Abstract
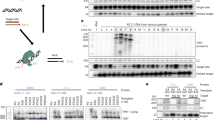
Tx1L is a site-specific non-LTR retrotransposon (NLR) that has been identified in the genome of Xenopus laevis. Using microinjection into Xenopus oocytes, several aspects of RNA expression by these elements were investigated. With constructs carrying various parts of the element we saw no evidence of promoter activity, unlike what has been shown for several other elements of this class. Tx1L transcription was induced by linking a whole element to a promoter that is active in oocytes. Among the RNAs produced, about half had 3′ ends located near the end of the element, suggesting that instructions for 3′ end formation are encoded in the element or its target. Deletion of the 3′ UTR of Tx1L and of surrounding target sequences indicated that these regions are not required for termination or processing of the RNA. PolyA or very A-rich sequences were added at these 3′ ends, despite the absence of canonical polyA addition signals. A significant proportion of non-A residues was found in the 3′ untemplated tails, and this is reminiscent of non-templated insertions often found at the 3′ junction of new genomic copies of some NLRs.
Similar content being viewed by others
References
Carroll, D., D.S. Knutzon & J.E. Garrett, 1989. Transposable elements in Xenopus, pp. 567-574 in Mobile DNA, edited by M.M. Howe and D.E. Berg. ASM Press, Washington, DC.
Chaboissier, M.-C., I. Busseau, J. Prosser, D.J. Finnegan & A. Bucheton, 1990. Identification of a potential RNA intermediate for transposition of the LINE-like element I factor in Drosophila melanogaster. EMBO J. 9: 3557-3563.
Chiang, C.C., J.C. Kennell, L.A. Wanner & A.M. Lambowitz, 1994. A mitochondrial retroplasmid integrates into mitochondrial DNA by a novel mechanism involving the synthesis of a hybrid cDNA and homologous recombination. Mol. Cell. Biol. 14: 6419-6432.
Cohen, D., S.M. Higman, S.I. Hsu & S.B. Horwitz, 1992. The involvement of a LINE-1 element in a DNA rearrangement upstream of the mdr1a gene in a taxol multidrug-resistant murine cell line. J. Biol. Chem. 267: 20248-20254.
Dawson, A., E. Hartswood, T. Paterson & D.J. Finnegan, 1997. A LINE-like transposable element in Drosophila, the I factor, encodes a protein with properties similar toothose of retroviral nucleocapsids. EMBO J. 16: 4448-4455.
Dombroski, B.A., Q. Feng, S.L. Mathias, D.M. Sassaman, A.F. Scott, H.H. Hazazian, Jr. & J.D. Boeke, 1994. An in vivo assay for the reverse transcriptase of human retrotransposon L1 in Saccharomyces cerevisiae. Mol. Cell. Biol. 14: 4485-4492.
Dumont, J.N., 1972. Oogenesis in Xenopus laevis (Daudin). 1. Stages of oocyte development in laboratory maintained animals. J. Morphol. 136: 153-180.
Eickbush, T.H., 1992. Transposing without ends: the non-LTR retrotransposable elements. New Biologist 4: 430-440.
Evans, J.P. & R.D. Palmiter, 1991. Retrotransposition of a mouse L1 element. Proc. Natl. Acad. Sci. USA 88: 8792-8795.
Feinberg, P.A. & B. Vogelstein, 1983. A technique for labeling restriction endonuclease fragments to high specific activity. Anal. Biochem. 132: 6-13.
Feng, Q., J.V. Moran, H.H. Kazazian, Jr. & J.D. Boeke, 1996. Human L1 retrotransposon encodes a conserved endonuclease required for transposition. Cell 87: 905-916.
Frohman, M.A., 1990. RACE: Rapid amplification of cDNA ends, pp. 28-38 in PCR Protocols: A Guide to Methods and Applications, edited by M. A. Innis, D. H. Gelfand, J. J. Sninsky and T. J. White. Academic Press, San Diego.
