Abstract
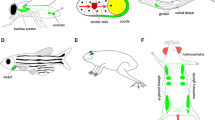
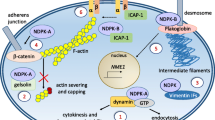
The role of nucleoside diphosphate (NDP) kinases in cell growth, differentiation, and tumormetastasis in relation to signal transduction was investigated. The essential role of NDP kinasein cell growth was validated by coupling between reduced NDP kinase levels, induced byantisense oligonucleotides, and the suppression of proliferative activity of a cultured cell line.In addition, because NDP kinase levels are often enhanced with development and differentiation,as has been demonstrated in postmitotic cells and tissues, such as the heart and brain, wefurther examined this possibility using the bone tissue (osteoblasts) and a cultured cell linePC12D. The enhanced NDP kinase accumulation was demonstrated in the matured osteoblastsin vivo and in vitro by immunohistochemistry. In PC12D cells neurite outgrowth took placein NDP kinase β-transfected clones without differentiation inducers, which was accompaniedby prolongation of doubling time. Neurite outgrowth, triggered by nerve growth factor and acyclic AMP analog, was down-regulated upon forced expression of inactive mutant NDPkinase by virtue of a dominant negative effect. NDP kinase α-transfected rat mammaryadenocarcinoma cells (MTLn3) and nm23-H2-transfected human oral squamous cell carcinomacells (LMF4) manifested reduced metastatic potential and were associated with an alteredsensitivity to environmental factors, such as motility and growth factors. NDP kinase α,compared to NDP kinase β, was involved in a wide variety of the cellular phenomena examined.Taken together, NDP kinase isoforms appear to elicit both their own respective and commoneffects. They may have an ability to lead cells to both proliferative and differentiated statesby modulating responsiveness to environmental factors, but their fate seems to depend on theirsurrounding milieu.
Similar content being viewed by others
REFERENCES
Ali, S. Y., Sajdera, S. W., and Anderson, H. C. (1970). Proc. Natl. Acad. Sci. USA 67, 1513-1520.
Baba, H., Urano, T., Okada, K., Furukawa, K., Nakayama, E., Tanaka, H., Iwasaki, K., and Shiku, H. (1995). Cancer Res. 55, 1977-1981.
Caligo, M. A., Cipollini, G., Fiore, L., Calvo, S., Basolo, F., Collecchi, P., Ciardiello, F., Pepe, S., Petrini, M., and Bevilacqua, G. (1995). Intern. J. Cancer 60, 837-842.
Cipollini, G., Berti, A., Fiore, L., Rainaldi, G., Basolo, F., Merlo, G., Bevilacqua, G., and Caligo, M. A. (1997). Intern. J. Cancer 73, 297-302.
Creanor, J., and Mitchison, J. M. (1989). J. Cell Sci. 93, 185-189.
Fukuda, M., Ishii, A., Yasutomo, Y., Shimada, N., Ishikawa, N., Hanai, N., Nagata, N., Irimura, T., Nicolson, G. L., and Kimura, N. (1996). Intern. J. Cancer 65, 531-537.
Howlett, A. R., Petersen, O. W., Steeg, P. S., and Bissell, M. J. (1994). J. Natl. Cancer Inst. 86, 1838-1844.
Igawa, M., Rukstalis, D. B., Tanabe, T., and Chodak, G.W. (1994). Cancer Res. 54, 1313-1318.
Ishijima, Y., Shimada, N., Fukuda, M., Miyazaki, H., Orlov, N. Y., Orlova, T. G., Yamada, T., and Kimura, N. (1999). FEBS Lett. 445, 155-159.
Ishikawa, N., Taniguchi-Seto, H., Munakata, Y., Takagi, Y., Shitomada, N., and Kimura, N. (1997). J. Biol. Chem. 272, 3289-3295.
Kantor, J. D., McCormick, B., Steeg, P. S. and Zetter B. R. (1993). Cancer Res. 55, 1971-1973.
Keim, D., Melhem, R., Zhu, X. X., Lascu, I., Véron, M., and Strahler, J. R. (1992). J. Clin. Invest. 89, 919-924.
Kimura, N. (1993). In Handbook of Experimental Pharmacology: GTPases in Biology II (B. F. Dickey, and L. Birnbaumer, eds.), Vol. 108II, Springer-Verlag, Berlin Heidelberg, pp. 485-498.
Kimura, N., Shimada, N., Nomura, K., and Watanabe, K. (1990). J. Biol. Chem. 265, 15744-15749.
Lakso, M., Steeg, P. S., and Westphal, H. (1992). Cell Growth Different. 3, 873-879.
Leone, A., Flatow, U., King, C. R., Sandeen, M. A., Margulies, I. M. K., Liotta, L. A., and Steeg, P. S. (1991). Cell 65, 25-35.
Leone, A., Flatow, U., VanHoutte, K., and Steeg, P. S. (1993). Oncogene 8, 2325-2333.
Liotta, L. A., Steeg, P. S., and Stetler-Stevenson, W. G. (1991). Cell 64, 327-336.
Miyazaki, H., Fukuda, M., Ishijima, Y., Takagi, Y., Iimura, T., Negishi, A., Hirayama, R., Ishikawa, N., Amagasa, T., and Kimura, N. (1999). Clin. Cancer Res. 5, 4301-4307.
Moréra, S., Lacombe, M-L., Yingwu, X., LeBras, G., Janin, J. (1995). Structure 3, 1307-1314.
Ohneda, K., Fukuda, M., Shimada, N., Ishikawa, N., Icho, T., Kaji, K., Toyota, T., and Kimura, N. (1994). FEBS Lett. 384, 273-277.
Orlov, N. Y., Orlova, T. G., Nomura, K., Hanai, N., and Kimura, N. (1996). FEBS Lett. 389, 186-190.
Parks, R. E., Jr., and Agarwal, R. P. (1973). In The enzymes, Vol. 8, P. D. Boyer, ed.), Academic Press, New York, pp. 307-333.
Postel, E. H., Berberich, S. J., Flint, S. J., and Ferrone, C. A. (1993). Science 261, 478-480.
Shimada, N., Ishikawa, N., Munakata, Y., Toda, T., Watanabe, K., and Kimura, N. (1993). J. Biol. Chem. 268, 2583-2589.
Sorscher, S. M., Steeg, P., Feramisco, J. R., Buckmaster, C., Boss, G. R. and Meinkoth, J. (1993). Biochem. Biophys. Res. Commun. 195, 336-345.
Steeg, P. S., Bevilacqua, G., Kopper, L., Thogeirsson, U. P., Talmadge, J. E., Liotta, L. A., and Sobel, M. E. (1988). J. Natl. Cancer Inst. 80, 200-204.
Stein, G. S., and Lian, J. B. (1993). Endocrine Rev. 14, 424-442.
Yasutomo, Y., Ishikawa, N., Nagata, N., and Kimura, N. (1998). J. Bone Mineral Metab. 16, 215-226.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kimura, N., Shimada, N., Fukuda, M. et al. Regulation of Cellular Functions by Nucleoside Diphosphate Kinases in Mammals. J Bioenerg Biomembr 32, 309–315 (2000). https://doi.org/10.1023/A:1005549315846
Issue Date:
DOI: https://doi.org/10.1023/A:1005549315846