Abstract
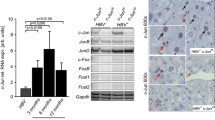
Immediate–early protooncogenes (IEP) are thought to play an important role in hepatocyte replication. Whether the extent of their expression correlates with the strength of the proliferative stimulus and subsequent regenerative activity has yet to be documented in vivo. Data are also lacking with respect to the level at which liver disease is associated with biochemical evidence of hepatic dysfunction. Thus, the objectives of this study were to determine whether a correlation exists between IEP gene mRNA expression and varying extents of partial hepatectomy (PHx) and to document the extent of resection required to result in increases in serum bilirubin levels. Eighty-nine adult, male Sprague-Dawley rats underwent either sham surgery or 20%, 35%, 55%, 70% or 90% PHx. Postoperatively, rats were killed (N = 3–6/group) at 15 and 30 mins and 8 and 24 hrs for c-fos, c-jun, and c-myc mRNA expression by northern blot analyses. Rats killed at 24 hrs also had hepatic regenerative activity documented by [3H]thymidine incorporation into hepatic DNA and serum bilirubin determinations. While c-fos mRNA expression at 15 mins and c-myc mRNA expression at 8 hrs after PHx did not correlate with the extent of PHx (r2 = 0.478 and 0.018, respectively), a weak correlation existed between c-jun mRNA expression at 30 mins and the extent of PHx (r2 = 0.662, P < 0.05). In terms of IEP mRNA expression and hepatic regenerative activity, a strong correlation existed between c-fos mRNA expression and [3H]thymidine incorporation (r2 = 0.851, P < 0.01) but not c-jun or c-myc mRNA expression. Compared to sham operated controls, [3H]thymidine incorporation was 2.0×, 3.4×, 3.2×, 7.8×, and 2.2× increased following 20%, 35%, 55%, 70%, and 90% PHx, respectively. Serum bilirubin levels remained unchanged until 70% PHx, when they increased from baseline values of 0.54 ± 0.05 mg/dl to 1.02 ± 0.15 mg/dl (P < 0.05). A further increase occurred following 90% PHx (1.83 ± 0.30 mg/dl, P < 0.01). In conclusion these findings indicate that c-fos mRNA expression 15 mins after PHx correlates with hepatic regenerative activity but not the strength of the regenerative stimulus and that hepatic parenchymal loss of 55–70% must occur prior to the detection of elevated serum bilirubin levels. The results also indicate that relative to a 70% PHx, 90% PHx is associated with decreased rather than increased hepatic regenerative activity.
Similar content being viewed by others
REFERENCES
Michalopoulos GK, DeFrances MC: Liver regeneration. Science 276:60–66, 1997.
Pistoi S, Morello D: Liver regeneration 7. Prometheus' myth revisited: Transgenic mice as a powerful tool to study liver regeneration. FASEB J 10:819–828, 1996.
Dubois RN: Early changes in gene expression during liver regeneration: What do they mean? Hepatology 11:1079–1082, 1990.
Thompson NL, Mead JE, Braun L, Goyette M, Shank PR, Fausto N: Sequential protoocogene expression during rat liver regeneration. Cancer Res 46:3111–3117, 1986.