Furano, A.V., C.C. Somerville, P.N. Tsichlis & E. D'Ambrosio, 1986. Target sites for the transposition of rat long interspersed repeated DNA elements (LINEs) are not random. Nucleic Acids Res. 14: 3717-3727.
Furano, A.V. & K. Usdin, 1995. DNA ‘fossils’ and phylogenetic analysis. Using L1 (LINE-1, long interspersed repeated) DNA to determine the evolutionary history of mammals. J. Biol. Chem. 270: 25301-25304.
Gabriel, A. & J.D. Boeke, 1991. Reverse transcriptase encoded by a retrotransposon from the trypanosomatid Crithidia fasciculata. Proc. Natl. Acad. Sci. USA 88: 9794-9798.
Garrett, J.E. & D. Carroll, 1986. Tx1: a transposable element from Xenopus laevis with some unusual properties. Mol. Cell. Biol. 6: 933-941.
Garrett, J.E., D.S. Knutzon & D. Carroll, 1989. Composite transposable elements in the Xenopus laevis genome. Mol. Cell. Biol. 9: 3018-3027.
Hohjoh, H. & M.F. Singer, 1996. Cytoplasmic ribonucleoprotein complexes containing human LINE-1 protein and RNA. EMBO J. 15: 630-639.
Holmes, S.E., B.A. Dombroski, C.M. Krebs, C.D. Boehm & H.H. Kazazian, Jr., 1994. A new retrotransposable human L1 element from the LRE2 locus on chromosome 1q produces a chimaeric insertion. Nat. Genet. 7: 143-148.
Hutchison, C.A., III, S.C. Hardies, D.D. Loeb, W.R. Shehee & M.H. Edgell, 1989. LINEs and related retroposons: long interspersed repeated sequences in the eukaryotic genome, pp. 593–617 in Mobile DNA, edited by M.M. Howe and D. E. Berg. ASM Press, Washington, DC.
Ivanov, V.A., A.A. Melnikov, A.V. Siunov, I.I. Fodor & Y.V. Ilyin, 1991. Authentic reverse transcriptase is coded by jockey, a mobile Drosophila element related to mammalian LINEs. EMBO J. 10: 2489-2495.
Jensen, S. & T. Heidmann, 1991. An indicator gene for detection of germline retrotransposition in transgenic Drosophila demonstrates RNA-mediated transposition of the LINE I element. EMBO J. 10: 1927-1937.
Kinsey, J.A., 1993. Transnuclear retrotransposition of the Tad element of Neurospora. Proc. Natl. Acad. Sci. USA 90: 9384-9387.
Kolosha, V.O. & S.L. Martin, 1997. In vitro properties of the first ORF protein from mouse LINE-1 support its role in ribonucleoprotein particle formation during retrotransposition. Proc. Natl. Acad. Sci. USA 94: 10155-10160.
Luan, D.D. & T.H. Eickbush, 1995. RNA template requirements for target DNA-primed reverse transcription by the R2 retrotransposable element. Mol. Cell. Biol. 15: 3882-3891.
Luan, D.D. & T.H. Eickbush, 1996. Downstream 28S gene sequences on the RNA template affect the choice of primer and the accuracy of initiation by the R2 reverse transcriptase. Mol. Cell. Biol. 16: 4726-4734.
Luan, D.D., M.H. Korman, J.L. Jakubczak & T.H. Eickbush, 1993. Reverse transcription of R2Bm RNA is primed by a nick at the chromosomal target site: a mechanism for non-LTR retrotransposition. Cell 72: 595-605.
Martin, S.L., 1991. Ribonucleoprotein particles with LINE-1 RNA in mouse embryonal carcinoma cells. Mol. Cell. Biol. 11: 4804- 4807.
