Summary
Voltage-gated calcium channels (VGCC) play obligatory roles in diverse physiological functions. Pathological conditions leading to changes in their biophysical properties and expression levels may cause malfunctions of VGCC-mediated activities, resulting in disease states. It is believed that changes in VGCC properties under pain-inducing conditions may play a causal role in the development of chronic pain, including nerve injury-induced pain or neuropathic pain. For the past several decades, preclinical and clinical research in developing VGCC blockers or modulators for chronic pain management has been fruitful, leading to some U.S. Food and Drug Administration-approved drugs currently available for chronic pain management. However, their efficacy in pain relief is limited in some patients, and their long-term use is limited by their side-effect profiles. Certainly, there is room for improvement in developing more subtype-specific VGCC blockers or modulators for chronic pain conditions. In this review, we summarized the most recent preclinical and clinical studies related to chronic pain medications acting on the VGCC. We also included clinical trials aiming to expand the application of approved VGCC drugs to different pain states derived from various pathological conditions, as well as drug combination therapies trying to improve the efficacies and side-effect profiles of current pain medications.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mitka M. “Virtual textbook” on pain developed: effort seeks to remedy gap in medical education. JAMA 2003;290: 2395.
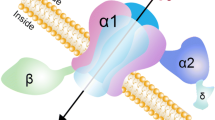
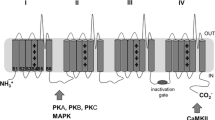
Catterall WA. Structure and regulation of voltage-gated Ca2+ channels. Annu Rev Cell Dev Biol 2000;16: 521–555.
Dolphin AC. Beta subunits of voltage-gated calcium channels. J Bioenerg Biomembr 2003;35: 599–620.
Dolphin AC. Beta subunits of voltage-gated calcium channels. J Bioenerg Biomembr 2003;35: 599–620.
Klugbauer N, Lacinova L, Marais E, Hobom M, Hofmann F. Molecular diversity of the calcium channel a2d subunit. J Neurosci 1999;19: 684–691.
Qin N, Yagel S, Momplaisir ML, Codd EE, D’Andrea MR. Molecular cloning and characterization of the human voltage-gated calcium channel alpha(2)delta-4 subunit. Mol Pharmacol 2002;62: 485–496.
Marais E, Klugbauer N, Hofmann F. Calcium channel alpha2delta subunits-structure and gabapentin binding. Mol Pharmacol 2001;59: 1243–1248.
Kim HL, Kim H, Lee P, King RG, Chin H. Rat brain expresses an alternatively spliced form of the dihydropyridine-sensitive L-type calcium channel alpha 2 subunit. Proc Natl Acad Sci U S A 1992;89: 3251–3255.
Angelotti T, Hofmann F. Tissue-specific expression of splice variants of the mouse voltage-gated calcium channel alpha2/delta subunit. Febs Letters 1996;397: 331–337.
Ellis SB, Williams ME, Ways NR, et al. Sequence and expression of mRNAs encoding the alpha 1 and alpha 2 subunits of a DHP-sensitive calcium channel. Science 1988;241: 1661–1664.
De Jongh KS, Warner C, Catterall WA. Subunits of purified calcium channels. Alpha 2 and delta are encoded by the same gene. J Biol Chem 1990;265: 14738–14741.
Brickley K, Campbell V, Berrow N, et al. Use of site-directed antibodies to probe the topography of the alpha 2 subunit of voltage-gated Ca2+ channels. Febs Letters 1995;364: 129–133.
Gurnett CA, De Waard M, Campbell KP. Dual function of the voltage-dependent Ca2+ channel alpha 2 delta subunit in current stimulation and subunit interaction. Neuron 1996;16: 431–440.
Wiser O, Trus M, Tobi D, Halevi S, Giladi E, Atlas D. The alpha 2/delta subunit of voltage sensitive Ca2+ channels is a single transmembrane extracellular protein which is involved in regulated secretion. Febs Letters 1996;379: 15–20.
Mikami A, Imoto K, Tanabe T, et al. Primary structure and functional expression of the cardiac dihydropyridine-sensitive calcium channel. Nature 1989;340: 230–233.
Mori Y, Friedrich T, Kim MS, et al. Primary structure and functional expression from complementary DNA of a brain calcium channel. Nature 1991;350: 398–402.
Hullin R, Singer-Lahat D, Freichel M, et al. Calcium channel beta subunit heterogeneity: functional expression of cloned cDNA from heart, aorta and brain. Embo J 1992;11: 885–890.
Williams ME, Feldman DH, McCue AF, et al. Structure and functional expression of alpha 1, alpha 2, and beta subunits of a novel human neuronal calcium channel subtype. Neuron 1992;8: 71–84.
Brust PF, Simerson S, McCue AF, et al. Human neuronal voltage-dependent calcium channels: studies on subunit structure and role in channel assembly. Neuropharmacology 1993;32: 1089–1102.
Kang MG, Felix R, Campbell KP. Long-term regulation of voltage-gated Ca(2+) channels by gabapentin. FEBS Lett 2002;528: 177–182.
Gurnett CA, Felix R, Campbell KP. Extracellular interaction of the voltage-dependent Ca2+ channel alpha2delta and alphal sub-units. J Biol Chem 1997;272: 18508–18512.
Wolf M, Eberhart A, Glossmann H, Striessnig J, Grigorieff N. Visualization of the domain structure of an L-type Ca2+ channel using electron cryo-microscopy. J Mol Biol 2003;332: 171–182.
