Abstract
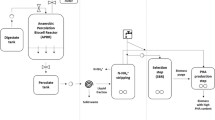
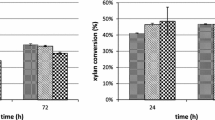
On partial analysis of the solid oligomeric waste of a nylon-6 production plant, it was found to contain ε-caprolactam, 6-aminocaproic acid (6-ACA) and its linear and cyclic oligomers. Out of four bacterial isolates capable of utilizing caprolactam as the sole growth substrate, Alcaligenes faecalis was found to be the most potent and utilized 90% of caprolactam in 24 h. In shake flask experiments, when the solid waste after solubilization was treated with a consortium of bacteria of four different genera, except the cyclic oligomers, all the other constituents were found to be degraded. A reduction of the chemical oxygen demand (COD) of the solid waste to the level of 63–66% was obtained when it was treated with either a consortium of the bacterial isolates or only a single isolate, A. faecalis. Alcaligenes faecalis could bring about a decrease of 95% in the caprolactam content of the solid waste, while 6-ACA and its linear oligomers were almost completely degraded. Alcaligenes faecalis cells adapted on solid waste could degrade the linear oligomers at a faster rate as compared to cells adapted on caprolactam. However, cyclic oligomers could not be degraded in either case. When solid waste, partially hydrolysed with acid to yield 6-ACA as the major constituent, was treated with the consortium of bacterial isolates, a 95% reduction in the COD was achieved.
Similar content being viewed by others
References
Ashtaputre, A.A. & Shah, A.K. 1995 Studies on a viscous, gel-forming exopolysaccharide from Sphingomonas paucimobilis GS1. Applied and Environmental Microbiology 61, 1159-1162.
Bartha, R. 1990 Isolation of microorganisms that metabolise xenobiotic compounds. In Isolation of Biotechnological Organisms from Nature, ed. Labeda, D.P., pp. 283-307. New York: McGraw-Hill Co. Inc. ISBN 0-07-035701-3.
Bergmann, F. 1952 Colorimetric determination of amides as hydroxamic acids. Analytical Chemistry 24, 1367-1369.
Bonifaci, L., Frezzotti, D., Cavalca, G., Malagutti, E. & Ravanetti, G.P. 1991 Analysis of e-caprolactam and its cyclic oligomers by high performance liquid chromatography. Journal of Chromatography 585, 333-336.
Clesceri, L.S., Greenberg, A.E. & Trussel, R.R. 1989 Chemical oxygen demand. In Standard Methods for the Examination of Water and Waste Water, 17th edn. pp. 5-15. Washington DC: American Public Health Association (APHA). American Water Works Association and Water Pollution Control Federation. ISBN 0-87553-161-X.
Fukumura, T., Takeuchi, M. & Banno, I. 1982 Stepwise loss of e-aminocaproic acid cyclic dimer in Alcaligenes species D-2. European Journal of Applied Microbiology and Biotechnology 14, 120-124.
Fukumura, T. 1966a Bacterial breakdown of ɛ-caprolactam and its cyclic oligomers. Plant and Cell Physiology 7, 93-104.
Fukumura, T. 1966b Hydrolysis of cyclic and linear oligomers of 6-aminocaproic acid by a bacterial cell extract. Journal of Biochemistry 59, 531-536.
Fukumura, T. 1966c Two bacterial enzymes hydrolysing oligomers of 6-aminocaproic acid. Journal of Biochemistry 59, 537-544.
Gross, P. 1984 Biologic activity of ɛ-caprolactam. CRC Critical Reviews in Toxicology 13, 205-216.
Kinoshita, S., Kageyama, S., Iba, K., Yamada, Y. & Okada, H. 1975 Utilisation of a cyclic dimer and linear oligomers of ɛ-aminocaproic acid by Achromobacter guttatus K172. Agricultural and Biological Chemistry 39, 1219-1223.
Kobayashi, Y. 1966 Thin layer chromatography of ɛ-caprolactam and its cyclic oligomers. Journal of Chromatography 24, 447-450.
Kulkarni, R.S. & Kanekar, P.P. 1998 Bioremediation of ɛ-caprolactam from nylon-6 waste water by use of Pseudomonas aeruginosa MCM B-407. Current Microbiology 37, 191-194.
Kulkarni, R.S. & Kanekar, P.P. 1997 Simultaneous determination of e-caprolactam and ɛ-aminocaproic acid by high performance liquid chromatography. Process Control and Quality 9, 31-37.
Mori, S. & Takeuchi, T. 1970a Gel permeation chromatography of the cyclic monomers and oligomers in nylon-6 and nylon-6,6. Journal of Chromatography 49, 230-238.
Mori, S. & Takeuchi, T. 1970b Gel permeation chromatography of the linear monomer and oligomers in polyamides. Journal of Chromatography 50, 419-428.
Plummer, D.T. 1971 An Introduction to Practical Biochemistry. pp. 153-154. New Delhi: Tata McGraw-Hill Co. Ltd.
Schmidt, O. 1986 Natural materials. In Biotechnology Vol 8-Microbial Degradations, eds. Rehm, H.J., Reed, G. & Schönborn, W., pp. 557-582. Weinheim: VCH Pub. ISBN 3-527-25770-5.
Schönborn, W. 1986 Historical developments and ecologic fundamentals. In Biotechnology Vol 8-Microbial Degradations, eds. Rehm, H.J., Reed, G. & Schönborn, W., pp. 3-41. Weinheim: VCH Pub. ISBN 3-527-25770-5.
Shama, G. & Wase, D.A.J. 1981 The biodegradation of ö-caprolactam and some related compounds. A Review. International Biodeterioration Bulletin 17, 1-9.
Sheldon, T. 1989 Chromosomal damage induced by caprolactam in human lymphocytes. Mutation Research 224, 325-327.
Snider, O.E. & Richardson, R.J. 1969 Polyamide fibres. In Encyclopedia of Polymer Science and Technology, ed. Bikales, N.M., vol 10, pp. 347-460. NewYork: Interscience.
Vogel, E.W. 1989 Caprolactam induces genetic alterations in early germ cell stages and in somatic tissue of D. melanogaster. Mutation Research 224, 339-342.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baxi, N., Shah, A. Biological treatment of the components of solid oligomeric waste from a nylon-6 production plant. World Journal of Microbiology and Biotechnology 16, 835–840 (2000). https://doi.org/10.1023/A:1008971216941
Issue Date:
DOI: https://doi.org/10.1023/A:1008971216941