Maryon, E. & D. Carroll, 1989. Degradation of linear DNA by a strand-specific exonuclease activity in Xenopus laevis oocytes. Mol. Cell. Biol. 9: 4862-4871.
Mathias, S.L., A.F. Scott, H.H. Kazazian, Jr., J.D. Boeke & A. Gabriel, 1991. Reverse transcriptase encoded by a human transposable element. Science 254: 1808-1810.
McClure, M.A., 1991. Evolution of retroposons by acquisition or deletion of retrovirus-like genes. Mol. Biol. Evol. 8: 835-856.
McLean, C., A. Bucheton & D.J. Finnegan, 1993. The 5' untranslated region of the I factor, a long interspersed nuclear element-like retrotransposon of Drosophila melanogaster, contains an internal promoter and sequences that regulate expression. Mol. Cell. Biol. 13: 1042-1050.
Miki, Y., I. Nishisho, A. Horii, Y. Miyoshi, J. Utsunomiya, K.W. Kinzler, B. Vogelstein & Y. Nakamura, 1992. Disruption of the APC gene by a retrotransposable insertion of L1 sequence in a colon cancer. Cancer Res. 52: 643-645.
Minakami, R., K. Kurose, K. Etoh, Y. Furuhata, M. Hattori & Y. Sakaki, 1992. Identification of an internal cis-element essential for the human L1 transcription and a nuclear factor(s) binding to the element. Nucleic Acids Res. 20: 3139-3145.
Minchiotti, G. & P.P. Di Nocera, 1991. Convergent transcription initiates from oppositely oriented promoters within the 5' end regions of Drosophila melanogaster F elements. Mol. Cell. Biol. 11: 5171-5180.
Mizrokhi, L.J., S.G. Georgieva & Y.V. Ilyin, 1988. jockey, a mobile Drosophila element similar to mammalian LINEs, is transcribed from the internal promoter by RNA polymerase II. Cell 54: 685- 691.
Moran, J.V., S.E. Holmes, T.P. Naas, R.J. DeBerardinis, J.D. Boeke & H.H. Kazazian, Jr., 1996. High frequency retrotransposition in cultured mammalian cells. Cell 87: 917-927.
Niman, H.L., R.A. Houghten, L.E. Walker, R.A. Reisfeld, I.A. Wilson, J.M. Hogle & R.A. Lerner, 1983. Generation of protein-reactive antibodies by short peptides is an event of high frequency: implications for the structural basis of immune recognition. Proc. Natl. Acad. Sci. USA 80: 4949-4953.
Nur, I., E. Pascale & A.V. Furano, 1988. The left end of rat L1 (L1Rn, long interspersed repeat) DNA which is a CpG island can function as a promoter. Nucleic Acids Res. 16: 9233-9251.
Ogretman, B., H. Ratajczak, A. Kats, B.C. Stark & S.M. Gendel, 1993. Effects of staining of RNA with ethidium bromide before electrophoresis on performance of northern blots. Biotechniques 14: 932-035.
Packer, A.I., K. Manova & R.F. Bachvarova, 1993. A discrete LINE-1 transcript in mouse blastocysts. Dev. Biol. 157: 281-283.
Pelisson, A., D.J. Finnegan & A. Bucheton, 1991. Evidence for retrotransposition of the I factor, a LINE element of Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 88: 4907-4910.
Perou, C.M., R.J. Pryor, T.P. Naas & J. Kaplan, 1997. The bg allele mutation is due to a LINE1 element retrotransposition. Genomics 42: 366-368.
Pont-Kingdon, G., 1994. Construction of chimeric molecules by a two-step recombinant PCR method. BioTechniques 16: 1010- 1011.
Pont-Kingdon, G., E. Chi, S. Christensen & D. Carroll, 1997. Ribonucleoprotein formation by the ORF1 protein of the non-LTR retrotransposon Tx1L in Xenopus oocytes. Nucleic Acids Res. 25: 3088-3094.
