Abstract
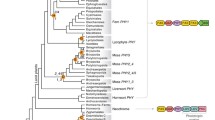
Knowledge of the genes in gymnosperms encoding the apoproteins of the plant photoreceptor phytochrome is currently scanty as for gymnosperm nuclear protein coding sequences in general. Here we report two complete cDNA-derived sequences which code for two different types of gymnosperm phytochrome. One sequence stems from Norway spruce (Picea abies) and the other from Scots pine (Pinus sylvestris). More detailed studies have shown that both types of phytochrome gene are present in Norway spruce. From phylogenetic analyses, these types appear to branch off from progenitors that are also the common ancestors of the angiosperm PHYA/PHYC and PHYB/PHYD/PHYE lineages. Partial phytochrome sequences of other gymnosperms cluster with either the one type or the other of the gymnosperm phytochrome genes characterized here. Southern blot analysis of Picea DNA using probes derived from the full-length Picea gene indicated a family of at least five members. Whether they code for new types may be doubted since only two phylogenetic clusters were found. Studies using RNA-PCR of Picea RNA extracted from either light- or dark-grown seedlings indicated that the steady-state levels of the transcripts of two PHYA/C-related genes were hardly affected by light.
Similar content being viewed by others
References
Aukerman, M.J., Hirschfeld, M., Wester, L., Weaver, M., Clack, T., Amasino, R.M. and Sharrock, R.A. 1997. A deletion in the PHYD gene of the arabidopsis Wassilewskija ecotype defines a role for phytochrome D in red/far-red light sensing. Plant Cell 9: 1317-1326.
Casal, J.J., Sanchez, R.A. and Botto J.F. 1998. Modes of action of phytochromes. J. Exp. Bot. 49: 127-138.
Chang, S., Puryear, J. and Cairney, J. 1993. A simple and efficient method for isolating RNA from pine trees. Plant Mol. Biol. Rep. 11: 113-116.
Childs, K.L., Miller, F.R., Cordonier-Pratt, M.M., Pratt, L.H., Morgan, P.W. and Mullet, J.E. 1997. The sorghum photoperiod sensitivity gene, Ma3, encodes a phytochrome B. Plant Physiol. 113: 611-619.
Clack, T., Mathews, S. and Sharrock, R.A. 1994. The phytochrome apoprotein family in Arabidopsis is encoded by five genes: the sequences and expression of PHYD and PHYE. Plant Mol. Biol. 25: 413-427.
Clapham, D.H., Dormling, I., Ekberg, I., Eriksson, G., Qamaruddin, M. and Vince-Prue, D. 1998a. Latitudinal cline of requirement for far-red light for the photoperiodic control of budset and extension growth in Picea abies (Norway spruce). Physiol. Plant. 102: 71-78.
Clapham, D., Ekberg, I., Dormling, I., Eriksson, G., Qamaruddin, M. and Vince-Prue, D. 1998b. Dormancy: night timekeeping and day timekeeping for the photoperiodic control of budset in Norway spruce. In: P.J. Lumsden and A.J. Millar (Eds.) Biological Rhythms and Photoperiodism in Plant, Bios Scientific Publishers, Oxford, UK.
Clapham, D.H., Newton, R.J., Sen, S. and von Arnold, S. In press. Transformation of Picea species. In: S.M. Jain and S.C. Minocha (eds.), Molecular Biology of Woody Plants. Kluwer Academic Publishers, Dordrecht, Netherlands.
D'Aoust, A.L. and Hubac, C. 1986. Phytochrome action and frost hardening in black spruce seedlings. Physiol. Plant. 67: 141-144.
Dehesh, K., Tepperman, J., Christensen, A.H. and Quail, P.H. 1991. phyB is evolutionarily conserved and constitutively expressed in rice seedling shoots. Mol. Gen. Genet. 225: 305-313.
