Abstract
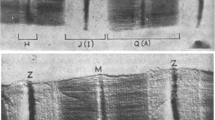
In the current study, the process of α-actinin binding to the myofibrillar Z-line was investigated to determine its mechanism. Pretreatment of rigor myofibrils with unlabeled α-actinin did not prevent or slow the incorporation of fluorescein skeletal α-actinin into myofibrils suggesting that incorporation was not the filling of empty binding sites but rather an exchange reaction. Further support for this was obtained using quantitative measures of labeled α-actinin incorporation and measures of total myofibrillar α-actinin. These results showed that there was no change in myofibrillar α-actinin content when up to 15% of the total α-actinin was the labeled protein. Measurement of the time-course of fluorescein α-actinin incorporation by quantitative fluorescence microscopy showed that the increase in Z-line fluorescence was well described by a rapid (unresolved) incorporation of fluorescence followed by a much slower phase. The slower phase was independent of fluorescein α-actinin concentration (2.5–160 nM) and had an apparent rate of 0.008–0.016 min−1. Pretreatment of myofibrils with fluorescein α-actinin followed by incubation with unlabeled α-actinin resulted in a decrease in Z-line fluorescence with an apparent rate of 0.021 min−1. The slow phase was interpreted as representing the dissociation rate of intrinsic Z-line α-actinin. Thus, the dissociation rate for the in situ interaction of α-actinin with actin appears to be three orders of magnitude slower than that determined from solution studies.
Similar content being viewed by others
References
Blanchard A, Ohanian V and Critchley D (1989) The structure and function of a-actinin. J Muscle Res Cell Motil 10: 280–289.
Casella JF, Craig SW, Maack DJ and Brown AE (1987) CapZ(36/32), a barbed end actin-capping protein, is a component of the Z-line of skeletal muscle. J Cell Biol 105: 371–379.
Edwards RJ, Goldstein MA, Schroeter JP and Sass RL (1989) The Z-band lattice in skeletal muscle in rigor. J Ultrastruc Mol Struct Res 102: 59–65.
Feramisco JR and Burridge K (1980) A rapid purification of a-actinin, filamin, and a 130,000-dalton protein from smooth muscle. J Biol Chem 255: 1194–1199.
Fritz JD, Swartz DR and Greaser ML (1989) Factors aecting polyacrylamide gel electrophoresis of high molecular weight myofibrillar proteins. Anal Biochem 180: 205–210.
Fukami K, Furuhashi K, Inagaki M, Endo T, Hatano S and Takenawa T (1992) Requirement of phosphatidylinositol 4,5-bisphosphate for a-actinin function. Nature 359: 150–152.
Fukami K, Endo T, Imamura Mand, Takenawa T (1994) a-actinin and vinculin are PIP2-binding proteins involved in signaling by tyrosine kinase. J Biol Chem 269: 1518–1522.
Goldmann WH and Isenberg G (1993) Analysis of filamin and a-actinin binding to actin by the stopped flow method. FEBS Lett 336: 408–410.
Goldstein MA, Michael LH, Schroeter JP and Sass RL (1988) Structural states in the Z band of skeletal muscle correlate with states of active and passive tension. J Gen Physiol 92: 113–119.
Gornall AG, Bardawill CJ and David MM (1949) Determination of serum protein by means of the biuret reaction. J Biol Chem 177: 751–766.
Grazi E, Trombetta G and Guidoboni M(1991) Binding of a-actinin to F-actin or to tropomyosin F-actin is a function of both a-actinin concentration and gel structure. J Muscle Res Cell Motil 12: 579–584.
Grazi E, Cuneo P, Magri E and Schweinbacher C (1992) Preferential binding of a-actinin to actin bundles from chicken gizzard. FEBS Lett 314: 348–350.
Grazi E, Cuneo P, Magri E, Schweinbacher C and Trombetta G (1993) Diusion hindrance and geometry of filament crossing account for the complex interactions of F-actin and a-actinin. Biochemistry 32: 8896–8901.
Kojima H, Ishijima A and Yanigida T (1994) Direct measurement of stiness of single actin filaments with and without tropomyosin by in vitro nanomanipulation. Proc Natl Acad Sci 91: 12962–12966.
