Abstract
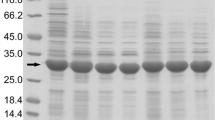
Escherichia coli PrlC is a trypsin-like proteinase regulating the cell cycle. The Escherichia coli prlC gene has been cloned into the pET28a prokaryotic expression vector. The recombinant fusion protein was produced mostly in the soluble, active form and the expression level amounted to approximately 70% of total protein. The recombinant proteinase was efficiently adsorbed to a resin containing immobilized Ni2+ via its amino terminal fusion hexahistidine tail to give a PrlC proteinase affinity column. The adsorbed fusion proteinase hydrolyzed 4-methylcoumaryl-7-amide of tert-butoxycarbonyl-l-valyl-l-prolyl-l-arginine (Boc-Val-Pro-Arg-NH-Mec), the specific substrate for the trypsin-like proteinase activity of E. coli PrlC.
Similar content being viewed by others
References
Conlin CA, Miller CG (1992) Cloning and nucleotide sequence of opdA, the gene encoding oligopepitdase A in Salmonella typhimurium. J. Bacteriol. 174: 1631–1640.
Conlin CA, Trun NJ, Miller CG (1992a) Escherichia coli prlC encodes an endopeptidase and is homologous to Salmonella typhimurium opdA gene. J. Bacteriol. 174: 5881–5887.
Conlin CA, Vimr ER, Miller CG (1992b) Oligopepitdase A is required for normal phage P22 development. J. Bacteriol. 174: 5869–5880.
Emr SD, Bassford PJ Jr (1982) Localization and processing of outer membrane and periplasmic proteins in Escherichia coli strains harboring export-specific suppressor mutations. J. Biol. Chem. 257: 5852–5860.
Jiang X, Zhang M, Ding Y, Yao J, Chen H, Zhu D, Muramatu M (1998) Escherichia coli prlC gene encodes a trypsin-like proteinase regulating the cell cycle. J. Biochem. 124: 980–985.
Kato M, Irisawa T, Morimoto Y, Muramatu M (1993) A trypsin inhibitor trans-4-guanidinomethylcyclohexanecarboxylic acid 4-tert-butylphenyl ester suppresses the onset of DNA synthesis in Escherichia coli cells synchronized by phosphate starvation. Biol. Pharm. Bull. 16: 552–557.
Kato M, Irisawa T, Ohtani M, Muramatu M (1992) Purification and characterization of proteinase In, a trypsin-like proteinase, in Escherichia coli. Eur. J. Biochem. 210: 1007–1014.
Murry MG, Thompson WF (1980) Rapid isolation of highmolecular-weight plant DNA. Nucl. Acids Res. 8: 4321–4325.
Novak P, Ray PH, Dev IK (1986) Localization and purification of two enzymes from Escherichia coli capable of hydrolyzing a signal peptide. J. Biol. Chem. 261: 420–427.
Schatz PJ, Beckwith J (1990) Genetic analysis of protein export in Escherichia coli. Annu. Rev. Genet. 24: 215–248.
Trun NJ, Silhavy TJ (1987) Characterization and in vivo cloning of prlC, a suppressor of signal sequence mutations in Escherichia coli K12. Genetics 116: 513–521.
Trun NJ, Silhavy TJ (1989) PrlC, a suppressor of signal sequence mutations in Escherichia coli, can direct the insertion of the signal sequence into the membrane. J. Mol. Biol. 205: 665–676.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jiang, X., Jin, Z., Cheng, N. et al. Fusion expression of Escherichia coli prlC gene and preparation of PrlC proteinase affinity column. Biotechnology Letters 22, 1741–1745 (2000). https://doi.org/10.1023/A:1005607000906
Issue Date:
DOI: https://doi.org/10.1023/A:1005607000906