Fausto N, Webber EM: Liver regeneration. In The Liver: Biology and Pathobiology. 3rd ed. IM, Arias, JL Boyer, N Fausto, W Jakoby, B Schacter, DA Shafritz, (eds). 1994 New York, Raven Press pp 1059–1084
Haber BA, Mohn KL, Diamond RH, Taub R: Induction patterns of 70 genes during nine days after hepatectomy define the temporal course of liver regeneration. J Clin Invest 91:1319–1326 1993
Steer CJ: Liver regeneration. FASEB J 9:1396–1400, 1995
Higgins GM, Anderson RM: Experimental pathology of liver: Restoration of liver of the white rat following partial surgical removal. Arch Pathol 12:186–202, 1931
Gaub J, Iversen J: Rat liver regeneration after 90% partial hepatectomy. Hepatology 4:902–904, 1984
Baruch Y, Frith LO, Kirsch RE: Twenty-one percent partial hepatectomy. In vivo rat model for the study of liver regeneration. J Hepatol 12:316–320, 1991
Braun L, Mead JE, Panzica M, Mikumo R, Bell GI, Fausto N: Transforming growth factor b mRNA increases during liver regeneration: A possible paracrine mechanism of growth regulation. Proc Natl Acad Sci USA 85:1539–1543, 1988
Mead JE, Braun L, Martin DA, Fausto N: Induction of replicative competence (“priming”) in normal liver. Cancer Res 50:7023–7030, 1990
Luk GD: Essential role of polyamine metabolism in hepatic regeneration: inhibition of deoxyribonucleic acid and protein synthesis and tissue regeneration by difluoromethylomithine in the rat. Gastroenterology 90:1261–1267, 1986
Fausto N, Laird AD, Webber EM: Role of growth factors and cytokines in hepatic regeneration. FASEB J 9:1527–1536, 1995
Skullman S, Adrian TE, Permert J, Larsson J: Humoral growth factors in plasma and liver tissue during liver regeneration and malnutrition. Eur J Gastroenterol Hepatol 6:797–802, 1994
Assy N, Minuk GY: Liver regeneration: Methods for monitoring and their applications. J Hepatol 26:945–952, 1997
Zhang XK, Gauthier T, Burczynski FJ, Wang GQ, Gong YW, Minuk GY: Changes in liver membrane potentials after partial hepatectomy in rats. Hepatology 23:549–551, 1996
Alison MR: Regulation of hepatic growth. Am Physiol Soc 66:499–541, 1986
Kruijer W, Skelly H, Botteri F, van der Putten H, Barber JR, Verma IM, Leffert HI: Protooncogene expression in regenerating liver is simulated in cultures of primary adult rat hepatocytes. J Biol Chem 261:7929–7933, 1986
Minuk GY, Gauthier T, Benarroch A: Changes in serum and hepatic polyamine concentrations after 30%, 70% and 90% partial hepatectomy in rats. Hepatology 12:542–546, 1990
Tuczek HV, Rabes H: Verlust der Proliferationsfahigkeit der hepatozyten nach subtotalen hepatektomie. Experientia 27:526, 1971
Zieve L, Anderson RW, Lindblad S: Course of hepatic regeneration after 80% to 90% resection of normal rat liver. Comparison with two-lobe and one-lobe hepatectomy. J Lab Clin Med 105:331–336, 1985
Vic P, Saint-Aubert B, Astre C, Bories P, Bonardet A, Descomps B, Humeau C, Joyeux H: Complete liver regeneration in one-stage 90% hepatectomized rats treated with testosterone. Hepatology 2:247–248, 1982
Ellis WR, Chu PK, Murray-Lyon IM: The influence of ammonia and octanoic acid on liver regeneration in the rat. Clin Sci 56:95–97, 1979
Zieve L, Shekleton M, Lyftogt C, Draves K: Ammonia, octanoate and a mercaptan depress regeneration of normal rat liver after partial hepatectomy. Hepatology 5:28–31, 1985
Minuk GY, Gauther T: The effect of g-aminobutyric acid on hepatic regenerative activity following partial hepatectomy in rats. Gastroenterology 104:217–221, 1993
Nadori F, Lardeux B, Rahmani M, Bringuier A, Durand-Schneider AM, Bernuau D: Presence of distinct AP-1 dimers in normal and transformed rat hepatocytes under basal conditions and after epidermal growth factor stimulation. Hepatology 26:1477–1483, 1997
McIntrye N: The limitations of conventional liver function tests. Semin Liver Dis 3:265–274, 1983
Rights and permissions
About this article
Cite this article
Moser, M., Gong, Y., Zhang, M. et al. Immediate-Early Protooncogene Expression and Liver Function Following Various Extents of Partial Hepatectomy in the Rat. Dig Dis Sci 46, 907–914 (2001). https://doi.org/10.1023/A:1010791915733
Issue Date:
DOI: https://doi.org/10.1023/A:1010791915733