Abstract
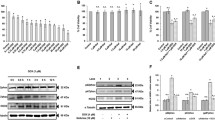
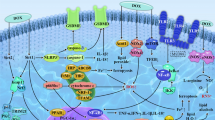
In this review, we discuss the role of nitric oxide synthase in doxorubicin (DOX)-induced cardiomyopathy, a prominent side effect of DOX chemotherapy in cancer patients. It is becoming increasingly clear that apoptosis of myocardial cells plays a critical role in the onset of cardiomyopathy. DOX exposure to endothelial cells and cardiomyocytes caused apoptotic cell death at sub-micromolar concentrations. DOX-induced generation of H2O2 has been shown to be responsible for this drug's toxicity and apoptosis. H2O2 in turn enhanced endothelial nitric oxide synthase (eNOS) transcription in endothelial cells and myocytes. Antisense eNOS depressed DOX-induced oxidative stress and apoptosis. Redox-metal chelators inhibited DOX-induced apoptosis, clearly suggesting a role for reactive oxygen species in DOX-induced apoptosis. Here, we will focus on the role of eNOS expression, iron chelation, and iron signaling on DOX-mediated apoptosis.
Similar content being viewed by others
References
Bristow MR, Billingham ME, Mason JW, Daniels JR: Clinical spectrum of anthracycline cardiotoxicity. Cancer Treat Rep 62: 873–879, 1978
Minow RA, Benjamin RS, Gottlieb JA: Adriamycin (NSC 123127) cardiomyopathy - an overview with determination of risk factors. Cancer Chemother Rep 6: 198–201, 1975
Singal PK, Iliskovic N: Doxorubicin-induced cardiomyopathy. N Eng J Med 339: 900–905, 1998
Singal PK, Deally CMR, Weinberg LE: Subcellular effects of adriamycin in the heart: A concise review. J Mol Cell Cardiol 19: 817–828, 1987
Sawyer DB, Fukazawa R, Arstall MA, Kelly RA: Daunorubicin-induced apoptosis in rat cardiac myocytes is inhibited by dexrazoxane. Circ Res 84: 257–265, 1999
Kotamraju S, Konorov EA, Joseph J, Kalyanaraman B: Doxorubicininduced apoptosis in endothelial cells and cardiomyocytes is ameliorated by nitrone spin traps and ebselen. J Biol Chem 275: 33585–33592, 2000
Wang L, Ma W, Markovich R, Chen J-W, Wang PH: Regulation of cardiomyocyte apoptotic signaling by insulin-like growth factor 1. Circ Res 83: 516–522, 1998
Narula J, Haider N, Virmani R, DiSalvo TG, Kolodgie FD, Hajjar RJ, Schmidt U, Semigran MJ, Dec GW, Khaw BA: Apoptosis in myocytes in end-stage heart failure. New Eng J Med 335: 1182–1189, 1996
Kalivendi S, Kotamraju S, Zhao H, Joseph J, Kalyanaraman B: Doxorubicin-induced apoptosis is associated with increased transcription of endothelial nitric-oxide synthase: Effect of antiapoptotic antioxidants and calcium. J Biol Chem 276: 47266–47276, 2001
Gouazè V, Mirault ME, Carpentier S, Salvayre R, Levade T, Andrieu-Abadie N: Glutathione peroxidase-1 overexpression prevents ceramide production and partially inhibits apoptosis in doxorubicin-treated human breast carcinoma cells. Mol Pharmacol 60: 488–496, 2001
Campbell KA, Lashley MR, Wyatt JK, Nantz MH, Britt RD: Dualmode EPR study of Mn(III) salen and the Mn(III) salen-catalyzed epoxidation of cis-beta-methylstyrene. J Am Chem Soc 123: 5710–5719, 2001
Campbell KA, Yikilmaz E, Grant CV, Gregor W, Miller A-F, Britt RD: Parallel polarization EPR characterization of the Mn(III) center of oxidized manganese superoxide dismutase. J Am Chem Soc 121: 4714–4715, 1999
Minotti G, Cairo G, Monti E: Role of iron in anthracycline cardiotoxicity: New tunes for an old song? FASEB J 13: 199–212, 1999
Kalyanaraman B, Perez-Reyes E, Mason RP: Spin-trapping and direct electron spin resonance investigations of the redox metabolism of quinone anticancer drugs. Biochim Biophys Acta 630: 119–130, 1980
Goodman J, Hochstein P: Generation of free radicals and lipid peroxidation by redox cycling of adriamycin and daunomycin. Biochem Biophys Res Commun 77: 797–803, 1977
Myers C: The role of iron in doxorubicin-induced cardiomyopathy. Semin Oncol 25(suppl 10): 10–14, 1998
Sarvazyan N: Visualization of doxorubicin-induced oxidative stress in isolated cardiac myocytes. Am J Physiol 271 (Heart Circ Physiol 40): H2079–H2085, 1996