Kuhlman PA, Ellis J, Critchley DR and Bagshaw CR (1994) The kinetics of interaction between the actin-binding domain of a-actinin and F-actin. FEBS Lett 339: 297–301.
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Lazarides E and Burridge K (1975) a-actinin: immunofluorescent localization or a muscle structural protein in nonmuscle cells. Cell 6:289–298.
Masaki T, Endo M and Ebashi S (1967) Localization of 6s component of a-actinin at the Z-band. J Biochem 62: 630–632.
McKenna NM, Meigs JB and Wang Y-L (1985) Exchangeability of a-actinin in living cardiac fibroblasts and muscle cells. J Cell Biol 101: 2223–2232.
Meyer RK and Aebi U (1990) Bundling of actin filaments by a-actinin depends on its molecular length. J Cell Biol 110: 2013–2024.
Miyata H, Yasuda R and Knosita K (1996) Strength and lifetime of the bond between actin and a-actinin studied with an optical trapping technique. Biochim Biophys Acta 1290: 83–88.
Morris EP, Nneji G and Squire JM (1990) The three-dimensional structure of the nemaline rod Z-band. J Cell Biol 111: 2961–2978.
Ohtsuka H, Yajima H, Maruyama K and Kimura S (1997) The N-terminal Z repeat 5 of connectin/titin binds to the C-terminal region of a-actinin. Biochim Biophys Res Comm 235: 1–3.
Robson RM, Goll DE, Arakawa N and Stromer MH (1970) Purification and properties of a-actinin from rabbit skeletal muscle. Biochim Biophys Acta 200: 296–318.
Sanger JW, Mittal B and Sanger JM (1984a) Analysis of myofibrillar structure and assembly using fluorescently labeled contractile proteins. J Cell Biol 98: 825–833.
Sanger JW, Mittal B and Sanger JM (1984b)Interaction of fluores-cently-labeled contractile proteins with the cytoskeleton in cell models.J Cell Biol 99: 918–928.
Sato M, Schwarz WH and Pollard TD (1987) Dependence of the mechanical properties of actin/a-actinin gels on deformation rate. Nature 325: 828–830.
Schroeter JP, Bretaudiere J-P, Sass RL and Goldstein MA (1996) Three-dimensional structure of the Z band in a normal mammalian skeletal muscle. J Cell Biol 133: 571–583.
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke MN, Olson BJ and Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150: 76–85.
Swartz DR, Greaser ML and Marsh BB (1990) Regulation of binding of subfragment-1 in isolated rigor myofibrils. J Cell Biol 111: 2989–3001.
Swartz DR, Greaser ML and Marsh BB (1993) Structural studies of rigor bovine myofibrils using fluorescence microscopy. II. Influence of sarcomere length on the binding of myosin subfrag-ment-1, a-actinin and G-actin to rigor myofibrils. Meat Sci 33: 157–190.
Swartz DR (1996) Covalent labeling of proteins for imaging applica-tions. Scanning Microscopy 10 (Suppl): 273–284.
Suzuki A, Goll DE, Singh I, Allen RE, Robson RM and Stromer MH (1976) Some properties of purified skeletal muscle a-actinin. J Biol Chem 251: 6860–6870.
Vigoreaux JO (1994) The muscle Z band: lessons in stress manage-ment. J Muscle Res Cell Motil 15: 237–255.
Wachsstock DH, Schwarz WH and Pollard TD (1993) Anity of a-actinin for actin determines the structure and mechanical proper-ties of actin filament gels. Biophys J 65: 205–214.
Yamaguchi M, Robson RM and Stromer MH (1983) Evidence for actin involvement in cardiac Z-lines and Z-line analogues. J Cell Biol 96: 435–442.
Yamaguchi M, Izumimoto M, Robson RM and Stromer MH (1985) Fine structure of wide and narrow vertebrate muscle Z-lines. A proposed model and computer stimulation of Z-line architecture. J Mol Biol 184: 621–644.
Yates LD and Greaser ML (1983) Quantitative determination of myosin and actin in rabbit skeletal muscle.J Mol Biol 168: 123–141.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Swartz, D.R. Exchange of α-actinin in isolated rigor myofibrils. J Muscle Res Cell Motil 20, 457–467 (1999). https://doi.org/10.1023/A:1005561912352
Issue Date:
DOI: https://doi.org/10.1023/A:1005561912352