Abstract
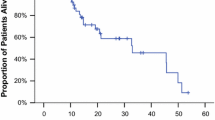
Gemcitabine has shown single-agent activity in metastatic breast cancer. Epirubicin is also widely used for the adjuvant and treatment of metastatic breast cancer. The toxicity profiles and modes of action are different which provides a good rationale for studying both drugs in combination. In a phase I study gemcitabine at a fixed dose of 1000 mg/m2 on days 1, 8, 15 of a 28 day cycle was combined with escalated weekly doses of epirubicin starting with an initial dose of 10 mg/m2. Patients had stage IV metastatic disease without previous chemotherapy except as adjuvant treatment. Nineteen patients were included in the study which defined the maximum tolerated dose (MTD) of epirubicin at 20 mg/m2. Myelosuppression was the dose limiting toxicity with leucopenia WHO grade 3 and 4 in 40.0% and 20.0%, neutropenia WHO grade 3 and 4 without neutropenic fever in 20.0% and 40.0% and thrombocytopenia WHO grade 4 in 20.0%. At the epirubicin 15 mg/m2 dose level, leucopenia (11.1% WHO grade 3) and neutropenia (12.5 and 37.5% WHO grade 3 and 4) were reported. Symptomatic toxicity was generally mild: nausea/vomiting in about 20% of patients (WHO grade 3 or 4) on both 15 and 20 mg/m2 epirubicin dose levels. Alopecia WHO grade 3 and 4 was seen in 2 patients at MTD. Four of 19 evaluable patients had a partial response. We conclude that the combination of gemcitabine and epirubicin is well tolerated and has promising activity. A phase II study is underway with gemcitabine 1000 mg/m2 and epirubicin 15 mg/m2 on days 1, 8 and 15 of a 28 day cycle.
Similar content being viewed by others
References
Jensen OM, Esteve J, Moller H, Renard H: Cancer in the European Community and its member states. Eur J Cancer 26: 1167–1256, 1990
Carmichael J, Possinger K, Phillip P, Beykirch M, Kerr H, Walling J, Harris A: Advanced breast cancer: a phase II trial with gemcitabine. J Clin Oncol 13: 2731–2736, 1995
Blackstein M, Vogel CL, Ambinder R, et al. Phase II study of gemcitabine in patients with metastatic breast cancer. Eur J Cancer 33 (suppl 8): S149, abstr 664, 1997
Spielmann M, Kalla S, Llombart-Cussac A, et al.: Activity of gemcitabine in metastatic breast cancer (MBC) patients previously treated with anthracycline-containing regimens. Eur J Cancer 33 (suppl 8): S149, abstr 663, 1997
García-Conde J, Lluch A, Pérez-Manga G, Palomero O, Alba E, Rueda A, Moreno Noguiera JA, Calvo E, Tarazona Y, López-Martin E: Gemcitabine + doxorubicin in advanced breast cancer: final results from an early phase II study. Proc ASCO 16: 147a, 1997
Coukell AJ, Faulds D: Epirubicin: An updated review of its pharmacodynamic and pharmacokinetic properties and therapeutic efficacy in the managment of breast cancer. Drugs 53: 453–482, 1997
Blomquist C, Elmoaa I, Rissanen P et al.: FEC (5–fluorouracil, epirubicin, cyclophosphamide) monthly versus FEC weekly in metastatic breast cancer. First results of a randomized trial. Acta Oncol 31(2): 231–236, 1992
Ricotti L, Zoli W, Barzanti F, Dal Susino M, Frassineti GL, Casadei Diunchi D, Milandri C, Amadori D: Schedule dependent interaction among doxorubicin, paclitaxel and gemcitabine in human breast cancer cell line. Proc ASCO 17: 959, 1998
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lüftner, D., Flath, B., Akrivakis, C. et al. Gemcitabine plus dose-escalated epirubicin in advanced breast cancer: results of a phase I study. Invest New Drugs 16, 141–146 (1998). https://doi.org/10.1023/A:1006121608414
Issue Date:
DOI: https://doi.org/10.1023/A:1006121608414