Abstract
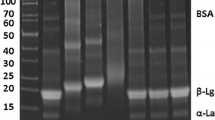
Non-dialyzable fraction of sweet whey was chromatographed on a column of phenyl-agarose equilibrated with 0.01 M sodium phosphate buffer, pH 6.8 containing 5 M NaCl. Most whey proteins were adsorbed on the column, while the glycomacropeptide (GMP) was not. Amino acid analysis of the GMP fraction showed presence of traces (each < 1 residue/peptide) of arginine, histidine and phenylalanine which are not found in GMP. The estimated yield of GMP fraction was approximately 1.6 g l−1 of sweet whey.
Similar content being viewed by others
References
Abd El-Salam MH, El-Shibiny S, Buchheim W (1996) Int. Dairy J. 6: 327-341.
AOAC (1998) Official Methods of Analysis of AOAC International, 16th ed. Gaithersburg, MD: Association of Official Analytical Chemists.
Coolbear KP, Elgar DF, Ayers JS (1996) Int. Dairy J. 6: 1055-1068.
Dische Z, Borenfreund E (1950) J. Biol. Chem. 184: 517-522.
Dziuba J, Minkiewicz P (1996) Int. Dairy J. 6: 1017-1044.
Eigel WN, Butler JE, Ernstrom CA, Farrel HM Jr, Harwalkar VR, Jenness R, Whitney RMcL (1984) J. Dairy Sci. 67: 1599-1631.
Kawakami H, Kawasaki Y, Dosako S, Tanimoto M Nakajima I (1992) Milchwissenschaft 47: 688-693.
Léonil J, Mollé D (1991) J. Dairy Res. 58: 321-328.
Mechanic GL (1979) Collagen biochemistry. In: Simmons DJ, Kunin AS, eds. Skeletal Research. New York: Academic Press, pp. 227-241.
Nakano T, Ozimek L (1998) Milchwissenschaft 53: 629-633.
Reid JR, Coolbear T, Ayers JS, Coolbear KP (1997) Int. Dairy J. 7: 559-569.
Trevelyan W, Harrison JS (1952) Biochem. J. 50: 298-303.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakano, T., Ozimek, L. Purification of glycomacropeptide from non-dialyzable fraction of sweet whey by hydrophobic interaction chromatography on phenyl-agarose. Biotechnology Letters 22, 413–416 (2000). https://doi.org/10.1023/A:1005649415447
Issue Date:
DOI: https://doi.org/10.1023/A:1005649415447