Abstract
Background
To compare pharmacologic effects of pirenzepine and AF-DX116, a selective competitive antagonist for M1 and M2 subtype muscarinic cholinergic receptors (mAChRs), respectively, with atropine, a non-selective competitive antagonist for mAChRs, on Lipopolysaccharide (LPS).
Methods
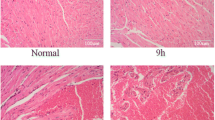
Male C57BL/6 mice were used to establish models of LPS-induced experimental endotoxemia. Mice were intraperitoneally injected 10 min prior to LPS injection with control (saline), atropine, pirenzepine and AF-DX116, respectively. Overall survival time was estimated using Kaplan-Meier plots. Inflammatory cytokine tumor necrosis factor-α (TNF-α) was monitored at various intervals after LPS injection and individual reagent administration. Pathological alternations in lungs and liver were analyzed.
Results
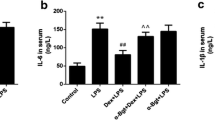
Pirenzepine and atropine pretreatment improved survival rate of LPS-induced septic shock; in contrast, AF-DX116 accelerated death from sepsis. Moreover, TNF-α plasma level was decreased in response to pirenzepine or atropine, whereas increased in response to AF-DX116. Pirenzepine and atropine relieved whereas AF-DX116 accelerated LPS-induced pulmonary and hepatic injury. Pirenzepine reduced proportion of M1 subtype of macrophages, while AF-DX116 promoted polarization of macrophages to M1 subtype. Pirenzepine pretreatment reduced while AF-DX116 enhanced expression of SOCS3 at mRNA level.
Conclusions
The administration of pirenzepine and atropine may have beneficial effects on septic shock.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for Sepsis and septic shock (Sepsis-3). JAMA 2016;315(8):801–10.
Shankar-Hari M, Phillips GS, Levy ML, Seymour CW, Liu VX, Deutschman CS, et al. Developing a new definition and assessing new clinical criteria for septic shock: for the third international consensus definitions for Sepsis and septic shock (Sepsis-3). JAMA 2016;315(8):775–87.
Seymour CW, Liu VX, Iwashyna TJ, Brunkhorst FM, Rea TD, Scherag A, et al. Assessment of clinical criteria for Sepsis For the third international consensus definitions for Sepsis and septic shock (Sepsis-3). JAMA 2016;315(8):762–74.
van Vught LA, Wiewel MA, Hoogendijk AJ, Frencken JF, Scicluna BP, Klein Klouwenberg PMC, et al. The host response in Sepsis patients developing intensive care unit-acquired secondary infections. Am J Respir Crit Care Med 2017;196(August (4)):458–70.
Nolan A, Weiden MD. Trends in Sepsis and infection sources in the United States. A population-based study. Ann Am Thorac Soc 2015;12(2):216–20.
van der Poll T, van de Veerdonk FL, Scicluna BP, Netea MG, et al. The immunopathology of sepsis and potential therapeutic targets. Nat Rev Immunol 2017;17(July (7)):407–20.
Opal SM. The evolution of the understanding of sepsis, infection, and the host response: a brief history. Crit Care Nurs Clin North Am 2011;23(1):1–27.
Yamamoto Y, Harashima A, Saito H, Tsuneyama K, Munesue S, Motoyoshi S, et al. Septic shock is associated with receptor for advanced glycation end products ligation of LPS. J Immunol 2011;186(5):3248–57.
Karpurapu M, Wang X, Deng J, Park H, Xiao L, Sadikot RT, et al. Functional PU.1 in macrophages has a pivotal role in NF-kB activation and neutrophilic lung inflammation during endotoxemia. Blood 2011;118(19):5255–66.
Huston JM. The vagus nerve and the inflammatory reflex: wandering on a new treatment paradigm for systemic inflammation and sepsis. Surg Infect (Larchmt) 2012;13(4):187–93.
Villegas-Bastida A, Torres-Rosas R, Arriaga-Pizano LA, Flores-Estrada J, Gustavo-Acosta A, Moreno-Eutimio MA. Electrical stimulation at the ST36 acupoint protects against Sepsis lethality and reduces serum TNF levels through vagus nerve- and catecholamine-dependent mechanisms. Evid Based Complement Alternat Med 2014;2014:451674.
