Abstract
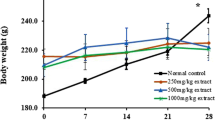
(−)-Hydroxycitric acid (HCA), a natural extract from the dried fruit rind of Garcinia cambogia (family Guttiferae), is a popular supplement for weight management. The dried fruit rind has been used for centuries as a condiment in Southeastern Asia to make food more filling and satisfying. A significant number of studies highlight the efficacy of Super CitriMax (HCA-SX, a novel 60% calcium-potassium salt of HCA derived from Garcinia cambogia) in weight management. These studies also demonstrate that HCA-SX promotes fat oxidation, inhibits ATP-citrate lyase (a building block for fat synthesis), and lowers the level of leptin in obese subjects. Acute oral, acute dermal, primary dermal irritation and primary eye irritation toxicity studies have demonstrated the safety of HCA-SX. However, no long-term safety of HCA-SX or any other (−)-hydroxycitric acid extract has been previously assessed. In this study, we have evaluated the dose- and time-dependent effects of HCA-SX in Sprague-Dawley rats on body weight, hepatic and testicular lipid peroxidation, DNA fragmentation, liver and testis weight, expressed as such and as a % of body weight and brain weight, and histopathological changes over a period of 90 days. The animals were treated with 0, 0.2, 2.0 and 5.0% HCA-SX as feed intake and the animals were sacrificed on 30, 60 or 90 days of treatment. The feed and water intake were assessed and correlated with the reduction in body weight. HCA-SX supplementation demonstrated a reduction in body weight in both male and female rats over a period of 90 days as compared to the corresponding control animals. An advancing age-induced marginal increase in hepatic lipid peroxidation was observed in both male and female rats as compared to the corresponding control animals. However, no such difference in hepatic DNA fragmentation and testicular lipid peroxidation and DNA fragmentation was observed. Furthermore, liver and testis weight, expressed as such and as a percentage of body weight and brain weight, at 30, 60 and 90 days of treatment, exhibited no significant difference between the four groups. Taken together, these results indicate that treatment of HCA-SX over a period of 90 days results in a reduction in body weight, but did not cause any changes in hepatic and testicular lipid peroxidation, DNA fragmentation, or histopathological changes.
Similar content being viewed by others
References
Jequier E: Pathways to obesity. Int J Obes Relat Metab Disord 260: S12-S17, 2002
Mayer-Davis EJ, Costacou T: Obesity and sedentary lifestyle: Modifiable risk factors for prevention of type 2 diabetes. Curr Diab Rep 1: 170-176, 2001
Haller CA, Benowitz NL: Adverse cardiovascular and central nervous system events associated with dietary supplements containing ephedra alkaloids. N Engl J Med 343: 1833-1838, 2000
Ramos RR, Saenz JLS, Agular RJA: Extract of Garcinia Cambogia in controlling obesity. Invest Med Int 22: 97-100, 1995
Sergio W: A natural food, the Malabar Tamarind, may be effective in the treatment of obesity. Med Hypotheses 29: 39-40, 1988
Lowenstein JM: Effect of (−)-hydroxycitrate on fatty acid synthesis by rat liver in vivo. J Biol Chem 246: 629-632, 1971
Triscari J, Sullivan AC: Anti-obesity activity of a novel lipid synthesis inhibitor. Int J Obes 8: 227-239, 1984
Loe YC, Bergeron N, Rodriguez N, Schwarz J-M: Gas chromatography/mass spectrometry method to quantify blood hydroxycitrate concentration. Anal Biochem 292: 148-154, 2001
Ohia SE, Awe O, LeDay AM, Opere CA, Bagchi D: Effect of hydroxycitric acid on serotonin release from isolated rat brain cortex. Res Commun Mol Pathol Pharmacol 109: 210-216, 2001
