Abstract
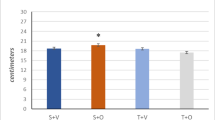
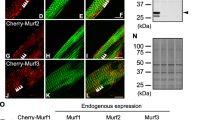
In smooth muscle, the phosphorylation/dephosphorylation of the 20-kDa regulatory light chain of myosin (MLC20) is known to regulate actomyosin interaction and force. However, a thin filament based regulatory system for actomyosin interaction has been suggested to exist in parallel to MLC20 phosphorylation. Calponin is a thin filament associated protein that in vitro inhibits actomyosin interaction, and has been suggested to reduce maximal shortening velocity (v max). Using antibodies to h1- and h2-calponin, we demonstrated that calponin was present in smooth muscle from Sprague Dawley (SD) rats, while calponin was not detectable in the smooth muscle from Wistar Kyoto (WKY) rats. v max determined from the force vs. velocity relationship at maximal Ca2+ activation was not different for either the aorta or the portal vein of SD vs. WKY rats. These results suggest that physiological levels of calponin do not contribute to a thin filament-based secondary regulation to inhibit smooth muscle contraction.
Similar content being viewed by others
References
Abe M, Takahashi K and Hiwada K (1992) Effect of calponin on actin-activated myosin ATPase activity. J Biochem (Tokyo) 108: 835–838.
Bárány M and Bárány K (1993) Calponin expression in porcine smooth muscle. Biochim Biophys Acta 1179: 229–233.
Bárány K, Polyak E and Bárány M (1992a) Involvement of calponin and caldesmon in sustained contraction of arterial smooth muscle. Biochem Biophys Res Commun 187: 847–852.
Bárány M, Rokolya A and Bárány K (1992b) Absence of calponin phosphorylation in contracting or resting arterial smooth muscle. FEBS Left 279: 65–68.
Barsotti RJ, Ikebe M and Hartshorne DJ (1987) Effects of Ca2+, Mg2+, and myosin phosphorylation on skinned smooth muscle fibers. Am J Physiol 252: C543-C554.
Brozovich FV and Yamakawa M (1995) Thin filament regulation of force activation is not essential in single vascular smooth muscle cells. Am J Physiol: Cell Physiol 268: C237-C242.
Dillon PF, Aksoy MO, Driska SP and Murphy RA (1981) Myosin phosphorylation and crossbridge cycle in arterial smooth muscle. Science 211: 495–497.
Fisher SA, Ikebe M and Brozovich F (1997) Endothelin-I alters the contractile phenotype of cultured embryonic smooth muscle cells. Circ Res 80: 885–893.
Goldman YE, Hibberd MG and Trentham DR (1984) Relaxation of rabbit psoas muscle fibres from rigor by photochemical generation of adenosine-5′-triphosphate. J Physiol 354: 577–604.
Haeberle JR (1994) Calponin decreases the rate of cross-bridge cycling and increases maximum force production by smooth muscle myosin in an in vitro motility assay. J Biol Chem 269: 12424 12431.
Hartshorne DJ (1987) Biochemistry of the Contractile Process in Smooth Muscle. Physiology of the Gastrointestinal Tract. (pp 432–482) New York, Raven Press.
Hathaway DR and Haeberle J (1985) A redioimmunoblotting method for measuring myosin light chain phosphorylation levels in smooth muscle. Am J Physiol: Cell Physiol 249: C345-C351.
Itoh T, Ikebe M, Kargacin GJ, Hartshorne DJ, Kemp BE and Fay FS (1989) Effects of modulators of myosin light-chain kinase activity in single smooth muscle cells. Nature 338: 164–167.
Jaworowski A, Anderson KI, Arner A, Engstroem M, Gimona M, Strasser P and Small JV (1995) Calponin reduces shortening velocity in skinned taenia coli smooth muscle fibres. FEBS Left 365: 167–171.
Jin J-P, Walsh MP, Reset ME and McMartin GA (1996) Expression and epitopic conservation of calponin in different smooth muscles and during development. Biochem Cell Biol 74: 187–196.
Kargacin GJ, Ikebe M and Fay FS (1992) Peptide modulators of myosin light chain kinase affect smooth muscle cell contraction. Am J Physiol 259: C315-C324.
Kelley CA, Takahashi M, Yu JH and Adelstein RS (1993) An Insert of 7 amino acids confers functional differences between smooth muscle myosins from the intestines and vasculature. J Biol Chem 268: 12848–12854
Lehman W (1991) Calponin and composition of the smooth muscle thin filament. J Muscle Res Cell Moti 12: 221–224.
Malmqvist U and Arner A (1991) Correlation between isoform composition of the 17 kDa myosin light chain and maximal shortening velocity in smooth muscle. Pflugers Arch 418: 523–530.
Malmqvist U, Trybus KM, Yagi S, Carmichael J and Fay FS (1997) Slow cycling of un phosphorylated myosin is inhibited by calponin, thus keeping smooth muscle relaxed. Proc Natl Acad Sci USA 94: 7655–7660.
Marston SB and Smith CW (1985) The thin filaments of smooth muscles. J Muscle Res Cell Moti 6: 669–708.
