Abstract
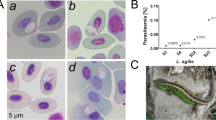
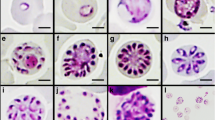
Fallisia biporcati n. sp. parasitises thrombocytes and lymphocytes of Anolis biporcatus and A. lionotus in Panama. Round or oval schizonts average 13.3 × 11.5 (10.5–16 × 9–13) μm, with LW 153.8 (94–208) μm2, and produce 38.3 (28–60) merozoites. Gametocytes are variably shaped, from round or oval to nearly triangular or rectangular, and average 12.6 × 9.0 (10–15 × 6–12) μm, with LW 113.1 (82–150) μm2 and L/W ratio 1.43 (1.0–2.2). Thrombocytes and lymphocytes of A. poecilopus in Panama are parasitised by F. poecilopi n. sp. Schizonts, oval to elongate in shape, 7.7 × 4.7 (5.5–9 × 3–6) μm, with LW 36.5 (22–54) μm2, are filled with 31.0 (20–51) tiny nuclei or merozoites. Gametocytes are 10.1 × 8.0 (7.5–14 × 6–11) μm, with LW 82.0 (45–132) μm2, round to elongate with L/W ratio 1.27 (1.0–1.6). F. thecadactyli n. sp. parasitises thrombocytes and lymphocytes of Thecadactylus rapicaudus in Panama and Venezuela. Oval, oblong, or triangular schizonts average 10.3 × 8.0 (7–13 × 5–12) μm, with LW 86.6 (37–156) μm2, and produce 40.2 (26–61) merozoites. Gametocytes are round, oval, triangular or elongate, 10.4 × 7.0 (7–15 × 5–11) μm, with LW 74.8 (40–154) μm2 and L/W ratio 1.51 (1.1–2.2). F. dominicensis n. sp. parasitises thrombocytes of A. cybotes on Hispaniola. Schizonts, 6.0 × 4.8 (4–8 × 3–7) μm, with LW 29.1 (12–56) μm2, round, oval, elongate, oblong or lentiform in shape, produce 12.4 (8–22) merozoites. Gametocytes are 6.6 × 5.0 (5–9 × 4–7) μm , with LW 33.8 (20–56) μm2, round, oval or elongate, and L/W ratio of 1.34 (1.1–2.0).
Similar content being viewed by others
References
Boulard, Y., Landau, I., Baccam, D., Petit, G. & Lainson, R. (1987) Observations ultrastructurales sur les formes sanguines des Garnides (Garnia gonatodi, G. uranoscodoni et Fallisia effusa) parasites de lezards Sud-Americains. European Journal of Protistology, 23, 66–75.
Gabaldon, A., Ulloa, G. & Zerpa, N. (1985) Fallisia (Plasmodioides) neotropicalis subgen. nov., sp. nov. from Venezuela. Parasitology, 90, 217–225.
Kimsey, R.B. (1992) Host association and the capacity of sand flies as vectors of lizard malaria in Panama. International Journal for Parasitology, 22, 657–664.
Lainson, R., Landau, I. & Shaw, J.J. (1974) Further parasites of the family Garniidae (Coccidiida: Haemosporidiidea) in Brazilian lizards. Fallisia effusa gen. nov., sp. nov. and Fallisia modesta gen. nov., sp. nov. Parasitology, 68, 117–125.
Lainson, R., Shaw, J.J. & Landau, I. (1975) Some blood parasites of the Brazilian lizards Plica umbra and Uranoscodon superciliosa (Iguanidae). Parasitology, 70, 119–141.
Levine, N.D. (1985) Phylum II. Apicomplexa Levine, 1970. In: Lee, J.J., Hutner, S.H. & Bovee, E.C. (Eds) An illustrated guide to the Protozoa. Lawrence, Kansas: Society of Protozoologists, pp. 322–374.
Levine, N.D. (1988) The protozoan phylum Apicomplexa. Vols 1 & 2. Boca Raton, Florida: CRC Press Inc., 357 pp.
Paperna, I. & Landau, I. (1990) Fallisia copemani (Haemosporidia: Garniidae) from the Australian skink Carlia rhomboidalis. Annales de Parasitologie Humaine et Comparée, 65, 16–21.
Peirce, M.A. & Bennett, G.F. (1996) A revised key to the avian subgenera of Plasmodium Marchiafava & Celli, 1885 (Apicomplexa). Systematic Parasitology, 33, 31–32.
Schall, J.J. & Vogt, S.P. (1993) Distribution of malaria in Anolis lizards of the Luquillo forest, Puerto Rico: implications for host community ecology. Biotropica, 25, 229–235.
Scorza, J.V. (1971) Asexual and sexual stages of a malaria parasite in the thrombocytes of Tropidurus torquatus (Iguanidae) infected with Plasmodium tropiduri. Journal of Protozoology, 18, 403–410.
Staats, C.M. & Schall, J.J. (1996) Distribution and abundance of two malarial parasites of the endemic Anolis lizard of Saba Island, Netherlands Antilles. Journal of Parasitology, 82, 409–413.
Telford, S.R., Jr (1970) Exoerythrocytic gametocytes of saurian malaria. Quarterly Journal of the Florida Academy of Sciences, 33, 77–79.
Telford, S.R., Jr (1973) Saurian malarial parasites from Guyana: their effect upon the validity of the family Garniidae and the genus Garnia, with descriptions of two new species. International Journal for Parasitology, 3, 829–842.
Telford, S.R., Jr (1974) The malarial parasites of Anolis species (Sauria: Iguanidae) in Panama. International Journal for Parasitology, 4, 91–102.
Telford, S.R., Jr (1975) Saurian malaria in the Caribbean: Plasmodium azurophilum sp. nov., a malaria parasite with schizogony and gametogony in both red and white cells. International Journal for Parasitology, 5, 383–394.
Telford, S.R., Jr (1979) A taxonomic reconsideration of some Plasmodium species from iguanid lizards. Annales de Parasitologie Humaine et Comparée, 54, 129–144.
Telford, S.R., Jr (1986) Fallisia parasites (Haemosporidia: Plasmodiidae) from the flying lizard, Draco maculatus (Agamidae) in Thailand. Journal of Parasitology, 72, 766–769.
Telford, S.R. Jr (1988) A contribution to the systematics of the reptilian malaria parasites, Family Plasmodiidae (Apicomplexa: Haemospororina). Bulletin of the Florida State Museum, Biological Sciences, 34, 65–96.
Telford, S.R. Jr, Johnson, R.N. & Young, D.G. (1989) Additional Plasmodium species from Anolis lizards of Hispaniola and Panama. International Journal for Parasitology, 19, 275–284.
Telford, S.R. Jr (1994) Plasmodia of Reptiles. In: Kreier, J.P. (Ed.) Parasitic Protozoa. 2nd Edit. San Diego, California: Academic Press, Vol.7, 1–71.
Thompson, P.E. & Hart, T.A. (1946) Plasmodium lacertiliae n. sp. and other saurian blood parasites from the New Guinea area. Journal of Parasitology, 32, 79–82.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Telford, S.R. Additional Fallisia spp. (Apicomplexa: Plasmodiidae) of Neotropical lizards. Syst Parasitol 40, 185–193 (1998). https://doi.org/10.1023/A:1006008301011
Issue Date:
DOI: https://doi.org/10.1023/A:1006008301011