Abstract
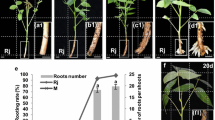
In vitro rooting constitutes a difficult step during the micropropagation process of forest species. The successive media culture technique represents one way to overcome this barrier and includes modifying physical (e.g. photoperiod) and chemical (e.g. flavonoids) factors during the rooting phases. The aim of this study was to obtain a successive media protocol based on the incorporation of flavonoids during the in vitro rooting of Nothofagus nervosa. The factors evaluated were the type, concentration, and combination of flavonoids in relation to the rooting phases, the presence of IBA in the culture medium, the photoperiod, and the effect of flavonoids on total tissue peroxidase activity. The photoperiod used included a darkness period during the rooting induction stage and the presence of 0.61 µMIBA in the culture medium. The results showed that flavonoid incorporation at a concentration of 20 µM accelerated the appearance of roots and improved the quality of the already formed ones. Each type and concentration of flavonoid produced different responses, with (±)naringenin giving the best results. The latter caused a peak in the peroxidase activity that was absent in the control treatments. This work allowed identifying an optimized rooting protocol through a successive media culture technique that improved the speed of appearance, as well as the quantity and quality of roots for a single N. nervosa clone.
Similar content being viewed by others
References
Altamura M.M. 1996. Root histogenesis in herbaceous and woody explants cultured in vitro. A critical review. Agronomie 16: 589-602.
Bellarosa R. 1989. Oak (Quercus sp). In: Bajaj Y.P.S. (ed.), Biotechnology in Agriculture and Forestry 5. Trees II. Springer-Verlag, pp. 387-401.
Blakesley D., Weston G.D. and Hall J.F. 1991. The role of endogenous auxin in root initiation. Part I: Evidence from studies in auxin application and analysis of endogenous levels. Plant Growth Regulation 10: 341-353.
Berthon J., Boyer N. and Gaspar T. 1987. Sequential rooting media and rooting capacity of Sequoiadendron giganteum in vitro. Plant Cell Report 6: 341-344.
Berthon J., Battraw M., Gaspar T. and Boyer N. 1993. Early test using phenolic compounds and peroxidase activity to improve in vitro rooting of Sequoiadendron giganteum. Saussurea 24: 7-13.
Bonga J. 1987. Clonal propagation of mature trees: Problems and possible solutions. In: Bonga J. and Durzan D. (eds), Cell and Tissue Culture in Forestry. Martinus Nijhoff Vol I., pp. 249-271.
Bradford M.M. 1976. A rapid and sensitive method for the quantification of microgram quantities of proteins using the principle of protein-dye binding. Anal. Biochem. 72: 248-254.
Calderon Baltierra X., Perez F., Salazar C. and Sanhueza V. 1994. Physiological aspects of in vitro preserving chilean native forest species: Gomortega keule and Nothofagus alpina. Noticiero de Biologia 2: 15-15.
Calderon Baltierra X., Martinez Pastur G., Jofre M.P. and Arena M. 1998. Activity variation of peroxidase during in vitro rooting of Nothofagus nervosa and Nothofagus antarctica. Phyton 62: 137-144.
Chalupa V. 1983. Micropropagation of conifer and broadleaved forest trees. Communicationes Instituti Forestalis Cechosloveniae 13: 7-39.
Davis F., Davis T. and Kester D. 1994. Commercial importance of adventitious rooting to horticulture. In: Davis T. and Haissig B. (eds), Biology of Adventitious Root Formation. Plenum Press, NewYork, pp. 53-59.
De Klerk G. 1996. Markers of adventitious root formation. Agronomie 16: 609-616.
Druart P., Kevers C., Boxus P. and Gaspar T. 1982. In vitro promotion of root formation by apple shoots through darkness effect on endogenous phenols and peroxidases. Z Pflanzenphysiol Bd 108: 429-436.
Faivre-Rampant O., Kevers C. and Gaspar T. 2000. IAA-oxidase activity and auxin protectors in non-rooting, rac, mutant shoots of tobacco in vitro. Plant Science 153: 73-80.
