Tariah NM (1992) Root distribution of Acioa barteri, Alchornea cordifolia, Cassia siameaand Gmelina arboreain an acid Ultisol. Agrofor Syst 27: 67–78
Schroth G (1995) Tree root characteristics as criteria for species selection and systems design in agroforestry. Agrofor Syst 30: 125–143
Toky OP and Bisht RP (1992) Observations on the rooting patterns of some agroforestry trees in an arid region of north-western India. Agrofor Syst 18: 245–263
Van Noordwijk M, Floris J and de Jager A (1985) Sampling schemes for estimating root density distribution in crop fields. Netherlands Journal of Agricultural Science 33: 241–262
Van Noordwijk M, Lawson G, Soumare A, Groot JJR and Hairiah K (1996) Root distribution of trees and crops: Competition and/or complementarity. In: Ong CK and Huxley P (eds) Tree-Crop Interactions, pp 319–364. CAB International, Wallingford, Oxon, UK
White RE, Haigh RA and Mcduff JH (1987)Frequency distributions and spatially dependent variability of ammonium and nitrate concentrations in soil under grazed and ungrazed grasslands. Fert Res 11: 193–208
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mekonnen, K., Buresh, R.J., Coe, R. et al. Root length and nitrate under Sesbania sesban: Vertical and horizontal distribution and variability. Agroforestry Systems 42, 265–282 (1998). https://doi.org/10.1023/A:1006180401982
Issue Date:
DOI: https://doi.org/10.1023/A:1006180401982