Conte AA: A non-prescription alternative in weight reduction therapy. Am J Bariat Med (Summer): 17-19, 1993
Mattes DR, Bormann L: Effects of (−)-hydroxycitric acid on appetitive variables. Physiol Behav 71: 87-94, 2000
Westerterp-Plantenga MS, Kovacs EMR: The effect of (−)-hydroxycitrate on energy intake and satiety in overweight humans. Int J Obes 26: 870-872, 2002
Kovacs EM, Westerterp-Plantenga MS, de Vries M, Brouns F, Saris WH: Effects of 2-week ingestion of (−)-hydroxycitrate and (−)-hydroxycitrate combined with medium-chain triglycerides on satiety and food intake. Physiol Behav 74: 543-549, 2001
Ohia SE, Opere CA, LeDay AM, Bagchi M, Bagchi D, Stohs SJ: Safety and mechanism of appetite suppression by a novel hydroxycitric acid extract (HCA-SX). Mol Cell Biochem 238: 89-103, 2002
Preuss HG, Bagchi D, Rao CVS, Echard BW, Satyanarayana S, Bagchi M: Effect of (−)-hydroxycitric acid on weight loss, body mass index and plasma leptin levels in human subjects. FASEB 16: (abstr) 742.16, 2002
Buege J, Aust S: Microsomal lipid peroxidation. Meth Enzymol 52: 302-310, 1978
Bagchi D, Carryl OR, Tran MX, Krohn RL, Bagchi DJ, Garg A, Bagchi M, Mitra S, Stohs SJ: Stress, diet and alcohol-induced oxidative gastrointestinal mucosal injury in rats and protection by bismuth subsalicylate. J Appl Toxicol 18: 3-13, 1998
Largilliere C, Melancon SB: Free malondialdehyde determination in human plasma by high-performance liquid chromatography. Anal Biochem 170: 123-126, 1988
Bagchi M, Balmoori J, Bagchi D, Ray SD, Kuszynski C, Stohs SJ: Smokeless tobacco, oxidative stress, apoptosis, and antioxidants in human oral keratinocytes. Free Rad Biol Med 26: 992-1000, 1999
Bagchi D, Ray SD, Patel D, Bagchi M: Protection against drug-and chemical-induced multiorgan toxicity by a novel IH636 grape seed proanthocyanidin extract. Drugs Exp Clin Res 27: 3-15, 2001
Sullivan AC, Triscari J, Hamilton JG, Miller ON: Effect of (−)-hydroxycitrate upon the accumulation of lipid in the rat. II. Appetite. Lipids 9: 129-134, 1974
Preuss HG, Bagchi D, Bagchi M., Rao CVS, Dey DK, Das S, Satyanarayana S: Weight management and mechanism of action of a novel, natural extract of (−)-hydroxycitric acid (HCA-SX) and a combination of HCA-SX plus niacin-bound chromium and gymnema sylvestre extract. JAMA (submitted)
Al-Bayati ZAF, Wahba ZZ, Stohs SJ: 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD)-induced alterations in lipid peroxidation, enzymes, and divalent cations in rat testis. Xenobiotica 18: 1281-1289, 1988
Wahba ZZ, Murray WJ, Hassan MQ, Stohs SJ: Comparative effects of pair-feeding and 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) on various biochemical parameters in female rats. Toxicology 59: 311-323, 1989
Bagchi D, Shara MA, Bagchi M, Hassoun EA, Stohs SJ: Time-dependent effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin on serum and urine levels of malondialdehyde, formaldehyde, acetaldehyde, and acetone in rats. Toxicol Appl Pharmacol 123: 83-88, 1993
Latchoumycandane C, Chitra KC, Mathur PP: The effect of 2,3,7,8-tetrachlorodibenzo-p-dioxin on the antioxidant system in mitochondrial and microsomal fractions of rat testis. Toxicology 171: 127-135, 2002
Park JD, Habeebu SSM, Klaassen CD: Testicular toxicity of di-(2-ethylhexyl)phthalate in young Sprague-Dawley rats. Toxicology 171: 105-115, 2002
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shara, M., Ohia, S.E., Yasmin, T. et al. Dose- and time-dependent effects of a novel (−)-hydroxycitric acid extract on body weight, hepatic and testicular lipid peroxidation, DNA fragmentation and histopathological data over a period of 90 days. Mol Cell Biochem 254, 339–346 (2003). https://doi.org/10.1023/A:1027358106407
Issue Date:
DOI: https://doi.org/10.1023/A:1027358106407