Abstract
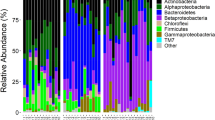
Aquatic larval habitat conditions influence the development, fitness and vectorial capacity of mosquitoes. In turn, human activities can influence these conditions and shape mosquito vector distribution, thus affecting pathogen circulation and transmission. We measured environmental factors (chemical and microbial) in mosquito larval habitats and evaluated their potential as predictors for the occurrence of different mosquito species in an arbovirus and malaria endemic region of western Kenya. We found significantly greater proportions of Aedes aegypti mosquito larvae in ammonium and phosphate rich habitats, suggesting that fertilizer usage could potentially increase aquatic habitat suitability and Aedes vectored disease transmission. Anopheles gambiae larval habitats correlated significantly with higher temperatures. However, none of the Culex species’ habitats correlated with investigated variables, indicating greater larval plasticity compared to Aedes or Anopheles. Profiling of bacterial communities by 16S rRNA pyrosequencing revealed Proteobacteria, Bacterioidetes and Firmicutes as the major bacteria phyla present in mosquito aquatic microhabitats. Although there were no correlations between microbiota composition or diversity and larval species abundance, the dominant genera of microbes detected in larval habitats were reported in larval and adult mid-guts, suggesting that bacteria acquired from the larval habitats are transmitted to adult stages. This study identified the chemical and bacterial composition of aquatic microhabitats that are conducive to the development of different mosquito vectors in western Kenya. This information can inform potential vector control strategies by assessing management of breeding sites based on likely exposure to fertilizer, light and bacterial fauna.
Similar content being viewed by others
References
Altschul S. F., Gish W., Miller W., Myers E. W. and Lipman D. J. (1990) Basic local alignment search tool. Journal of Molecular Biology 215, 403–410.
Bayoh M. N. and Lindsay S. W. (2003) Effect of temperature on the development of the aquatic stages of Anopheles gambiae sensu stricto (Diptera: Culicidae). Bulletin of Entomological Research 93, 375–381.
Bennun L. A. and Njoroge P. (2000) Important bird areas in Kenya. Ostrich — Journal of African Ornithology 71, 164–167. doi: https://doi.org/10.1080/00306525.2000.9639900.
Briones A. M., Shililu J., Githure J., Novak R. and Raskin L. (2008) Thorsellia anophelis is the dominant bacterium in a Kenyan population of adult Anopheles gambiae mosquitoes. ISME Journal 2, 74–82. doi: https://doi.org/10.1038/ismej.2007.95.
Caporaso J. G., Kuczynski J., Stombaugh J., Bittinger K., Bushman F. D., Costello E. K., Fierer N., Peña A. G., Goodrich J. K., Gordon J. I., Huttley G. A., Kelley S. T., Knights D., Koenig J. E., Ley R. E., Lozupone C. A., McDonald D., Muegge B. D., Pirrung M., Reeder J., Sevinsky J. R., Turnbaugh P. J., Walters W. A., Widmann J., Yatsunenko T., Zaneveld J. and Knight R. (2010) QIIME allows analysis of high-throughput community sequencing data. Nature Methods 7, 335–336. doi:https://doi.org/10.1038/nmeth.f.303.
Darriet F. (2015) An anti-mosquito mixture for domestic use, combining a fertiliser and a chemical or biological larvicide. Pest Management Science. doi: https://doi.org/10.1002/ps.4157.
Darriet F. and Corbel V. (2008) Aedes aegypti oviposition in response to NPK fertilizers [Article in French]. Parasite 15, 89–92.
Darriet F., Zumbo B., Corbel V. and Chandre F. (2010) Influence of plant matter and NPK fertilizer on the biology of Aedes aegypti (Diptera: Culicidae) [article in French]. Parasite 17, 149–154.
Djadid N. D., Jazayeri H., Raz A., Favia G., Ricci I. and Zakeri S. (2011) Identification of the midgut microbiota of An. stephensi and An. maculipennis for their application as a paratransgenic tool against malaria. PLOS ONE 6, e28484.
Dong Y., Manfredini F. and Dimopoulos G. (2009) Implication of the mosquito midgut microbiota in the defense against malaria parasites. PLOS Pathogens 5, e1000423. doi:https://doi.org/10.1371/journal.ppat.1000423.
Duguma D., Rugman-Jones P., Kaufman M. G., Hall M. W., Neufeld J. D., Stouthamer R. and Walton W. E. (2013) Bacterial communities associated with Culex mosquito larvae and two emergent aquatic plants of bioremediation importance. PLOS ONE 8, e72522. doi:https://doi.org/10.1371/journal.pone.0072522.
Edgar R. C. (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461.