Roth, M.B., A.M. Zahler & J.A. Stolk, 1991. A conserved family of nuclear phosphoproteins localized to sites of polymerase II transcription. J. Cell Biol. 115: 587-596.
Sambrook, J., E.F. Fritsch & T. Maniatis, 1989. Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor.
Schumann, G., I. Zundorf, J. Hofmann, R. Marschalek & T. Dingermann, 1994. Internally located and oppositely oriented polymerase II promoters direct convergent transcription of a LINE-like retroelement, the Dictyostelium repetitive element, from Dictyostelium discoideum. Mol. Cell. Biol. 14: 3074-3084.
Segal-Bendirdjian, E. & T. Heidmann, 1991. Evidence for a reverse transcription intermediate for a marked LINE transposon in tumoral rat cells. Biochem. Biophys. Res. Comm. 181: 863-870.
Sheets, M.D., M. Wu & M. Wickens, 1995. Polyadenylation of c-mos mRNA as a control point in Xenopus meiotic maturation. Nature 374: 511-516.
Skowronski, J., T.G. Fanning & M.F. Singer, 1988. Unit-length Line-1 transcripts in human teratocarcinoma cells. Mol. Cell. Biol. 8: 1385-1397.
Swergold, G.D., 1990. Identification, characterization, and cell specificity of a human LINE-1 promoter. Mol. Cell. Biol. 10: 6718-6729.
Swick, A.G., M. Janicot, T. Cheneval-Kastelic, J.C. McLenithan & M.D. Lane, 1992. Promoter-cDNA-directed heterologous protein expression in Xenopus laevis oocytes. Proc. Natl. Acad. Sci. USA 89: 1812-1816.
Tsang, S.S., X. Yin, C. Guzzo-Arkuran, V.S. Jones & A.J. Davison, 1993. Loss of resolution in gel electrophoresis of RNA: a problem associated with the presence of formaldehyde gradients. Biotechniques 14: 380-381.
Usdin, K. & A.V. Furano, 1988. Rat L (long interspersed repeated DNA) elements contain guanine-rich homopurine sequences that induce unpairing of contiguous duplex DNA. Proc. Natl. Acad. Sci. USA 85: 4416-4420.
Vaury, C., M.C. Chaboissier, M.E. Drake, O. Lajoinie, B. Dastugue & A. Pelisson, 1994. The Doc transposable element in Drosophila melanogaster and Drosophila simulans: genomic distribution and transcription. Genetica 93: 117-124.
Wallace, R.A., 1973. Protein incorporation by isolated amphibian oocytes. III. Optimum incubation conditions. J. Exp. Zool. 184: 321-324.
Wickens, M., 1990. How the messenger got its tail: addition of poly(A) in the nucleus. Trends Biochem. Sci. 15: 277-281.
Wyatt, J.R., E.J. Sontheimer & J.A. Steitz, 1992. Site-specific cross-linking of mammalian U5 snRNP to the 5' splice site before the first step of pre-mRNA splicing. Genes Dev. 6: 2542-2553.
Xiong, Y. & T.H. Eickbush, 1988. Functional expression of a sequence-specific endonuclease encoded by the retrotransposon R2Bm. Cell 55: 235-246.
Xiong, Y. & T.H. Eickbush, 1990. Origin and evolution of retroelements based upon their reverse transcriptase sequences. EMBO J. 9: 3353-3362.
Zimmerly, S., H. Guo, P.S. Perlman & A.M. Lambowitz, 1995. Group II intron mobility occurs by target DNA-primed reverse transcription. Cell 82: 545-554.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pont-Kingdon, G., Chi, E., Christensen, S. et al. RNA expression from a site-specific non-LTR retrotransposon microinjected into Xenopus oocytes. Genetica 104, 67–76 (1998). https://doi.org/10.1023/A:1003472404777
Issue Date:
DOI: https://doi.org/10.1023/A:1003472404777