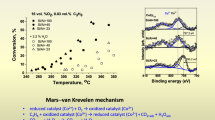
Abstract
At lower temperatures or in the presence of O2, metal oxides showed higher turnover frequency for the decomposition of N2O than corresponding metal ion exchanged ZSM-5.
Similar content being viewed by others
REFFERENCES
M.H. Thiemens, W.C. Trogler: Science, 251, 932 (1991).
R.M. Dell, F.S. Stone, P.F. Tiley: Trans. Faraday Soc., 49, 201 (1952).
E.R.S. Winter: J. Catal., 34, 431 (1974).
E.R.S. Winter: Trans. Faraday Soc., 57, 183 (1960).
K. Tanaka, K. Tamaru: Bull. Chem. Soc. Japan, 40, 420 (1967).
A.K. Vijh: J. Catal., 31, 51 (1973)
M. Iwamoto, K. Maruyama, N. Yamazoe, T. Seiyama: J. Phys. Chem., 81, 662 (1977).
S. Akbar, R. Joyner: J. Chem. Soc. Farady Trans. I, 77, 803 (1981).
J. Leglise, J.O. Petunchi, W.K. Hall: J. Catal., 86, 392 (1984).
Y. Li, J. N. Armor: Appl. Catal. B, 1, L21 (1992).
G.D. Lei, B.J. Adelman, J. Sárkány, W.M.H. Sachtler: Appl. Catal. B, 5, 245 (1995).
F. Kaptejin, G. Marban, J. Rodriguez-Mirasol, J.A. Moulijin: J. Catal., 167, 256 (1997).
A. Dandekar, M.A. Vannice: Appl. Catal. B, 22, 179 (1999).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Goto, T., Niimi, A., Hirano, K. et al. Comparative Study of Decomposition of N2O Over Metal Oxides and Metal Ion Exchanged ZSM-5 Zeolites. Reaction Kinetics and Catalysis Letters 69, 375–378 (2000). https://doi.org/10.1023/A:1005624606218
Issue Date:
DOI: https://doi.org/10.1023/A:1005624606218