Dormling, I. 1993. Bud dormancy, frost hardiness, and frost drought in seedlings of Pinus sylvestris and Picea abies. In: P.H. Li and L. Christersson (Eds.), Advances in Plant Cold Hardiness. CRC Press, Boca Raton, FL, pp. 285-298.
Elmlinger, M.W., Bolle, C., Batschauer, A., Oelmüller, R. and Mohr, H. 1994. Coaction of blue light and light absorbed by phytochrome in control of glutamine synthetase gene expression in Scots pine (Pinus sylvestris L.) seedlings. Planta 192: 189-194.
Fernbach, E. and Mohr, H. 1990. Coaction of blue/ultraviolet-A light and light absorbed by phytochrome in controlling growth of pine (Pinus sylvestris L.) seedlings. Planta 180: 212-216.
Frohman, M.A., Dush, M.K. and Martin, G.R. 1988. Rapid production of full-length cDNAs from rare transcripts: amplification using a single gene-specific oligonucleotide primer. Proc. Natl. Acad. Sci. USA 85: 8998-9002.
Furuya, M. 1993. Phytochromes: their molecular species, gene families, and functions. Annu. Rev. Plant Physiol. Plant Mol. Biol. 44: 617-645.
Halliday, K.J., Thomas, B. and Whitelam, G.C. 1997. Expression of heterologous phytochromes A, B or C in transgenic tobacco plants alters vegetative development and flowering time. Plant J. 12: 1079-1090.
Hauser, B.A., Pratt, L.H. and Cordonnier-Pratt, M.M. 1997. Absolute quantification of five phytochrome transcripts in seedlings and mature plants of tomato (Solanum lycopersicum L.). Planta 201: 379-387.
Howe, G.T., Bucciaglia, P.A., Hackett, W.P., Furnier, G.R., Cordonnier-Pratt, M.-M. and Gardner, G. 1998. Evidence that the phytochrome gene family in black cottonwood has one PHYA locus and two PHYB loci but lacks members of the PHYC/F and PHYE subfamilies. Mol. Biol. Evol. 15: 160-175.
Jackson, S. and Thomas, B. 1997. Photoreceptors and signals in the photoperiodic control of development. Plant Cell Environ. 20: 790-795.
Kolukisaoglu, H.Ñ., Marx, S., Wiegmann, C., Hanelt, S. and Schneider-Poetsch, H.A.W. 1995. Divergence of the phytochrome gene family predates angiosperm evolution and suggests that Selaginella and Equisetum arose prior to Psilotum. J. Mol. Evol. 41: 329-337.
Kvarnheden, A., Tandre, K. and Engström, P. 1995. A cdc2 homologue and closely related processed retropseudogenes from Norway spruce. Plant Mol. Biol. 27: 391-403.
Martin, W., Nock, S., Meyer-Gauen, G., Häger, K.-P., Jensen, U. and Cerff, R. 1993. A method for isolation of cDNAquality mRNA from immature seeds of a gymnosperm rich in polyphenolics. Plant Mol. Biol. 22: 555-556.
Mathews, S. and Sharrock, R.A. 1997. Phytochrome gene diversity. Plant Cell Environ. 20: 666-671.
Mathews, S., Lavin, M. and Sharrock, R.A. 1995. Evolution of the phytochrome gene family and its utility for phylogenetic analyses of angiosperms. Ann. Miss. Bot. Gard. 82: 296-321.
Mo, L.H., von Arnold, S. and Lagercrantz, U. 1989. Morphogenic and genetic stability in longterm embryogenic cultures and somatic embryos of Norway spruce (Picea abies L. Karst). Plant Cell Rep. 8: 375-378.
Morand, L.Z., Kidd, D.G. and Lagarias, J.C. 1993. Phytochrome levels in the green alga Mesotaenium caldariorum are light regulated. Plant Physiol 101: 97-103.
