Abstract
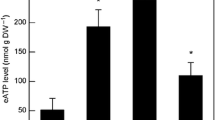
All the protoplasts analysed in this study whatever the original plant material were able to accumulate ABA under osmotic stress. The time course of ABA accumulation strongly differed according to the plant material. In both rose petal or Amaranthus leaf protoplasts, the increase in ABA level was significant but transient. Protoplasts prepared from Amaranthus cell suspensions behaved differently, showing a late and durable accumulation of ABA. Similar patterns of changes in ABA accumulation were observed in the original plant material under osmotic stress. A pretreatment of plant material by fluridone induced a strong inhibition of ABA accumulation whatever the origin of protoplasts was. This result suggests that ABA could be synthesised via the carotenoid pathway in the absence of the cell wall.
Similar content being viewed by others
References
Alibert G, Carrasco A and Boudet AM (1982) Changes in biochemical composition of vacuoles isolated from Acer pseudoplatanus L. during cell culture. Biochimica Biophysica Acta 721: 22-29
Barthe P, Vaillant V and Gudin S (1991) pH of cell sap and vacuolar pH during senescence of the rose petal. Acta Hort 298: 135-139
Belefant H and Fong F (1991) Abscisic acid biosynthesis in Zea mays embryos. Plant Science 78: 19-25
Bianco-Colomas J and Hugues M (1990) Establishment and characterization of a betacyanin producing cell line of Amaranthus tricolor: inductive effects of light and cytokinin. J Plant Physiol 136: 734-739
Bianco-Colomas J, Barthe P, Orlandini M and Le Page-Degivry MTh (1991) Carrier-mediated uptake of abscisic acid by suspension-cultured Amaranthus tricolor cells. Plant Physiol 95: 990-996
Bianco-Trinchant J, Guigonis JM and Le Page-Degivry MTh (1993) Early release of ABA from cell walls during rose petal protoplast isolation. J Exp Bot 44: 957-962
Bokemeyer D, Sorokin A, Yang M, Ahn NG, Templeton DJ and Dunn MJ (1996) Induction of mitogen-activated protein kinase phosphatase 1 by the stress-activated protein kinase signaling pathway but not by extracellular signal-regulated kinase in fibroblasts. J Biol Chem 271: 639-642
Bostock RM and Quatrano RS (1992) Regulation of Em gene expression in Rice: interaction between osmotic stress and abscisic acid. Plant Physiol 98: 1356-1363
Creelman RA and Zeevaart JAD (1985) Abscisic acid accumulation in spinach leaf slices in the presence of penetrating and nonpenetrating solutes. Plant Physiol 77: 25-28
Galbraith DW (1981) Microfluorimetric quantitation of cellulose biosynthesis by plant protoplast using calcofluor white. Physiol Plant 53: 111-116
Jonak C, Kiegerl S, Ligterink W, Barker PJ, Huskisson NS and Hirt H (1996) Stress signaling in plants: a mitogen-activated protein kinase pathway is activated by cold and drought. Proc Natl Acad Sci 93: 11274-11279
Lahr W and Raschke K (1988) Abscisic-acid contents and concentrations in protoplasts from guard cells and mesophyll cells of Vicia faba L. Planta 173: 528-531
Le Page-Degivry MTh, Duval D, Bulard C and Delaage M (1984) A radioimmunoassay for abscisic acid. J Immunol Methods 67: 119-128
Le Page-Degivry MTh, Orlandini M, Garello G, Barthe Ph and Gudin S (1991) Regulation of ABA levels in senescing petals of rose flowers. J Plant Growth Regul 10: 67-72
Loveys BR and Robinson SP (1987) Abscisic acid synthesis and metabolism in barley leaves and protoplasts. Plant Science 49: 23-30
Loveys BR, Brien CJ and Kriedemann PE (1975) Biosynthesis of abscisic acid under osmotic stress: studies bases on a dual labelling technique. Physiol Plant 33: 166-170
Pierce M and Raschke K (1980) Correlation between loss of turgor and accumulation of abscisic acid in detached leaves. Planta 148: 174-182
Radin JN and Hendrix DL (1986) Accumulation and turnover of abscisic acid in osmotically stressed cotton leaf tissues in relation to temperature. Plant Science 45: 37-42
Rös M and Wernicke W (1991) The first cell division cycle in Nicotiana mesophyll protoplast culture in vitro. I. Method to determine cell kinetics. J Plant Physiol 138: 150-155
Weiler EW, Schnabl H and Hornberg C (1982) Stress-related levels of abscisic acid in guard cell protoplasts of Vicia faba L. Planta 154: 24-28
Wright STC (1977) The relationship between leaf water potential and the levels of abscisic acid and ethylene in excised wheat leaves. Planta 134: 183-189
Zabadal TJ (1974) A water potential threshold for the increase of abscisic acid in leaves. Plant Physiol 53: 125-127
Zeevaart JAD (1980) Changes in the level of abscisic acid and its metabolites in excised leaf blades of Xanthium strumarium during and after water stress. Plant Physiol 66: 672-678
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bianco-Trinchant, J., Le Page-Degivry, M.T. ABA synthesis in protoplasts of different origin in response to osmotic stress. Plant Growth Regulation 25, 135–141 (1998). https://doi.org/10.1023/A:1006085327980
Issue Date:
DOI: https://doi.org/10.1023/A:1006085327980