Edwards F. W. (1941) Mosquitoes of the Ethiopian region. III. Culicine Adults and Pupae. British Museum (Natural History), London. 499 pp.
Engel P. and Moran N. A. (2013) The gut microbiota of insects — diversity in structure and function. FEMS Microbiology Reviews 37, 699–735. doi: https://doi.org/10.1111/1574-6976.12025.
Fillinger U., Ndenga B., Githeko A. and Lindsay S. W. Integrated malaria vector control with microbial larvicides and insecticide-treated nets in western Kenya: a controlled trial. Bulletin of the World Health Organization 87, 655–665.
Folmer O., Black M., Hoeh W., Lutz R. and Vrijenhoek R. (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Molecular Marine Biology and Biotechnology 3, 294–299.
Fouda M. A., Hassan M. I., Al- Daly A. G. and Hammad K. M. (2001) Effect of midgut bacteria of Culex pipiens L. on digestion and reproduction. Journal of the Egyptian Society of Parasitology 31, 767–780.
Gaio A. D., Gusmao D. S., Santos A. V., Berbert-Molina M. A., Pimenta P. F. P. and Lemos F. J. A. (2011) Contribution of midgut bacteria to blood digestion and egg production in Aedes aegypti (Diptera: Culicidae) (L.). Parasites & Vectors 4, 105. doi:https://doi.org/10.1186/1756-3305- 4-105.
Garcia-Rejon J. E., Blitvich B.J., Farfan-Ale J. A., Loroño-Pino M. A., Chi Chim W. A., Flores-Flores L. F., Rosado-Paredes E., Baak-Baak C., Perez-Mutul J., Suarez-Solis V., Fernandez-Salas I. and Beaty B. J. Host-feeding preference of the mosquito, Culex quinquefasciatus, in Yucatan State, Mexico. Journal of Insect Science 10, 32. doi: https://doi.org/10.1673/031.010.3201.
Gerberg E. J., Barbard D. R. and Ward R. A. (1994) Manual for Mosquito Rearing and Experimental Techniques. American Mosquito Control Association Inc., Lake Charles, Louisiana. 98 pp.
Geser A., Henderson B. E. and Christensen S. (1970) A multipurpose serological survey in Kenya. 2. Results of arbovirus serological tests. Bulletin of the World Health Organization 43, 539–552.
Gillies M. T. and Coetzee M. (1987) A Supplement to the Anophelinae of Africa South of the Sahara (Afrotropical Region). South African Institute for Medical Research, Johannesburg, South Africa. Vol. 55, 143 pp.
Gusmäo D. S., Santos A. V., Marini D. C., Russo E. D., Peixoto A. M. D., Bacci M., Berbert-Molina M. A. and Lemos F. J. A. (2007) First isolation of microorganisms from the gut diverticulum of Aedes aegypti (Diptera: Culicidae): new perspectives for an insect-bacteria association. Memórias do Instituto Oswaldo Cruz 102, 919–924.
Hoffmann A. A., Montgomery B. L., Popovici J., Iturbe-Ormaetxe I., Johnson P. H., Muzzi F., Greenfield M., Durkan M., Leong Y. S., Dong Y., Cook H., Axford J., Callahan A. G., Kenny N., Omodei C., McGraw E. A., Ryan P. A., Ritchie S. A., Turelli M. and O’Neill S. L. Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 476, 454–457. doi:https://doi.org/10.1038/nature10356.
Lane D. J. (1991) 16S/23S rRNA sequencing, pp. 115–175. In Nucleic Acid Techniques in Bacterial Systematics (edited by E. Stackebrandt and M. Goodfellow). John Wiley and Sons, New York.
Le Menach A., McKenzie F. E., Flahault A. and Smith D. L. (2005) The unexpected importance of mosquito oviposition behaviour for malaria: non-productive larval habitats can be sources for malaria transmission. Malaria Journal 4, 23. doi: https://doi.org/10.1186/1475-2875-4-23.
Lutomiah J., Bast J., Clark J., Richardson J., Yalwala S., Oullo D., Mutisya J., Mulwa F., Musila L., Khamadi S., Schnabel D., Wurapa E. and Sang R. (2013) Abundance, diversity, and distribution of mosquito vectors in selected ecological regions of Kenya: public health implications. Journal of Vector Ecology 38, 134–142.
Mala A. O. and Irungu L. W. (2011) Factors influencing differential larval habitat productivity of Anopheles gambiae complex mosquitoes in a western Kenyan village. Journal of Vector Borne Diseases 48, 52–57.
Mease L. E., Coldren R. L., Musila L. A., Prosser T., Ogolla F., Ofula V. O., Schoepp R.J., Rossi C.A. and Adungo N. (2011) Seroprevalence and distribution of arboviral infections among rural Kenyan adults: a cross-sectional study. Virology Journal 8, 371. doi: https://doi.org/10.1186/1743-422X-8-371.
