Abstract
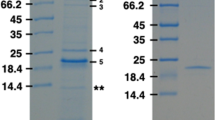
Porin from Roseobacter denitrificans was isolated and purified to homogeneity. The pore characteristics from this marine bacterium were compared to those of its phylogenetically closely related freshwater bacteria Rhodobacter capsulatus 37b4, Rhodobacter sphaeroides and Rhodopseudomonas blastica. The porin formed weakly cation-selective, general diffusion pores in lipid bilayer membranes. High transmembrane potentials caused channel closing in steps that were of one or two thirds of the initial on-steps indicating that the porin of R. denitrificans comprised three more or less independent channels similar to PhoE and OmpC of Escherichia coli and the porin of Rhodobacter capsulatus. Prediction of the secondary structure of the 36 N-terminal amino acid residues indicated two transmembrane β-strands similar to those of the porins of Rhodobacter capsulatus 37b4 and Rhodopseudomonas blastica. Differences of the single channel conductivities between the porin of R. denitrificans and those of the related freshwater bacteria show that R. denitrificans evolved porin channels that are well adapted to the marine habitat.
Similar content being viewed by others
References
Benz R, Janko K & Läuger P (1979) Ionic selectivity of pores formed by the matrix protein (porin) of Escherichia coli. Biochim. Biophys. Acta 551: 238–247
Benz R, Schmid A & Hancock REW (1985) Ion selectivity of gramnegative bacterial porins. J. Bacteriol. 162: 722–727
Benz R, Woitzik D, Flammann HT & Weckesser J (1987) Pore forming activity of the major outer membrane protein of Rhodobacter capsulatus in lipid bilayer membranes. Arch. Microbiol. 14: 226–230
Benz R & Bauer K (1988) Permeation of hydrophilic molecules through the outer membrane of gram-negative bacteria. Eur. J. Biochem. 176: 1–19
Benz R (1988) Structure and function of porins from Gram-negative bacteria. Annu. Rev. Microbiol. 42: 359–393
Benz R (1994) Solute uptake through bacterial outer membranes. In: Bacterial Cell Wall, pp. 397–423 Edited by R. Hackenbek & J.-M. Ghuysen. Amsterdam: Elsevier
Butz S, Benz R, Wacker T, Welte W, Lustig A, Plapp R & Weckesser J (1993) Biochemical characterization and crystallization of porin from Rhodospseudomonas blastica. Arch. Microbiol. 159: 301–307
Capaldi RA & Vanderkooi G (1972) The low polarity of many membrane proteins. Proc. Natl. Acad. Sci. USA 69: 930–932
Cowan SW, Schirmer T, Rummel G, Steiert M, Gosh R, Pauptit RA, Jansonius JN & Rosenbusch JP (1992) Crystal structures explain functional properties of two E. coli porins. Nature 358: 727–733
Fuerst JA, Hawkins JA, Holmes A, Sly LI, Moore CJ & Stackebrandt E (1993) Porphyrobacter neustonensis gen. nov., sp. nov., an aerobic bacteriochlorophyll-synthesizing budding bacterium from fresh water. Int. J. Syst. Bacteriol. 43: 125–134
Imhoff JF (1989) The family Ectothiorhodospiraceae. In: Staley JT, Bryant MP, Pfennig N, Holt JG (Eds.) Bergey's manual of systematic bacteriology, 1st edn, vol 3. Williams & Wilkins, Baltimore, pp 1654–1658
Jap BK (1989) Molecular design of PhoE porin and its functional consequences. J. Mol. Biol. 205: 407–419
Karshikoff A, Spassov V, Cowan SW, Ladenstein R & Schirmer T (1994) Electrostatic properties of two porin channels from Escherichia coli. J. Mol. Biol. 240: 372–84
Kreusch A, Neubüser A, Schiltz E, Weckesser J & Schulz GE (1994) Structure of the membrane channel porin from Rhodopseudomonas blastica at 2.0 Å resolution. Prot. Sci. 3: 58–63
Kyte J & Doolittle RF (1982) A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 157: 105–132
Lowry OH, Roberts NJ, Farr AL & Randall RJ (1951) Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–275
Neumann U, Mayer H, Schiltz E, Benz R & Weckesser J (1995) Lipopolysaccharide and porin of Roseobacter denitrificans, confirming its phylogenetic relationship to the α-3-subgroup of Proteobacteria. Microbiology 141: 2013–2017
Nikaido H (1992) Porins and specific channels of bacterial outer membranes. Molec. Microbiol. 6: 435–442
Nikaido H & Rosenberg EY (1981) Effect of solute size on diffusion through the transmembrane pores of the outer membrane of Escherichia coli. J. Gen. Physiol. 77: 121–135
Rodriguez-Vico F, Martinez-Cayuela M, Garcia-Peregrin E & Ramirez H (1989) A procedure for eliminating interferences in the Lowry method of protein determination. Anal Biochem 183: 275–278
Schiltz E, Kreusch A, Nestel U & Schulz GE (1991) Primary structure of porin from Rhodobacter capsulatus. Eur. J. Biochem. 199: 587–594
Sen K, Hellman J & Nikaido H (1988) Porin channels in intact cells of Escherichia coli are not affected by Donnan potentials across the outer membrane. J. Biol. Chem. 263: 1182–1187
Shioi Y (1986). Growth characteristics and substrate specifity of aerobic photosynthetic bacterium Erythrobacter sp. (OCh 114). Plant Cell Physiol. 27: 567–572
Todt CJ & McGroarty JE (1992) Involvement of histidine-21 in the pH-induced switch in porin channel size. Biochemistry 31: 10479–82
Vogel H & Jähnig F (1986) Models for the structure of outer-membrane proteins of Escherichia coli derived from Raman spectroscopy and prediction methods. J. Mol. Biol. 190: 191–199
Weckesser J, Zalman S & Nikaido H (1984) Porin from Rhodopseudomonas sphaeroides. J Bacteriol. 159: 199–205
Weiss MS, Kreusch A, Schiltz E, Nestel U, Welte W Weckesser J & Schulz GE (1991) The structure of Porin from Rhodobacter capsulatus at 1.8 Å resolution. FEBS Lett. 280: 379–382
Wolf E, Zahr M, Benz R, Imhoff JF, Lustig A, Schiltz E, Stahl-Zeng J & Weckesser J (1996) The porins from the halophilic species Ectothiorhodospira shaposhnikovii and Ectothiorhodospira vacuolata. Arch. Microbiol. 166: 169–175
Woitzik D, Weckesser J, Benz R, Stevanovic S, Jung G & Rosenbusch JP (1990) Porin of Rhodobacter capsulatus: biochemical and functional characterization. Z. Naturforsch. 45c: 576–582
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Neumann, U., Maier, E., Schiltz, E. et al. Characterization of Porin from Roseobacter denitrificans. Antonie Van Leeuwenhoek 72, 135–140 (1997). https://doi.org/10.1023/A:1000262802010
Issue Date:
DOI: https://doi.org/10.1023/A:1000262802010