Konorev EA, Kennedy MC, Kalyanaraman B: Cell-permeable superoxide dismutase and glutathione peroxidase mimetics afford superior protection against doxorubicin-induced cardiotoxicity: The role of reactive oxygen and nitrogen intermediates. Arch Biochem Biophys 368: 421–428, 1999
Davies KJA, Doroshow JH: Redox cycling of anthracyclines by cardiac mitochondria. I. Anthracycline radical formation by NADH dehydrogenase. J Biol Chem 261: 3060–3067, 1986
Singal PK, Panagia V: Direct effects of adriamycin on the rat heart sarcolemma. Res Commun Chem Path Pharmacol 43: 67–77, 1984
Solem LE, Heller LJ, Wallace KB: Dose-dependent increase in sensitivity to calcium-induced mitochondrial dysfunction and cardiomyocyte cell injury by doxorubicin. J Mol Cell Cardiol 28: 1023–1032, 1996
Simmons TW, Jamall IS: Relative importance of intracellular glutathione peroxidase and catalase in vivo for prevention of peroxidation to the heart. Cardiovasc Res 23: 774–779, 1989
Chance B, Sies H, Boveris A: Hydroperoxide metabolism in mammalian organs. Physiol Rev 59: 527–605, 1979
Oster O, Prellwitz W: Selenium and cardiovascular disease. Biol Trace Elem Res 24: 91–103, 1990
Vásquez-Vivar J, Martasek P, Hogg N, Masters BSS, Pritchard KA Jr, Kalyanaraman B: Endothelial nitric oxide synthase-dependent superoxide generation from adriamycin. Biochemistry 36: 11293–11297, 1997
Garner AP, Paine MJ, Rodriguez-Crespo I, Chinje EC, Ortiz De Montellano P, Stratford IJ, Tew DG, Wolf CR: Nitric oxide synthases catalyze the activation of redox cycling and bioreductive anticancer agents. Cancer Res 59: 1929–1934, 1999
Thomas CE, Aust SD: Release of iron from ferritin by cardiotoxic anthracycline antibiotics. Arch Biochem Biophys 248: 684–689, 1986
Minotti G, Recalcati S, Mordente A, Liberi G, Calafiore AM, Mancuso C, Preziosi P, Cairo G: The secondary alcohol metabolite of doxorubicin irreversibly inactivates aconitase/iron regulatory protein-1 in cytosolic fractions from human myocardium. FASEB J 12: 541–552, 1998
Myers CE, Gianni L, Simone CB, Klecker R, Greene R: Oxidative destruction of erythrocyte ghost membranes catalyzed by the doxorubiciniron complex. Biochemistry 21: 1707–1712, 1982
Muindi JR, Sinha BK, Gianni L, Myers CE: Hydroxyl radical production and DNA damage induced by anthracycline-iron complex. FEBS Lett 172: 226–230, 1984
Minotti G, Cairo G, Monti E: Role of iron in anthracycline cardiotoxicity: New tunes for an old song? FASEB J 13: 199–212, 1999
Herman EH, Ferrans VJ, Myers CE, Van Vleet JF: Comparison of the effectiveness of (± )-1,2-bis(3,5-dioxopiperazinyl-1-yl)propane (ICRF-187) and N-acetylcysteine in preventing chronic doxorubicin cardiotoxicity in beagles. Cancer Res 45: 276–281, 1985
Speyer JL, Green MD, Kramer E, Rey M, Sanger J, Ward C, Dubin N, Ferrans V, Stecy P, Zeleniuch-Jacquotte A: Protective effect of the bispiperazinedione ICRF-187 against doxorubicin-induced cardiac toxicity in women with advanced breast cancer. N Eng J Med 319: 745–752, 1988
Curran CF, Narang PK, Reynolds RD: Toxicity profile of dexrazoxane (Zinecard, ICRF-187, ADR-529, NSC-169780), a modulator of doxorubicin cardiotoxicity. Cancer Treat Rev 18: 241–252, 1991
Seifert CF, Nesser ME, Thompson DE: Dexrazoxane in the prevention of doxorubicin-induced cardiotoxicity. Ann Pharmacother 28: 1063–1072, 1994
Tetef ML, Synold TW, Chow W, Leong L, Margolin K, Morgan R, Raschko J, Shibata S, Somlo G, Yen Y, Groshen S, Johnson K, Lenz HJ, Gandara D, Doroshow JH: Phase I trial of 96-h continuous infusion of dexrazoxane in patients with advanced malignancies. Clin Cancer Res 7: 1569–1576, 2001
Klausner RD, Rouault TA, Harford JB: Regulating the fate of mRNA: The control of cellular iron metabolism. Cell 72: 19–28, 1993
Martins EAL, Robalinho RL, Meneghini R: Oxidative stress induces activation of a cytosolic protein responsible for control of iron uptake. Arch Biochem Biophys 316:128–134, 1995
