Abstract
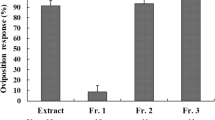
Host-recognizing kairomones for the stinging behavior of the parasitic wasp, Anisopteromalus calandrae, were identified on host azuki bean weevil larvae, Callosobruchus chinensis (L.). The kairomones were extracted with acetone from Chinese green beans, from which emerged wasps and host weevils had been removed. The kairomones are a mixture of triacylglycerols and fatty acids, each of which is separately active, and with no observable synergistic effect between them. These compounds are known to be constituents of an oviposition-marking pheromone of host azuki bean weevils. However, they differ from the previously reported saturated hydrocarbons and diacylglycerols of the kairomone that another parasitic wasp, Dinarmus basalis, uses for the host recognition of C. chinensis. Thus, A. calandrae and D. basalis selectively utilize different constituents of the oviposition-marking pheromone of C. chinensis as host-recognizing kairomones.
Similar content being viewed by others
REFERENCES
Arbogast, R. T. and Mullen, M. A. 1990. Interaction of maize weevil (Coleoptera: Curculionidae) and parasitoid Anisopteromalus calandrae (Hymenoptera: Pteromalidae) in a small bulk of stored corn. J. Econ. Entomol. 83:2462–2468.
Carroll, K. K. 1961. Separation of lipid classes by chromatography on florisil. J. Lipid Res. 2:135–141.
Cline, L. D., Press, J. W., and Flaherty, B. R. 1985. Suppression of the rice weevil, Sitophilus oryzae (Coleoptera: Curculionidae), inside and outside of burlap, woven polypropylene, and cotton bags by the parasitic wasp, Anisopteromarus calandrae (Hymenoptera: Pteromalidae). J. Econ. Entomol. 78:835–838.
Ghani, M. A. and Sweetman, H. L. 1955. Ecological studies on the granary weevil parasite, Aplastomorpha calandrae (Howard). Biologia (Lahore) 1:115–139.
Golding, B. T., Griffin, A. L., and Robinson, D. H. 1993. Synthesis of a “reverse ester” analogue of 1,2-sn-diglycerides from (S)-1,2-di-O-isopropylidene-glycerol; efficient, stereospecific nucleophilic displacement via a triflate at glycerol C-2. Tetrahedron Lett. 34:6459–6462.
Hayashi, A. 1991. Chapter 9. Handling of Lipids, pp. 149–170, in T. Goto, T. Shiba, and T. Matsuura (eds.). Introduction to Experiments for Organic Chemistry. Kagakudojin, Kyoto (in Japanese).
Honda, H. and Ohsawa, K. 1990. Chemical ecology for stored product insects. J. Pestic. Sci. 15:263–270. (in Japanese with an English abstract).
Kumazaki, M., Matsuyama, S., Suzuki, T., Kuwahara, Y., and Fujii, K. 2000. Parasitic waspDinarmus basalis, utilizes oviposition-marking pheromone of host azuki bean weevils as hostrecognizing kairomone. J. Chem. Ecol. 26:2677–2695.
Leblanc, Y., Fitzsimmons, B. J., Adams, J., Perez, F., and Rokach, J. 1986. The total synthesis of 12-HETE and 12, 20-DiHETE. J. Org. Chem. 51:789–793.
Matsumoto, H., Tebayashi, S., Kuwahara, Y., Matsuyama, S., Suzuki, T., and Fujii, K. 1994.Identification of taxifolin present in the azuki bean as an oviposition stimulant of the azuki bean weevil. J. Pestic. Sci. 19:181–186.
Murata, T. and Takahashi, S. 1973. Analysis of triglycerides mixtures by gas chromatography-mass spectrometry. Anal. Chem. 45:1816–1823.
Ohshima, K., Honda, H., and Yamamoto, I. 1973. Isolation of an oviposition marker from azuki bean weevil, Callosobruchus chinensis (L.). Agric. Biol. Chem. 37:2679–2680.
Okamoto, K. 1971. The synchronization of the life cycles between Callosobruchus chinensis (L) and its parasite Anisopteromalus carandrae (Howard). Jpn. J. Ecol. 20:233–237 (in Japanese with an English synopsis).
Okamoto, K. 1972. The synchronization of the life cycles between Callosobruchus chinensis (L) and its parasite Anisopteromalus calandrae (Howard) II. The relationship between the development of the parasite and the developmental stage of the host. Jpn. J. Ecol. 22:238–244 (in Japanese with an English synopsis).
Press, J. W., Cline, L. D., and Flaherty, B. R. 1984. Suppression of residual population of the rice weevil, Sitophilus oryzae, by the parasitic wasp, Anisopteromalus calandrae. J. Ga. Entomol. Soc. 19:110–113.
Sakai, A., Honda, H., and Yamamoto, I. 1986. Oviposition marking pheromone of two bean weevils,Callosobruchus chinensis and Callosobruchus maculatus. J. Pestic. Sci. 11:163–168.
Smith, L. 1992. Effect of temperature on life history characteristics of Anisopteromalus calandrae (Hymenoptera: Pteromalidae) parasitizing maize weevil larvae in corn kernels. Environ. Entomol. 21:877–887.
Sokal, R. R. and Rohlf, F. J. 1973. Introduction to Biostatistics. Kyoritsu Publishing, Tokyo, 449 pp. (K. Fujii Japanese translation, 1983).
Tanaka, K., Ohsawa, K., Honda, H., and Yamamoto, I. 1981. Copulation release pheromone, electin, from the azuki bean weevil (Callosobruchus chinensis L.). J. Pestic. Sci. 6:75–82.
Tebayashi, S., Matsuyama, S., Suzuki, T., Kuwahara, Y., Nemoto, T., and Fujii, K. 1995. Quercimeritrin: The third oviposition stimulant of the azuki bean weevil from the host azuki bean. J. Pestic. Sci. 20:299–305.
Ueno, T., Kuwahara, Y., Fujii, K., Taper, M. L., Toquenaga, Y., and Suzuki, T. 1990. D-Katechin:An oviposition stimulant of azuki bean weevil Callosobruchus chinensis in the host azuki bean. J. Pestic. Sci. 15:573–578.
Utida, S. and Nagasawa, S. 1949. On the developmental period and that of adult life of Neocatolaccus mamezophagus Ishii et Nagasawa, a parasite wasp of the azuki bean weevil. Kontyu 17:1–21 (in Japanese).
van den Assem, J., Putters, F. A., and Prints, T. C. 1984. Host quality effects on sex ratio of the parasitic wasp Anisopteromalus calandrae (Chalcidoidea, Pteromalidae). Neth. J. Zool. 34:33–62.
Williams, R. N. and Floyd, E. H. 1971. Effect of two parasites, Anisopteromalus calandrae and Choetospila elegans, upon populations of the maize weevil under laboratory and natural conditions. J. Econ. Entomol. 64:1406–1408.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Onodera, J., Matsuyama, S., Suzuki, T. et al. Host-Recognizing Kairomones for Parasitic Wasp, Anisopteromalus calandrae, from Larvae of Azuki Bean Weevil, Callosobruchus chinensis . J Chem Ecol 28, 1209–1220 (2002). https://doi.org/10.1023/A:1016285801824
Issue Date:
DOI: https://doi.org/10.1023/A:1016285801824