Abstract
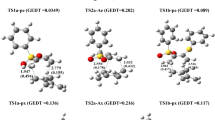
The semiempirical PM3 method is used to calculate the potential functions of internal rotation of the functional groups –SO2Cl, –NO2, –CH3, –OCH3, and –NH2 of benzenesulfonyl halide molecules (PhSO2Hal, Hal = F, Cl, Br, I) and twelve substituted derivatives of benzenesulfonyl chloride. Molecular conformations have been determined and internal rotation barriers of the functional groups have been calculated. For meta- and para-substituted benzenesulfonyl chlorides, the projection of the S–Hal bond is perpendicular to the plane of the benzene ring. The rotation barriers of the –SO2Hal group of benzenesulfonyl halides increase in the series Hal = F, Cl, Br, I. The rotation barriers of the –SO2Cl group of benzenesulfonyl chloride with meta- and para-substituents slightly increase with the electron-donor properties of the substituent. The rotation barriers of the functional groups of ortho-substituted benzenesulfonyl chlorides are 3 or 4 times as high as those of the meta- and para-isomers. For para-substituted benzenesulfonyl chlorides, the rotation barriers of the functional groups increase in the order –CH3, –NO2, –SO2Cl, –OCH3, –NH2.
Similar content being viewed by others
REFERENCES
J. Brunvoll and I. Hargittai, J. Mol. Struct., 30, 361–378 (1976).
V. M. Petrov, V. N. Petrova, V. V. Kislov, et al., Zh. Strukt. Khim., 40, No. 4, 653–664 (1999).
J. Brunvoll, O. Exner, I. Hargittai, et al., J. Mol. Struct., 117, 317–322 (1984).
I. Hargittai, The Structure of Volatile Sulfur Compounds, Reidel, Dordrecht (1985).
S. Oae (ed.), Organic Chemistry of Sulfur, Plenum, New York (1977).
B. G. Gnedin and S. N. Ivanov, Zh. Org. Khim., 14, No. 4, 772–777 (1978).
A. R. Haughhton, R. M. Laird, and M. J. Spence, J. Chem. Soc., Perkin Trans. II, No. 6, 637–643 (1975).
V. V. Kislov, S. N. Ivanov, and B. G. Gnedin, Zh. Org. Khim., 32, No. 5, 716–721 (1996).
R. V. Vizgert, L. I. Rubleva, and N. N. Maksimenko, ibid., 25, No. 4, 810–814 (1989).
L. I. Rubleva, S. N. Lyashchuk, N. N. Maksimenko, and R. V. Vizgert, ibid., 30, No. 6, 261–266 (1994).
J. J. P. Stewart, J. Comput. Chem., No. 10, 209–217 (1989).
J. J. P. Stewart, J. Comput.-Aided Mol. Des., 4, 1-105 (1990); J. J. P. Stewart, Mopac Manual (6th ed.); Frank J. Seiler Research Laboratory, US Air Force Academy CO 80840 (1990).
G. A. Chmutova and H. Albrecht, Zh. Obshch. Khim., 68, No. 11, 1854–1859 (1998).
R. W. Taft and I. C. Lewis, J. Am. Chem. Soc., 80, 2436 (1958).
R. W. Taft, S. Ehrenson, I. C. Lewis, and R. E. Glick, ibid., 81, 5352 (1959).
O. Exner, Coll. Czech. Chem. Commun., 31, 65 (1966).
I. F. Shishkov, N. I. Sadova, V. P. Novikov, and L. V. Vilkov, Zh. Strukt. Khim., 25, No. 2, 98–102 (1984).
M. Takezaki, N. Hirota, M. Terazima, et al., J. Phys. Chem. A, 101, 5190–5195 (1997).
J. H. Hog, L. Nygaard, and G. O. Sorensen, J. Mol. Struct., 7, 111 (1970).
O. G. Batyukhnova, N. I. Sadova, Yu. N. Syshchikov, et al., Zh. Strukt. Khim., 29, No. 4, 53–63 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kislov, V.V., Ivanov, S.N. & Petrov, V.M. Quantum Chemical Calculation of Conformations and Rotation Barriers of Functional Groups in Arylsulfonyl Halides. Journal of Structural Chemistry 41, 749–756 (2000). https://doi.org/10.1023/A:1004845815690
Issue Date:
DOI: https://doi.org/10.1023/A:1004845815690