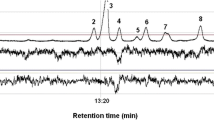
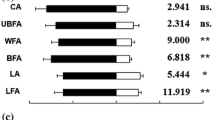
Abstract
Y-tube olfactometer bioassays and combined gas chromatography–electroantennographic detector (GC-EAD) analyses were performed to investigate the specific odors utilized as host location cues by the bark beetle parasitoid, Roptrocerus xylophagorum, originating from the southeastern United States. R. xylophagorum parasitizes several economically important holarctic bark beetle species and females oviposit preferentially on late larval stages. Both female and male parasitoids were tested with volatiles derived from host infestations of either Dendroctonus frontalis, the southern pine beetle, or Ips grandicollis, the southern pine engraver. Tested volatiles were steam distillates from the bark of loblolly pine, Pinus taeda, infested with larvae of the respective bark beetle species. Combined gas chromatography–mass spectrometry (GC-MS) was employed for identification and quantification of the compounds in the steam distillates. To confirm the EAD activity of identified compounds, GC-EAD analyses were repeated with a synthetic blend composed predominantly of compounds in the crude extracts that had revealed apparent electrophysiological activity. In Y-olfactometer bioassays, female parasitoids were attracted to both of the above-mentioned distillates. Male parasitoids were tested with one of the distillates but failed to respond. In GC-EAD analyses, the sexes displayed similar relative sensitivities to the components of the blends. Males exhibited generally higher amplitudes of response to the tested compounds than females. Monoterpene hydrocarbons associated with the constitutive resin of the host tree did not elicit significant EAD responses. Compounds known to be associated specifically with the host–tree complex, such as certain oxygenated monoterpenes, generated the greatest EAD responses. Female parasitoids were attracted by a synthetic blend composed of several of the EAD active oxygenated monoterpenes.
Similar content being viewed by others
REFERENCES
Aldrich, J. R. 1995. Chemical communication in the true bugs and parasitoid exploitation, pp. 318–363, in R. T. Cardé and W. J. Bell (eds.). Chemical Ecology of Insects 2. Chapman & Hall, New York.
Berisford, C. W. 1980. Natural enemies and associated organisms, pp. 31–54, in R. C. Thatcher, J. L. Searcy, J. E. Coster, and G. D. Hertel (eds.). The Southern Pine Beetle. USDA Forest Service Science Education Administration Technical Bulletin 1631.
Berisford, C. W., and Dahlsten, D. L. 1989. Biological control of Ips grandicollis (Eichoff ) (Coleoptera: Scolytidae) in Australia—a preliminary evaluation, pp. 81–93, in D. L. Kulhavy and M. C. Miller (eds.). Potential for Biological Control of Dendroctonus and Ips Bark Beetles. University of Texas Press, Austin, Texas.
Birgersson, G., and LEUFVéN, A. 1988. The influence of host tree response to Ips typographus (Coleoptera: Scolytidae) and fungal attack on production of semiochemicals. Insect Biochem. 18:761–770.
Birgersson, G., Dalusky, M. J., Espelie, K. E., and Berisford, C. W. 1992. Isolation and identification of kairomones utilized by southern pine beetle parasitoids, p. 30, in Proceedings: North American Forest Insect Work Conference. USDA Forest Service General Technical Report PNW-GTR-294.
Borden, J. H., Hunt, D. W. A., Miller, D. R., and Slessor, K. N. 1986. Orientation in forest Coleoptera: An uncertain outcome of responses by individual beetles to variable stimuli, pp.97–109, in T. L. Payne, M. C. Birch, and C. E. J. Kennedy (eds.). Mechanisms in Insect Olfaction. Oxford University Press. Oxford.
Bushing, R. W. 1965. A synoptic list of the parasites of Scolytidae (Coleoptera) in North America north of Mexico. Can. Entomol. 97:449–492.
Cortesero, A. M., De Moraes, C. M., Stapel, J. O., Tumlinson, J. H., and Lewis, W. J. 1997. Comparisons and contrasts in host-foraging strategies of two larval parasitoids with different degrees of host specificity. J. Chem. Ecol. 23:1589–1606.
Coyne, J. F., and Keith, G. C. 1972. Geographic survey of monoterpenes in loblolly and shortleaf pines. USDA Forest Service Research Paper So-79.
Dahlsten, D. L. 1982. Relationships between bark beetles and their natural enemies, pp. 140–182, in J. B. Mitton and K. B. Sturgeon (eds.). Bark Beetles in North American Conifers—A System for the Study of Evolutionary Biology. University of Texas Press, Austin.
Dix, M. E., and Franklin, R. T. 1981. Observations on the behavior of the southern pine beetle parasite Roptrocerus eccoptogastri Ratz. (Hymenoptera: Torymidae). J. Ga. Entomol. Soc. 16:239–248.
Dixon, W. N., and Payne, T. L. 1980. Attraction of entomophagous and associate insects of the southern pine beetle to beetle-and host-tree-produced volatiles. J. Ga. Entomol. Soc. 15:378–389.
Kudon, L. H., and Berisford, W. 1981. An olfactometer for bark beetle parasites. J. Chem. Ecol. 7:359–366.
Leal, W. S., Mochizuki, F., Wakamura, S., and Yasuda, T. 1992. Electroantennographic detection of Anomala cuprea Hope (Coleoptera: Scarabaeidae) sex pheromone. Appl. Entomol. Zool. 27:289–291.