Matsuda A, Jacob A, Wu R, Aziz M, Yang WL, Matsutani T, et al. Novel therapeutic targets for sepsis: regulation of exaggerated inflammatory responses. J Nippon Med Sch 2012;79(1):4–18.
Borovikova LV, Ivanova S, Zhang M, Yang H, Botchkina GI, Watkins LR, et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 2000;405(6785):458–62.
Zhang M, Wang H, Tracey KJ. Regulation of macrophage activation and inflammation by spermine: a new chapter in an old story. Crit Care Med 2000;28(4 Suppl):N60–6.
Czura CJ, Friedman SG, Tracey KJ. Neural inhibition of inflammation: the cholinergic anti-inflammatory pathway. J Endotoxin Res 2003;9(6):409–13.
Wang H, Yu M, Ochani M, Amella CA, Tanovic M, Susarla S, et al. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature 2003;421(6921):384–8.
Olofsson PS, Katz DA, Rosas-Ballina M, Levine YA, Ochani M, Valdes-Ferrer SI, et al. alpha7 nicotinic acetylcholine receptor (alpha7nAChR) expression in bone marrow-derived non-T cells is required for the inflammatory reflex. Mol Med 2012;18:539–43.
Lu B, Kwan K, Levine YA, Olofsson PS, Yang H, Li J, et al. alpha7 nicotinic acetylcholine receptor signaling inhibits inflammasome activation by preventing mitochondrial DNA release. Mol Med 2014;20:350–8.
Haga T. Molecular properties of muscarinic acetylcholine receptors. Proc Jpn Acad, Ser B, Phys Biol Sci 2013;89(6):226–56.
Wess J. Muscarinic acetylcholine receptor knockout mice: novel phenotypes and clinical implications. Annu Rev Pharmacol Toxicol 2004;44:423–50.
Fuentes JM, Fulton WB, Nino D, Talamini MA, Maioe AD. Atropine treatment modifi es LPS-induced infl amatory response and increases survival. Inflamm Res 2008;57(3):111–7.
Lolmede K, Campana L, Vezzoli M, Bosurgi L, Tonlorenzi R, Clementi E, et al. Inflammatory and alternatively activated human macrophages attract vessel-associated stem cells, relying on separate HMGB1- and MMP-9-dependent pathways. J Leukoc Biol 2009;85(5):779–87.
Jensen KD, Wang Y, Wojno ED, Shastri AJ, Hu K, Cornel L, et al. Toxoplasma polymorphic effectors determine macrophage polarization and intestinal inflammation. Cell Host Microbe 2011;9(6):472–83.
Khanduja KL, Kaushik G, Khanduja S, Pathak CM, Laldinpuii J, Behera D. Corticosteroids affect nitric oxide generation, total free radicals production, and nitric oxide synthase activity in monocytes of asthmatic patients. Mol Cell Biochem 2011;346(1):31–7.
Catal F, Mete E, Tayman C, Topal E, Albayrak A, Sert H. A human monoclonal anti-TNF alpha antibody (adalimumab) reduces airway inflammation and ameliorates lung histology in a murine model of acute asthma. Allergol Immunopathol 2014;43(1):14–8.
Yoshimura A, Naka T, Kubo M. SOCS proteins, cytokine signalling and immune regulation. Nat Rev Immunol 2007;7(6):454–65.
Dalpke A, Heeg K, Bartz H, Baetz A. Regulation of innate immunity by suppressor of cytokine signaling (SOCS) proteins. Immunobiology 2008;213(3):225–35.
Liu Y, Stewart KN, Bishop E, Marek CJ, Kluth DC, Rees AJ. Unique expression of suppressor of cytokine signaling 3 is essential for classical macrophage activation in rodents in vitro and in vivo. J Immunol 2008;180(9):6270–8.
Yasukawa H, Ohishi M, Mori H, Murakami M, Chinen T, Aki D, etal. IL-6 induces an anti-inflammatory response in the absence of SOCS3 in macrophages. Nat Immunol 2003;4(6):551.
Author information
Authors and Affiliations
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit https://doi.org/creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Z., Li, M., Liu, L. et al. Muscarinic M1 and M2 receptor subtypes play opposite roles in LPS-induced septic shock. Pharmacol. Rep 71, 1108–1114 (2019). https://doi.org/10.1016/j.pharep.2019.06.005
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2019.06.005