Abstract
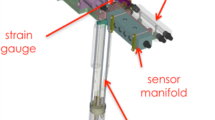
A simple device for taking in situ proton NMR measurements in 1H2O is described. This allows aeration of reactions in a 10 mm diameter NMR tube without modifying the magnet or the probe head. With this device, aerobic biotransformations can be monitored in the NMR-tube placed in the spectrometer. It allows in situ analyses of the transformations, separating the aeration period temporally from the measurement time, not unlike traditional Warburg respiratory experiments. Two reactions determining kinetic and stoichieometric parameters: (i) a biotransformation by a growing Pseudomonas putida culture and (ii) l-phenylalanine oxidation catalysed by l-amino acid oxidase [E.C. 1.4.3.2]; both incubations were contained in the magnet.
Similar content being viewed by others
References
Albert K (1988) Durchfluß-NMR-Spektroskopie. Habilitationsschrift. Tübingen: Universität.
Besse P, Combourieu B, Poupin P, Sancelme M, Truffaut N, Veschambre H, Delort AM (1998) Degradation of morpholine and thiomorpholine by an environmental Mycobacterium involves a cytochrome P450. Direct evidence of intermediates by in situ 1H NMR. J. Mol. Catal. B5: 403–409.
Brecker L, Ribbons DW (2000) Biotransformations monitored in situ by proton nuclear magnetic resonance spectroscopy. Trends Biotechnol. 18: 197–202.
Brecker L, Pogorevc M, Griengl H, Steiner W, Kappe T, Ribbons DW (1999a) Synthesis of 2,4-diketo acids and their aqueous solution structures. New J. Chem. 23: 437–446.
Brecker L, Weber H-J, Griengl H, Ribbons DW (1999b) In situ proton-NMR analyses of Escherichia coli HB101 fermentations in 1H2O and in D2O. Microbiology 145: 3389–3397.
Chen R, Bailey JE (1993) Observations of aerobic, growing Escherichia coli metabolism using an on-line nuclear magnetic resonance spectroscopy system. Biotechnol. Bioeng. 42: 215–221.
Fernandez EJ, Mancuso A, Murphy MK, Blanch HW, Clark DS (1990) Nuclear magnetic resonance methods for observing the intracellular environment of mammalian cells. Ann. N.Y. Acad. Sci. 589: 458–475.
deGraaf AA, Wittig RM, Probst U, Strohhäcker J, Schoberth SM, Sahm H (1992) Continuous-flow NMR bioreactor for in vivo studies of microbial cell suspensions with low biomass concentrations. J. Magn. Reson. 98: 654–659.
Guéron M, Plateau P, Decorps M (1991) Solvent signal suppression in NMR. Prog. NMR Spectrosc. 23: 135–209.
Hartbrich A, Weuster-Botz D, Wandrey C (1995) Cultivation of aerobic microorganisms at high cell density. German Patent DE P 44.07 440.9–41.
Herbert D, Phipps PJ, Strange RE (1971) Chemical analysis of microbial cells. In: Norris JR, Ribbons DW, eds. Methods Microbiology, Vol. 5b. London: Academic Press Ltd., pp. 210–383.
Hore JP (1989) Solvent suppression. In: Oppenheimer J, James JT, eds. Methods Enzymology, Vol. 176. San Diego: Academic Press Inc., pp. 64–77.
Jenkins GN, Ribbons DW, Widdowson DA, Slawin AMZ, Willams DJ (1995) Synthetic application of biotransformations: absolute stereochemistry and Diels-Alder reactions of the (1S,2R)-1,2-dihydroxycyclohexa-3,5-diene-1-car boxylic acid from Pseudomonas putida. J. Chem. Soc. Perkin Trans. I 1995: 2647–2655.
Knox WE, Pitt BM (1957) Enzymic catalysis of the keto-enol tautomerisation of phenylpyruvic acids. J. Biol. Chem. 225: 675–688.
Kramer HW, Bailey JE (1991) Mass transfer characterisation of an airlift probe for oxygenation and mixing cell suspensions in an NMR spectrometer. Biotechnol. Bioeng. 37: 205–209.
Morawski B, Casy G, Illaszewicz C, Griengl H, Ribbons DW (1997a) Stereochemical course of two arene-cic-diol dehydrogenases specifically induced in Pseudomonas putida. J. Bacteriol. 179: 4023–4029.
Morawski B, Eaton RW, Rossiter JT, Guoping S, Griengl H, Ribbons DW(1997b) 2-Naphthoate catabolic pathway in Burkholderia strain JT 1500. J. Bacteriol. 179: 115–121.
Ng FMW, Dawes EA (1973) Chemostat studies on regulation of glucose metabolism in Pseudomonas aeruginosa by citrate. Biochem. J. 132: 129–140.
Pokorny D, Brecker L, Pogorevc M, Steiner W, Griengl H, Kappe T, Ribbons DW (1999) Proton-nuclear magnetic resonance analyses of the substrate specificity of a _-ketolase from Pseudomonas putida, acetopyruvate hydrolase. J. Bacteriol. 181: 5051–5059.
Santos H, Turner DL (1986) Characterisation of the improved sensitivity obtained using a flow method for oxygenating and mixing cell suspensions in NMR. J. Magn. Reson. 68: 345–349.
Sprinson DB, Rittenberg D (1951) Nature of the activation process in enzymatic reactions. Nature 167: 484.
Taylor SJC, Ribbons DW, Slawin AMZ, Widdowson DA, Williams DJ (1987) Biochemically generated chiral intermediates for organic synthesis: the absolute stereochemistry of 4-bromo-cis-2,3-dihydrocyclohexa-4,6-diene-1-c arboxylic acid formed from 4-bromobenzoic acid by a mutant of Pseudomonas putida. Tetrahedron Lett. 28: 6391–6392.
Umbreit WW, Burris RH, Stauffer JT (1957) Manometric Techniques. Minneapolis: Burgess Publishing Co.
Wandelt A (1994) Neue NMR-Methoden zur PHIP-Untersuchung homogen und heterogen katalysierter Hydrierungen. Dissertationsschrift. Bonn: Universität.
Wellner D, Meister A (1960) Crystalline L-amino acid oxidase of Crotalus adamanteus. J. Biol. Chem. 235: 2013–2018.
Weuster-Botz D, deGraaf AA (1996) Reaction engineering methods to study intracellular metabolite concentrations. In: Scheper T, ed. Adv. Biochem. Eng. Biotechnol., Vol. 54, pp. 75–108.
Woelk K, Bargon J (1992) High-pressure NMR probes for in situ investigation of gas/liquid reactions. Rev. Sci. Instrum. 63: 3307–3310.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brecker, L., Urdl, P., Schmid, W. et al. Simple device to monitor aerobic biotransformations by in situ 1H-NMR. Biotechnology Letters 22, 1135–1141 (2000). https://doi.org/10.1023/A:1005664400918
Issue Date:
DOI: https://doi.org/10.1023/A:1005664400918