Gaspar T. 1981. Rooting and flowering to antagonistic phenomena from a hormonal point of view. In: Jeeffcoat B. (ed.), Aspects and Prospects of Plant Growth Regulators. British Plant Growth Regulator Group, Wantage Monograph 6., pp. 39-49.
Gaspar T., Penel C., Castillo F. and Greppin H. 1985. A two-step control of basic and acidic peroxidases and its significance for growth and development. Physiologia plantarum 64: 418-423.
Gaspar T., Moncousin C. and Greppin H. 1990. The place and role of exogenous and endogenous auxin in adventitious root formation. In: Millet B. and Greppin H. (eds), Intra-and intercellular communications in plants. INRA, Paris, pp. 125-139.
Gaspar T., Penel C., Hagege D. and Greppin H. 1991. Peroxidases in plant growth, differentiation and development processes. In: Lobarzewski J., Greppin H., Penel H. and Gaspar T. (eds), Biochemical Molecular and Physiological Aspects of Plant Peroxidases. University of Marie Curie, Sklodowska, pp. 249-280.
Gaspar T., Kevers C., Hausman J.F. and Ripetti V. 1993. Peroxidase activity and endogenous free auxin during adventitious root formation. Plant Growth Regulation (Lancaster Meeting).
George E., Puttock D. and George H. 1987. Plant Culture Media. Formulations and Uses. Exegetics Limited, England, 454 pp. 1.
Hausman J.F. 1993. Changes in peroxidase activity, auxin level and ethylene production during root formation by poplar shoots raised in vitro. Plant Growth Regulation 13: 263-268.
Jacobs M. and Rubery P.H. 1988. Naturally occurring auxin transport regulators. Science 241: 346-349.
Jordan M. and Velozo J. 1992. Micropropagacion de rauli (Nothofagus alpina). In: Segundo Taller Silvicola. Santiago, Chile, pp. 57-64.
Jordan M., Velozo J. and Sabja A. 1996. Organogenesis in vitro of Nothofagus alpina, Fagaceae. Plant Cell Report 15: 795-798.
Kevers C., Hausman J., Faivre-Rampant O., Evers D. and Gaspar T. 1997. Hormonal control of adventitious rooting: Progress and questions. Angew. Bot. 71: 71-79.
Mader M. 1992. Compartmentation of peroxidase isoenzymes in plant cells. In: Penel C., Gaspar T. and Greppin H. (eds), Plant Peroxidases 1980-1990. University of Geneva, pp. 37-46.
Martinez Pastur G. and Arena M. 1995. In vitro propagation of Nothofagus obliqua (Fagaceae). Australian Journal of Botany 43: 601-607.
Martinez Pastur G. and Arena M. 1996. In vitro propagation of Nothofagus nervosa. Phyton 58: 1-7.
Martinez Pastur G. and Arena M. 1997. Micropropagacion de Nothofagus pumilio (Poepp. et. Endl.) Krasser. Bosque 18: 43-50.
Martinez Pastur G. and Arena M. 1999. In vitro propagation of juvenile Nothofagus leoni Espinosa (Fagaceae). Journal of Forest Research 4: 295-298.
Martinez Pastur G., Arena M. and Caso O. 1997. In vitro propagation of Nothofagus antarctica from sphaeroblasts and saplings. Biocell 21: 231-236.
Martinez Pastur G., Arena M. and Caso O. 1998. Physiological variability arising from in vitro propagation of Nothofagus obliqua. Biocell 22: 149-155.
Martinez Pastur G., Arena M. and Curvetto N. 2000. Calcium and boron for in vitro rooting of-Nothofagus nervosa. Biocell 24: 65-71.
Stenlid G. 1976. Effects of flavonoids on the polar transport of auxins. Physiol. Plant. 38: 262-266.
Van Der Berg B.M., Chibbar R.N. and Van Huystee R.B. 1983. A comparative study of a cationic peroxidase from peanut and an anionic peroxidase from Petunia. Plant Cell Report 2: 304-307.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martínez Pastur, G., Arena, M., Curvetto, N. et al. Successive media to improve the in vitro rhizogenesis of Nothofagus nervosa (Phil.) Dim. et Mil.. New Forests 26, 201–215 (2003). https://doi.org/10.1023/A:1024421906002
Issue Date:
DOI: https://doi.org/10.1023/A:1024421906002