Leufvén, A. 1987. Chemical interactions between the spruce bark beetle (Ips typographus), its host tree (Picea abies), and associated microorganisms (especially yeasts) during tree colonisation. PhD dissertation. Department of Chemical Ecology, Göteborg University, Sweden.
Leufvén, A., and Birgersson, G. 1987. Quantitative variation of different monoterpenes around galleries of Ips typographus attacking Norway spruce. Can. J. Bot. 65:1038–1044.
Mills, N. J. 1983. The natural enemies of scolytids infesting conifer bark in Europe in relation to the biological control of Dendroctonus spp. in Canada. Biocontrol News Inf. 4:305–328.
Mills, N. J. 1994. Parasitoid guilds: Defining the structure of the parasitoid communities of endopterygote insect hosts. Environ. Entomol. 23:1066–1083.
Mills, N. J., KrÜger, K., and Schlup, J.1991. Short-range host location mechanisms of bark beetle parasitoids. J. Appl. Entomol. 111:33–43.
Mirov, N. T. 1961. Composition of gum turpentines of pines. USDA Technical Bulletin 1239.
Moore, G. E. 1972. Southern pine beetle mortality in North Carolina caused by parasites and predators. Environ. Entomol. 1:58–65.
Parra, J. R. P., Vinson, S. B., Gomes, S. M., and Consoli, F. L. 1996. Flight response of Habrobracon hebetor (Say) (Hymenoptera: Braconidae) in a wind tunnel to volatiles associated with infestations of Ephestia kuehniella Zeller (Lepidoptera: Pyralidae). Biol. Control 6:143–150.
Payne, T. L. 1980. Life history and habits, pp. 7–28, in R. C. Thatcher, J. L. Searcy, J. E. Coster, and G. D. Hertel (eds.). The Southern Pine Beetle. USDA Forest Service Science Education Administration Technical Bulletin 1631.
Payne, T. L. 1989. Olfactory basis for insect enemies of allied species, pp. 55–69, in D. L. Kulhavy and M. C. Miller (eds.). Potential for Biological Control of Dendroctonus and Ips Bark Beetles. University of Texas Press, Austin.
RÖse, U. S. R., Lewis, W. J., and Tumlinson, J. H. 1998. Specificity of systemically released cotton volatiles as attractants for specialist and generalist parasitic wasps. J. Chem. Ecol. 24:303–319.
Salom, S. M., Birgersson, G., Payne, T. L., and Berisford, C. W. 1991. Electroantennogram responses of the southern pine beetle parasitoid Dinotiscus dendroctoni (Ashmead) (Hymenoptera: Pteromalidae) to potential semichemicals. J. Chem. Ecol. 17:2527–2538.
Samson, P. R. 1984. The biology of Roptrocerus xylophagorum (Hym.: Torymidae), with a note on its taxonomic status. Entomophaga 29:287–298.
Samson, P. R., and Smibert, J. 1986. Preliminary studies on the efficacy and establishment of Roptrocerus xylophagorum (Hym.: Torymidae), a parasitoid of Ips grandicollis (Col.: Scolytidae), in Australia. Entomophaga 31:173–182.
Sokal, R. R., and Rohlf, F. J. 1995. Biometry, the Principles and Practice of Statistics in Biological Research. W. H. Freeman and Co., New York.
Sullivan, B. T. 1997. The chemical ecology of host habitat location by larval parasitoids of the southern pine beetle, Dendroctonus frontalis Zimmermann: Olfactory cues and their possible sources. PhD dissertation. Department of Entomology, University of Georgia, Athens.
Sullivan, B. T., Berisford, C. W., and Dalusky, M. J. 1997. Field response of southern pine beetle parasitoids to some natural attractants. J. Chem. Ecol. 23:837–856.
Sullivan, B. T., Seltmann, K. C., and Berisford, C. W. 1999. A simple continuous-rearing technique for the bark beetle parasitoid, Roptrocerus xylophagorum (Ratzeburg). J. Entomol. Sci. 34:260–264.
Van Baaren, J., and Nénon, J. 1996. Host location and discrimination mediated through olfactory stimuli in two species of Encyrtidae. Entomol. Exp. Appl. 81:61–69.
Van Dijken, M. J., and Van Alphen, J. J. M. 1998. The ecological significance of differences in host detection behavior in coexisting parasitoid species. Ecol. Entomol. 23:265–270.
Vet, L. E. M., and Dicke, M. 1992. Ecology ofinfochemical use by natural enemies in a tritrophic context. Annu. Rev. Entomol. 37:141–172.
Vinson, S. B. 1976. Host selection by insect parasitoids. Annu. Rev. Entomol. 21:109–133.
Wassgren, A. B., and BergstrÖm, G. 1984. A simple effluent splitter for capillary gas chromatography. J. High Resolut. Chromatogr. Chromatogr. Commun. 7:154–155.
White, R. A., Jr., Agosin, M., Franklin, R. T., and Webb, J. W. 1980. Bark beetle pheromones: Evidence for physiological synthesis mechanisms and their ecological implications. Z. Angew. Entomol. 90:255–274.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pettersson, E.M., Sullivan, B.T., Anderson, P. et al. Odor Perception in the Bark Beetle Parasitoid Roptrocerus xylophagorum Exposed to Host Associated Volatiles. J Chem Ecol 26, 2507–2525 (2000). https://doi.org/10.1023/A:1005580528046
Issue Date:
DOI: https://doi.org/10.1023/A:1005580528046