Merritt R. W., Dadd R. H. and Walker E. D. (1992) Feeding behavior, natural food, and nutritional relationships of larval mosquitoes. Annual Review of Entomology 37, 349–374. doi: https://doi.org/10.1146/annurev.en.37.010192.002025.
Minakawa N., Mutero C. M., Githure J. I., Beier J. C. and Yan G. (1999) Spatial distribution and habitat characterization of anopheline mosquito larvae in western Kenya. American Journal of Tropical Medicine and Hygiene 61, 1010–1016.
Minard G., Tran F. H., Raharimalala F. N., Hellard E., Ravelonandro P., Mavingui P. and Valiente Moro C. (2013) Prevalence, genomic and metabolic profiles of Acinetobacter and Asaia associated with field-caught Aedes albopictus from Madagascar. FEMS Microbiology Ecology 83, 63–73. doi: https://doi.org/10.1111/j.1574-6941.2012.01455.x.
Montero-Pau J., Gómez A. and Muñoz J. (2008) Application of an inexpensive and high-throughput genomic DNA extraction method for the molecular ecology of zooplanktonic diapausing eggs. Limnology and Oceanography: Methods 6, 218–222. doi: https://doi.org/10.4319/lom.2008.6.218.
Moro C. V., Tran F. H., Raharimalala F. N., Ravelonandro P. and Mavingui P. (2013) Diversity of culturable bacteria including Pantoea in wild mosquito Aedes albopictus. BMC Microbiology 13, 70. doi:https://doi.org/10.1186/1471-2180-13-70.
Mutero C. M., Ng’ang’a P. N., Wekoyela P., Githure J. and Konradsen F. (2004) Ammonium sulphate fertiliser increases larval populations of Anopheles arabiensis and culicine mosquitoes in rice fields. Acta Tropica 89, 187–192.
Mwangangi J. M., Muturi E. J., Shililu J.I., Muriu S., Jacob B., Kabiru E. W., Mbogo C. M., Githure J. I. and Novak R. J. (2007) Environmental covariates of Anopheles arabiensis in a rice agroecosystem in Mwea, Central Kenya. Journal of the American Mosquito Control Association 23, 371–377.
Mwangangi J. M., Shililu J., Muturi E. J., Muriu S., Jacob B., Kabiru E. W., Mbogo C. M., Githure J. and Novak R. J. (2010) Anopheles larval abundance and diversity in three rice agro-village complexes Mwea irrigation scheme, central Kenya. Malaria Journal 9, 228. doi: https://doi.org/10.1186/1475-2875-9-228.
Ochieng C., Lutomiah J., Makio A., Koka H., Chepkorir E., Yalwala S., Mutisya J., Musila L., Khamadi S., Richardson J., Bast J., Schnabel D., Wurapa E. and Sang R. (2013) Mosquito-borne arbovirus surveillance at selected sites in diverse ecological zones of Kenya; 2007–2012. Virology Journal 10, 140. doi: https://doi.org/10.1186/1743-422X-10-140.
Omondi D., Masiga D. K., Ajamma Y. U., Fielding B. C., Njoroge L. and Villinger J. (2015) Unraveling host- vector-arbovirus interactions by two-gene high resolution melting mosquito bloodmeal analysis in a Kenyan wildlife-livestock interface. PLOS ONE 10, e0134375. doi: https://doi.org/10.1371/journal.pone.0134375.
Osei-Poku J., Mbogo C. M., Palmer W. J. and Jiggins F. M. Deep sequencing reveals extensive variation in the gut microbiota of wild mosquitoes from Kenya. Molecular Ecology 21, 5138–5150. doi: https://doi.org/10.1111/j.1365-294X.2012.05759.x.
Ponnusamy L., Wesson D. M., Arellano C., Schal C. and Apperson C. S. (2010) Species composition of bacterial communities influences attraction of mosquitoes to experimental plant infusions. Microbial Ecology 59, 158–173. doi: https://doi.org/10.1007/s00248-009-9565-1.
Ponnusamy L., Xu N., Nojima S., Wesson D. M., Schal C. and Apperson C. S. (2008) Identification of bacteria and bacteria-associated chemical cues that mediate oviposition site preferences by Aedes aegypti. Proceedings of the National Academy of Sciences USA 105, 9262–9267. doi: https://doi.org/10.1073/pnas.0802505105.
Powell J. R. and Tabachnick W. J. (2013) History of domestication and spread of Aedes aegypti — A review. Memórias do Instituto Oswaldo Cruz 108(Suppl 1), 11–17. doi: https://doi.org/10.1590/0074-0276130395.