Quail, P.H. 1994. Phytochrome genes and their expression. In: R.E., Kendrick and G.H.M. Kronenberg (Eds.), Photomorphogenesis in Plants, 2nd ed. Kluwer Academic Publishers, Dordrecht, Netherlands, ISBN 0-7923-2551-6, pp. 71-104.
Quail, P.H., Boylan, M.T., Parks, B.M., Short, T.W., Xu, Y. and Wagner, D. 1995. Phytochromes: photosensory perception and signal transduction. Science 268: 675-680.
Qin, M., Kuhn, R., Moran, S. and Quail, P.H. 1997. Overexpressed phytochrome C has similar photosensory specificity to phytochrome B but a distinctive capacity to enhance primary leaf expansion. Plant J. 12: 1163-1172.
Rogers, S.O. and Bendich, A.J. 1988. Extraction of DNA from plant tissues. In: S.B. Gelvin, R.A. Schilperoort and D.P.S. Verma (eds.), Plant Molecular Biology Manual, Kluwer Academic Publishers, Dordrecht, Netherlands, pp. A6: 1-10.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
Schneider-Poetsch, H.A.W., Kolukisaoglu, Ñ., Clapham, D.H., Hughes, J. and Lamparter, T. 1998. Non-angiosperm phytochromes and the evolution of vascular plants. Physiol. Plant. 102: 612-622.
Sharrock, R.A. and Quail, P.H. 1989. Novel phytochrome sequences in Arabidopsis thaliana: structure, evolution, and differential expression of a plant regulatory photoreceptor family. Genes Dev. 3: 1745-1757.
Smith, H. 1995. Physiological and ecological function within the phytochrome family. Annu Rev. Plant Physiol. Plant Mol. Biol. 46: 289-315.
Smith, H. and Whitelam, G.C. 1997. The shade avoidance syndrome: multiple responses mediated by multiple phytochromes. Plant Cell Environ. 20: 840-844.
Smith, H., Xu, Y. and Quail, P.H. 1997. Antagonistic but complementary actions of phytochromes A and B allow optimum seedling de-etiolation. Plant Physiol. 114: 637-641.
Thümmler, F. and Dittrich, P. 1995. Phytochrome represents a small gene family in Picea abies: six partial sequences (Accession Nos. U38363 to U38368) encoding at least three different phytochrome isotypes. Plant Physiol. 109: 1498.
Thümmler, F., Dufner, M., Kreisl, P. and Dittrich, P. 1990. Molecular cloning of a novel phytochrome gene of the moss Ceratodon purpureus which contains a putative light-regulated protein kinase. Plant Mol. Biol. 20: 1003-1017.
Tomizawa, K.-I., Nagatani, A. and Furuya, M. 1990. Phytochrome genes: studies using the tools of molecular biology and photomorphogenetic mutants. Photochem. Photobiol. 52: 265-275.
Vince-Prue, D. 1984. Contrasting types of photoperiodic response in the control of dormancy. Plant Cell Environ. 7: 507-513.
Whitelam, G.C. and Devlin, P.F. 1997. Roles of different phytochromes in Arabidopsis photomorphogenesis. Plant Cell Environ. 20: 752-758.
Winands, A., Wagner, G., Marx, S. and Schneider-Poetsch, H.A.W. 1992. Partial nucleotide sequence of phytochrome from the Zygnematophycean green alga Mougeotia. Photochem. Photobiol. 56: 765-770.
Young, E. and Hanover, J.W. 1977. Effects of quality, intensity and duration of light breaks during a long night on dormancy in blue spruce (Picea pungens Engelm.) seedlings. Plant Physiol. 60: 271-273.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Clapham, D.H., Kolukisaoglu, H.Ü., Larsson, CT. et al. Phytochrome types in Picea and Pinus. Expression patterns of PHYA-related types. Plant Mol Biol 40, 669–678 (1999). https://doi.org/10.1023/A:1006204318499
Issue Date:
DOI: https://doi.org/10.1023/A:1006204318499