Abstract
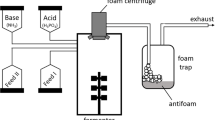
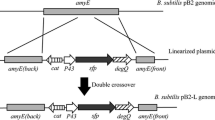
Bacillus subtilis C9 produces a lipopeptide-type biosurfactant, surfactin, and rapidly degrades alkanes up to a chain length of C19. The nucleotide sequence of the sfp gene cloned from B. subtilis C9 was determined and its deduced amino acid sequence showed 100% homology with the sfp gene reported before [Nakano et al. (1992) Mol. Gen. Genet. 232: 313–321]. To transform a non-surfactin producer, B. subtilis 168, to a surfactin producer, the sfp gene cloned from B. subtilis C9 was expressed in B. subtilis 168. The transformed B. subtilis SB103 derivative of the strain 168 was shown to produce surfactin measured by its decrease in surface tension, emulsification activity, and TLC analysis of the surface active compound isolated from the culture broth. Like B. subtilis C9, B. subtilis SB103 containing sfp gene readily degraded aliphatic hydrocarbons (C10−19), though its original strain did not. The addition of surfactin (0.5%, w/v) to the culture of B. subtilis 168 significantly stimulated the biodegradation of hydrocarbons of the chain lengths of 10–19; over 98% of the hydrocarbons tested were degraded within 24 h of incubation. These results indicate that the lipopeptide-type biosurfactant, surfactin produced from B. subtilis enhances the bioavailability of hydrophobic hydrocarbons.
Similar content being viewed by others
References
Anagnostopoulos C, Spizizen J (1961) Requirements for transformation in Bacillus subtilis. J. Bacteriol. 81: 741–746.
Andreas KK, Kappeli O, Fiechter A, Reiser J (1991) Hydrocarbon assimilation and biosurfactant production in Pseudomonas aeruginosa mutants. J. Bacteriol. 173: 4212–4219.
Banat IM (1995) Characterization of biosurfactants and their use in pollution removal-State of the art. Acta Biotechnol. 15: 251–267.
Cosmina P, Rodriguez F, de Ferra F, Grandi G, Perego M, Venema G, van Sinderen D (1993) Sequence and analysis of the genetic locus responsible for surfactin synthesis in Bacillus subtilis. Mol. Microbiol. 8: 821–831.
Desai JD (1987) Microbial surfactants: Evaluation, types, production and future applications. J. Sci. Ind. Res. 46: 440–449.
Doi RH (1983) Reading, In: Rodriguez RL, Tait RC, eds. Recombinant DNA Techniques. Reading, MA: Addison Wesley, pp. 161–163.
Georgiou G, Lin SC, Sharma MM(1992) Surface-active compounds from microorganisms. Bio/Technology 10: 60–65.
Inoue H, Nojima H, Okayama H (1990) High efficiency transformation of Escherichia coli with plasmids. Gene 96: 23–28.
Jain DK, Collins-Thompson DL, Lee H, Trevors JT (1991) A drop-collapsing test for screening surfactant-producing microorganisms. J. Microbiol. Meth. 13: 271–279.
Kim HS, Lee CH, Suh HH, Ahn KH, Oh HM, Kwon GS, Yang JW, Yoon BD (1997a) A lipopeptide biiosurfactant produced by Bacillus subtilis C9 selected through the oil film-collapsing assay. J. Microbiol. Biotechnol. 7: 180–188.
Kim HS, Yoon BD, Lee CH, Suh HH, Oh HM, Katsuragi T, Tani Y (1997b) Production and properties of a lipopeptide biosurfactant from Bacillus subtilis C9. J. Ferment. Bioengin. 84: 41–46.
Koch AK, Käppeli O, Fiechter A, Reiser J (1991) Hydrocarbon assimilation and biosurfactant production in Pseudomonas aeruginosa mutants. J. Bacteriol. 173: 4212–4219.
Miller JH (1972) Experiments in Molecular Genetics. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory.
Nakano MM, Zuber P (1989) Cloning and characterization of srfB, a regulatory gene involved in surfactin production and competence in Bacillus subtilis. J. Bacteriol. 171: 5347–5353.
Nakano MM, Corbell N, Besson J, Zuber P (1992) Isolation and characterization of sfp: a gene that functions in the production of the lipopeptide biosurfactant, surfactin, in Bacillus subtilis. Mol Gen. Genet. 232: 313–321.
Nakano MM, Marahiel MA, Z uber P (1988) Identification of a genetic locus required for biosynthesis of the lipopeptide antibiotic surfactin in Bacillus subtilis. J. Bacteriol. 170: 5662–5668.
Quadri LEN, Weinreb PH, Lei M, Nakano MM, Zuber P, Walsh CT (1998) Characterization of Sfp, a Bacillus subtilis phosphopantetheinyl transferase for peptidyl carrier protein domains in peptide synthetases. Biochemistry 37: 1585–1595.
Reuter K, Mofid MR, Marahiel MA, Ficner R (1999) Crystal structure of the surfactin synthetase-activating enzyme Sfp: a prototype of the 4′-phosphopantetheinyl transferase superfamily. EMBO J. 18: 6823–6831.
Rosenberg E, Zuckerberg A, Rubinovitz C, Gutnick DL (1979) Emulsifier of Arthrobacter sp. RAG-1: isolation and emulsifying properties. Appl. Environ. Microbiol. 37: 402–408.
Sanger R, Nicklen W, Coulson AR (1977) DNA sequencing with chain termination inhibitors. Proc. Natl. Acad. Sci. USA 74: 5463–5467.
Spizizen J (1958) Transformation of biochemically deficient strains of Bacillus subtilis by deoxyribonucleate. Proc. Natl. Acad. Sci. 44: 1072–1078.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, HS., Kim, SB., Park, SH. et al. Expression of sfp gene and hydrocarbon degradation by Bacillus subtilis. Biotechnology Letters 22, 1431–1436 (2000). https://doi.org/10.1023/A:1005675231949
Issue Date:
DOI: https://doi.org/10.1023/A:1005675231949