Abstract
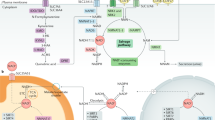
A hypothesis as to the nature of the ageing process is presented. Our studies lead us to emphasise that ageing is a stochastic process; it is individualistic, it is species specific, it is different for individuals within a species, it is organ and tissue specific as well as varying among individual cells of a tissue. Human ageing is a slow process which takes place over decades; it is a cellular process in a dynamic equilibrium of continuing damage and repair. Tissues are damage mosaics. We hypothesise that the characteristic of ageing post-mitotic tissues is the progressive, slow loss of cells; those cells which no longer adequately function are removed by an apoptotic or non-inflammatory necrotic process. A case is made for an encompassing role for coenzyme Q10 in the regulation of systemic disease, cellular metabolism and ageing.
Similar content being viewed by others
References
Crane FL, Sun IL, Barr R and Morre DJ (1984) Coenzyme Q in Golgi apparatus membrane redox activity and proton uptake. In: Folkers K and Yamamura Y (eds) Biomedical and Clinical Aspects of Coenzyme Q, Vol 4, pp 77-85. Elsevier, Amsterdam
Crane FL, Sun IL, Crowe RA, Alcain FJ and Low H (1994) Coenzyme Q10, plasma membrane oxidase and growth control. Mol Aspects Med 15 (Suppl.): 1-11
Gille L and Nohl H (2000) The existence of a lysosomal redox chain and the role of ubiquinone. Arch Biochem Biophys 375: 347-354
Glockshuber R (1999) Where do the electrons go? Nature 401: 30-31
Karim S, Winkler E and Klingenberg M (2000) Coenzyme Q is an obligatory cofactor for uncoupling protein function. Nature 408: 609-613
Kopsidas G, Kovalenko S and Heffernan D et al. (2000) Tissuemitochondrial DNA changes: a stochastic system. Ann NY Acad Sci 908: 226-243
Kopsidas G, Zhang C, Yarovaya N et al. (2002) Stochastic mitochondrial DNA changes: bioenergy decline in type I skeletal muscle fibres correlates with a decline in the amount of amplifiable full-length mtDNA. Biogerontology 3: 29-36 (this issue)
Kovalenko S, Harms PJ and Tanaka M et al. (1997) Method for in situ investigation of mitochondrial DNA deletions. Human Mutation 10: 489-495
Kovalenko S, Kopsidas G and Kelso J et al. (1998a) Tissue-specific distribution of multiple mitochondrial DNA rearrangements during human aging. Ann NY Acad Sci 854: 171-181
Kovalenko S, Kopsidas G and Islam MM et al. (1998b) The ageassociated decrease in the amount of amplifiable full-length mitochondrial DNA in human skeletal muscle. Biochem Mol Biol Int 46(6): 1233-1241
Lawen A, Martunus RD and McMullen et al. (1994) The universality of bioenergetic disease: the role of mitochondrial mutation and the putative inter-relationship between mitochondria and plasma membrane NADH oxidoreductase. Mol Aspects Med 15: 13-27
Linnane AW, Marzuki S, Ozawa T and Tanaka M (1989) Mitochondrial DNA mutations as a major contributor to aging and degenerative diseases. Lancet 1: 642-645
Linnane AW, Zhang C and Nagley P et al. (1992) Mitochondrial DNA mutation and the ageing process: bioenergy and pharmacological intervention. Mut Res 275: 195-208
Linnane AW, Esposti MD and Generowicz M et al. (1995) The universality of bioenergetic disease and amelioration with redox therapy. Biochim Biophys Acta 1271: 191-194
Linnane AW, Kovalenko S and Gingold EB (1998) The universality of bioenergetic disease: age-associated cellular bioenergetic degradation and amelioration therapy. Ann NY Acad Sci 854: 202-213
Linnane AW (2000) Human skeletal muscle ageing: a life style determinant. Proc of the Fifth Shizuoka Forum on Health and Longevity, pp 43-52. Shizuoka Perpetual Government, Japan
Linnane AW, Kopsidas G, Zhang C, Yarovaya N, Kovalenko S, Papakostopoulos P, Eastwood H, Graves S and Richardson M (2001) Cellular redox activity of coenzyme Q10: effect of CoQ10 supplementation on human skeletal muscle. Free Rad Res, in press
Nagley P, Zhang C and Martinus RD et al. (1993) Mitochondrial DNA mutation and human ageing: molecular biology, bioenergetics, and redox therapy. In: DiMauro S and Wallace DC (eds) Mitochondrial DNA Human Pathology, pp 137-157. Raven Press, New York
Olivetti G, Melissari M, Capasso JM and Anversa P (1991) Cardiomyopathy of the aging human heart: myocyte loss and reactive cellular hypertrophy. Circulation Res 68: 1560-1568
Pfannschmidt T, Nilsson A and Allen JF (1999) Photosynthetic control of chloroplast gene expression. Nature 397: 625-628
Rosenfeldt FL, Pepe S and Ou R et al. (1999) Coenzyme Q10 improves the tolerance of the senescent myocardium to aerobic and ischemic stress: studies in rats and in human atrial tissue. BioFactors 9: 291-299
Rowland MA, Nagley P and Linnane AW et al. (1998) Coenzyme Q10 treatment improves the tolerance of the senescent myocardium to pacing stress in the rat. Cardiovascular Res 40: 165-173
Smith J, Ladi E, Mayer-Proschel M and Noble M (2000) Redox state is a central modulator of the balance between self-renewal and differentiation in a dividing glial precursor cell. Proc Natl Acad Sci USA 97: 10032-10037
Sun IL and Crane FL (1990) In: Oxidation Reduction at the Plasma Membrane, Vol 1, pp 257-280. CRC Press, Boca Raton, Florida
Yarovaya NO, Kramarova L, Borg J, Kovalenko SA, Caragounis A and Linnane AW(2002) Age-related atrophy of rat soleus muscle is accompanied by changes in fibre type composition, bioenergy decline and mtDNA rearrangements. Biogerontology 3: 25-27 (this issue)
Zhang C, Bills M and Quigley A et al. (1997) Varied prevalence of age-associated mitochondrial DNA deletions in different species and tissues: a comparison between human and rat. Biochem Biophys Res Comm 230: 630-635
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Linnane, A.W. Cellular coenzyme Q10 redox poise constitutes a major cell metabolic and gene regulatory system. Biogerontology 3, 3–6 (2002). https://doi.org/10.1023/A:1015266204771
Issue Date:
DOI: https://doi.org/10.1023/A:1015266204771