Pantopoulos K, Hentze MW: Rapid response to oxidative stress mediated by iron regulatory protein. EMBO J 14: 2917–2924, 1995
Pantopoulos K, Mueller S, Atzberger A, Ansorge W, Stremmel W, Hentze MW: Differences in the regulation of iron regulatory protein-1 (IRP-1) by extra-and intracellular oxidative stress. J Biol Chem 272: 9802–9808, 1997
Gehring NH, Hentze MW, Pantopoulos K: Inactivation of both RNA binding and aconitase activities of iron regulatory protein-1 by quinoneinduced oxidative stress. J Biol Chem 274: 6219–6225, 1999
Rouault TA, Klausner RD: Iron-sulfur clusters as biosensors of oxidants and iron. Trends Biochem Sci 21: 174–177, 1996
Klausner RD, Rouault TA, Harford JB: Regulating the fate of mRNA: The control of cellular iron metabolism. Cell 72: 19–28, 1993
Cairo G, Castrusini E, Minotti G, Bernelli-Zazzera A: Superoxide and hydrogen peroxide-dependent inhibition of iron regulatory protein activity: A protective stratagem against oxidative injury. FASEB J 10: 1326–1335, 1996
Vile GF, Basu-Modak S, Waltner C, Tyrrell RM: Heme oxygenase 1 mediates an adaptive response to oxidative stress in human skin fibroblasts. Proc Natl Acad Sci USA 91: 2607–2610, 1994
Kang PM, Izumo S: Apoptosis and heart failure: A critical review of the literature. Circ Res 86: 1107–1120, 2000
Haunstetter A, Izumo S: (1998) Apoptosis - basic mechanisms and implications for cardiovascular disease. Circ Res 82: 1111–1129, 1998
Narula J, Pandey P, Arbustini E, Haider N, Narula N, Kolodgie FD, Dal Bello B, Semigran MJ, Bielsa-Masdeu A, Dec GW, Israels S, Ballester M, Virmani R, Saxena S, Kharbanda S: Apoptosis in heart failure: Release of cytochrome c from mitochondria and activation of caspase-3 in human cardiomyopathy. Proc Natl Acad Sci USA 96: 8144–8149, 1999
Konopa J: G2 block induced by DNA crosslinking agents and its possible consequences. Biochem Pharmacol 37: 2303–2309, 1988
Carmody JR, Cotter TG: Signalling apoptosis: A radical approach. Redox Rep 6: 77–88, 2001
Green D, Kroemer G: The central executioners of apoptosis: Caspases or mitochondria? Trends Cell Biol 8: 267–71, 1998
Green DR, Reed JC: Mitochondria and apoptosis. Science 281: 1309–1312, 1998
Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD: The release of cytochrome c from mitochondria: A primary site for Bcl-2 regulation of apoptosis. Science 275: 1132–1136, 1997
Fulda S, Meyer E, Friesen C, Susin SA, Kroemer G, Debatin KM: Cell type specific involvement of death receptor and mitochondrial pathways in drug-induced apoptosis. Oncogene 20: 1063–1075, 2001
Gross A, McDonnell JM, Korsmeyer SJ: Bcl-2 family members and the mitochondria in apoptosis. Genes Dev 13: 1899–1911, 1999
Oltvai ZN, Milliman CL, Korsmeyer SJ: Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 74: 609–619, 1993
von Harsdorf R, Li PF, Dietz R: Signaling pathways in reactive oxygen species-induced cardiomyocyte apoptosis. Circulation 99: 2934–2941, 1999
Wang S, Leonard SS, Ye J, Ding M, Shi X: The role of hydroxyl radical as a messenger in Cr(VI)-induced p53 activation. Am J Physiol 279: C868–C875, 2000
Huang C, Zhang Z, Ding M, Li J, Ye J, Leonard SS, Shen HM, Butterworth L, Lu Y, Costa M, Rojanasakul Y, Castranova V, Vallyathan V, Shi X: Vanadate induces p53 transactivation through hydrogen peroxide and causes apoptosis. J Biol Chem 275: 32516–32522, 2000
Gouaze V, Mirault M-E, Carpenter S, Salvayre R, Levade T, Andrieu-Abadie N: Glutathione peroxidase-1 overexpression prevents ceramide production and partially inhibits apoptosis in doxorubicin-treated human breast carcinoma cells. Mol Pharmacol 60: 488–496, 2001
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalyanaraman, B., Joseph, J., Kalivendi, S. et al. Doxorubicin-induced apoptosis: Implications in cardiotoxicity. Mol Cell Biochem 234, 119–124 (2002). https://doi.org/10.1023/A:1015976430790
Issue Date:
DOI: https://doi.org/10.1023/A:1015976430790