R Development Core Team (2008) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3900051-07-0, URL.https://doi.org/www.R-project.org.
Rani A., Sharma A., Rajagopal R., Adak T. and Bhatnagar R. K. (2009) Bacterial diversity analysis of larvae and adult midgut microflora using culture-dependent and culture-independent methods in lab-reared and field- collected Anopheles stephensi-an Asian malarial vector. BMC Microbiology 9, 96. doi: https://doi.org/10.1186/1471-2180-9-96.
Richards S. L., Anderson S. L. and Yost S. A. (2012) Effects of blood meal source on the reproduction of Culex pipiens quinquefasciatus (Diptera: Culicidae). Journal of Vector Ecology 37, 1–7.
Sumba L. A., Guda T. O., Deng A. L., Hassanali A., Beier J. C. and Knols B. G. J. (2004) Mediation of oviposition site selection in the African malaria mosquito Anopheles gambiae (Diptera: Culicidae) by semiochemicals of microbial origin. International Journal of Tropical Insect Science 24, 260–265. doi: https://doi.org/10.1079/IJT200433.
Turner S., Pryer K. M., Miao V. P. and Palmer J. D. (1999) Investigating deep phylogenetic relationships among cyanobacteria and plastids by small subunit rRNA sequence analysis. Journal ofEukaryotic Microbiology 46, 327–338.
Victor T. J. and Reuben R. (2000) Effects of organic and inorganic fertilisers on mosquito populations in rice fields of southern India. Medical and Veterinary Entomology 14, 361–368.
Walker T., Johnson P. H., Moreira L. A., Iturbe-Ormaetxe I., Frentiu F. D., McMeniman C. J., Leong Y. S., Dong Y., Axford J., Kriesner P., Lloyd A. L., Ritchie S. A., O’ Neill S. L. and Hoffmann A. A. (2011) The wMel Wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature 476, 450–453. doi:https://doi.org/10.1038/nature10355.
Wamae P. M., Githeko A. K., Menya D. M. and Takken W. (2010) Shading by Napier grass reduces malaria vector larvae in natural habitats in western Kenya highlands. EcoHealth 7, 485–497. doi: https://doi.org/10.1007/s10393-010-0321-2.
Wang Q., Garrity G. M., Tiedje J. M. and Cole J. R. (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Applied and Environmental Microbiology 73, 5261–5267.
Wang Y., Gilbreath T. M. III, Kukutla P., Yan G. and Xu J. (2011) Dynamic gut microbiome across life history of the malaria mosquito Anopheles gambiae in Kenya. PLOS ONE 6, e24767. doi:https://doi.org/10.1371/journal.pone.0024767.
Weaver S.C. and Barrett A.D.T. (2004) Transmission cycles, host range, evolution and emergence of arboviral disease. Nature Reviews Microbiology 2, 789–801. doi:https://doi.org/10.1038/nrmicro1006.
Weiss B. L., Maltz M. and Aksoy S. (2012) Obligate symbionts activate immune system development in the tsetse fly. Journal of Immunology 188, 3395–3403. doi: https://doi.org/10.4049/jimmunol.1103691.
Weiss B. L., Wang J. and Aksoy S. (2011) Tsetse immune system maturation requires the presence of obligate symbionts in larvae. PLOS Biology 9, e1000619. doi:https://doi.org/10.1371/journal.pbio.1000619.
Yeap H. L., Axford J. K., Popovici J., Endersby N. M., Iturbe-Ormaetxe I., Ritchie S. A. and Hoffmann A. A. (2014) Assessing quality of life-shortening Wolbachia- infected Aedes aegypti mosquitoes in the field based on capture rates and morphometric assessments. Parasites & Vectors 7, 58. doi: https://doi.org/10.1186/1756-3305-7-58.
Yuill T. M. (1986) The ecology of tropical arthropodborne viruses. Annual Review of Ecology, Evolution, and Systematics 17, 189–219.
Zouache K., Raharimalala F. N., Raquin V., Tran-Van V., Raveloson L. H., Ravelonandro P. and Mavingui P. (2011) Bacterial diversity of field-caught mosquitoes, Aedes albopictus and Aedes aegypti, from different geographic regions of Madagascar. FEMS Microbiology Ecology 75, 377–389. doi: https://doi.org/10.1111/j.1574-6941.2010.01012.x.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Onchuru, T.O., Ajamma, Y.U., Burugu, M. et al. Chemical parameters and bacterial communities associated with larval habitats of Anopheles, Culex and Aedes mosquitoes (Diptera: Culicidae) in western Kenya. Int J Trop Insect Sci 36, 146–160 (2016). https://doi.org/10.1017/S1742758416000096
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1017/S1742758416000096