Matthew JD, Khromov AS, Trybus KM, Somlyo A V and Somlyo AP (1998) Myosin essential light chain isoforms modulate the velocity of shortening propelled by nonphosphorylated cross-bridges. J Bioi Chem 273: 31289–31296.
Miki M, Walsh MP and Hartshorne DJ (1992) The mechanism of inhibition of the actin-activated myosin MgATPase by calponin. Biochem Biophys Res Commun 187: 867–871.
Nabeshima Y, Nonomura Y and Fujii-Kuriyama Y (1987) Nonmuscle and smooth muscle myosin light chain mRNAs are generated from a single gene by tissue-specific alternative RNA splicing. J Bioi Chem 262: 10608–10612.
Nigam R, Triggle CR and Jin JP (1998) hl-and h2-Calponins are not essential for norepinephrine-or sodium fluoride-induced contrac tion of rat aortic smooth muscle. J Muscle Res Cell Moti 19: 695–703.
Obara K, Szymanski PT, Tao T and Paul RJ (1996) Effects of calponin on isometric force and shortening velocity in permeabilized taenia coli smooth muscle. Am J Physiol: Cell Physiol 270: C481-C487.
Ogut O and Brozovich FV (1999) Determinants of the rate of force activation in the developing chicken and aorta. Biophys J 76: A284.
Pohl J, Winder SJ, Allen BG, Walsh MP, Sellers JR and Gerthoffer WT (1997) Phosphorylation of calponin in airway smooth muscle. Am J Physiol: Lung Cell Mol Physiol 272: L115-L123.
Rokolya A, Walsh MP and Moreland RS (1996) Calcium-and phorbol ester-dependent calponin phosphorylation in homogen ates of swine carotid artery. Am J Physiol: Heart and Circ Physiol 271: H776-H783.
Rovner AS, Freyzon Y and Trybus KM (1997) An insert in the motor domain determines the functional properties of expressed smooth muscle myosin isoforms. J Muscle Res Cell Moti 18: 103–110.
Shirinsky VP, Biryukov KG, Hettasch JM and Sellers JR (1992) Inhibition of the Relative Movement of Actin and Myosin by Caldesmon and Calponin. J Bioi Chem 267: 15886–15892.
Smith JP and Barsotti RJ (1993) A computer-based servo system for controlling isotonic contractions of muscle. Am J Physiol: Cell Physiol 265: C1424-C1432.
Strasser P, Gimona M, Herbert S, Herzog M and Small JV (1993) Mammalian calponin: Identification and expression of genetic variants. FEBS Left 330: 13–18.
Szymanski PT, Chacko TK, Rovner AS and Goyal RK (1998) Differences in contractile protein content and isoforms in phasic and tonic smooth muscles. Am J Physiol: Cell Physiol 275: C684-C692.
Takahashi K, Hiwada K and Kokubu T (1986) Isolation and characterization of a 34,000-dalton calmodulin and F-actin-bind ing protein from chicken gizzard smooth muscle. Biochem Biophys Res Commun 141: 20–26.
Takahashi K, Hiwada K and Kokubu T (1988) Vascular smooth, muscle calponin: a novel troponin T-like protein. Hypertension 11: 620–626.
Takahashi K, Mitsuo-Saito M, Fuchibe K, Yamamura H, Tanaguchi S, Katsuki M, Ozaki H, Tsuchiya T, Shibata Nand Karaki H (1998) Targeted disruption of calponin results in rapid cross-bridge cycling and reduced force in phasic smooth muscle. Biophys J 74: A38.
Takahashi K and Nadal-Ginard B (1991) Molecular cloning and sequence analysis of smooth muscle calponin. J. Bioi Chem 266: 13284–13288.
Uyama Y, Imaizumi Y, Watanabe M and Walsh MP (1996) Inhibition by calponin of isometric force in demembranated vascular smooth muscle strips: The critical role of serine-I 75. Biochem J 319: 551–558.
Walsh MP (1991) Calcium-dependent mechanisms of smooth muscle contraction. Biochem Cell Biol 69: 771–800.
Winder SJ, Allen BG, Clement-Chomienne O and Walsh MP (1998) Regulation of smooth muscle actin-myosin interaction and force by calponin. Acta Physiol Scandinavica 164: 415–426.
Winder SJ, Allen BG, Fraser ED, Kang HM, Kargacin GJ and Walsh MP (1993) Calponin phosphorylation in vitro and in intact muscle. Biochem J 296: 827–836.
Winder SJ, Sutherland C and Walsh MP (1992) A comparison of the effects of calponin on smooth and skeletal muscle actomyosin systems in the presence and absence of caldesmon. Biochem J 288: 733–739.
Winder SJ and Walsh MP (1990) Smooth muscle calponin: Inhibition of actomyosin MgATPase and regulation by phosphorylation. J Bioi Chem 265: 10148–10155.
Winder SJ and Walsh MP (1993) Calponin: thin filament-linked regulation of smooth muscle contraction. Cell Signals: 677–686.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Facemire, C., Brozovich, F.V. & Jin, JP. The maximal velocity of vascular smooth muscle shortening is independent of the expression of calponin. J Muscle Res Cell Motil 21, 367–373 (2000). https://doi.org/10.1023/A:1005680614296
Issue Date:
DOI: https://doi.org/10.1023/A:1005680614296