Bauer CS, Simerson S, McCue AF, et al. The increased trafficking of the calcium channel subunit alpha2delta-1 to presynaptic terminals in neuropathic pain is inhibited by the alpha2delta ligand pregabalin. J Neurosci 2009;29: 4076–4088.
Hendrich J, Van Minh AT, Heblich F, et al. Pharmacological disruption of calcium channel trafficking by the alpha2delta ligand gabapentin. Proc Natl Acad Sci U S A 2008;105: 3628–3633.
Takahashi M, Seagar MJ, Jones JF, Reber BF, Catterall WA. Subunit structure of dihydropyridine-sensitive calcium channels from skeletal muscle. Proc Natl Acad Sci U S A 1987;84: 5478–5482.
Catterall WA. Structure and function of voltage-sensitive ion channels. Science 1988;242: 50–61.
Catterall WA. Functional subunit structure of voltage-gated calcium channels. Science 1991;253: 1499–1500.
Black JL 3rd, Lennon VA. Identification and cloning of putative human neuronal voltage-gated calcium channel gamma-2 and gamma-3 subunits: neurologic implications. Mayo Clin Proc 1999;74: 357–361.
Kang MG, Chen CC, Felix R, et al. Biochemical and biophysical evidence for gamma 2 subunit association with neuronal voltage-activated Ca2+ channels. J Biol Chem 2001;276: 32917–32924.
Klugbauer N, Dai S, Specht V, et al. A family of gamma-like calcium channel subunits. FEBS Lett 2000;470: 189–197.
Letts VA, Felix R, Biddlecome GH, et al. The mouse stargazer gene encodes a neuronal Ca2+-channel gamma subunit. Nat Genet 1998;19: 340–347.
Moss FJ, Viard P, Davies A, et al. The novel product of a five-exon stargazin-related gene abolishes Ca(V)2.2 calcium channel expression. Embo J 2002;21: 1514–1523.
Eitel EA, Campbell KP, Harpold MM, et al. Nomenclature of voltage-gated calcium channels. Neuron 2000;25: 533–535.
Jan LY, Jan YN. Voltage-sensitive ion channels. Cell 1989;56: 13–25.
Spedding M, Lepagnol J. Pharmacology of sodium and calcium channel modulation in neurons: implications for neuroprotection. Biochem Soc Trans 1995;23: 633–636.
Tsien RW, Ellinor PT, Horne WA. Molecular diversity of voltage-dependent Ca2+ channels. Trends Pharmacol Sci 1991;12: 349–354.
Finkbeiner S, Greenberg ME. Ca2+ channel-regulated neuronal gene expression. J Neurobiol 1998;37: 171–189.
Belardetti F, Zamponi GW. Linking calcium-channel isoforms to potential therapies. Curr Opin Investig Drugs 2008;9: 707–715.
Davies JN, Zamponi GW. Old proteins, developing roles: the regulation of calcium channels by synaptic proteins. Channels (Austin) 2008;2: 130–138.
Zamponi GW, Lewis RJ, Todorovic SM, Arneric SP, Snutch TP. Role of voltage-gated calcium channels in ascending pain pathways. Brain Res Rev 2009;60: 84–89.
Catterall WA, Dib-Hajj S, Meisler MH, Pietrobon D. Inherited neuronal ion channelopathies: new windows on complex neurological diseases. J Neurosci 2008;28: 11768–11777.
Yaksh TL. Calcium channels as therapeutic targets in neuropathic pain. J Pain 2006;7: S13–30.
McGivem JG, McDonough SI. Voltage-gated calcium channels as targets for the treatment of chronic pain. Curr Drug Targets CNS Neurol Disord 2004;3: 457–478.
Bourinet E, Zamponi GW. Voltage gated calcium channels as targets for analgesics. Curr Top Med Chem 2005;5: 539–546.
Schroeder CI, Doering CJ, Zamponi GW, Lewis RJ. N-type calcium channel blockers: novel therapeutics for the treatment of pain. Med Chem 2006;2: 535–543.
Kisilevsky AE, Zamponi GW. Presynaptic calcium channels: structure, regulators, and blockers. Handb Exp Pharmacol 2008: 45–75.
Catterall WA, Few AP. Calcium channel regulation and presynaptic plasticity. Neuron 2008;59: 882–901.
Fuchs A, Rigaud M, Sarantopoulos CD, Filip P, Hogan QH. Contribution of calcium channel subtypes to the intracellular calcium signal in sensory neurons: the effect of injury. Anesthesiology 2007;107: 117–127.
Diaz A, Dickenson AH. Blockade of spinal N- and P-type, but not L-type, calcium channels inhibits the excitability of rat dorsal horn neurones produced by subcutaneous formalin inflammation. Pain 1997;69: 93–100.
Matthews EA, Dickenson AH. Effects of spinally delivered N- and P-type voltage-dependent calcium channel antagonists on dorsal horn neuronal responses in a rat model of neuropathy. Pain 2001;92: 235–246.
Matthews EA, Dickenson AH. Effects of ethosuximide, a T-type Ca(2+) channel blocker, on dorsal horn neuronal responses in rats. Eur J Pharmacol 2001;415: 141–149.
Vanegas H, Schaible H. Effects of antagonists to high-threshold calcium channels upon spinal mechanisms of pain, hyperalgesia and allodynia. Pain 2000;85: 9–18.
Westenbroek RE, Hell JW, Warner C, Dubel SJ, Snutch TP, Catterall WA. Biochemical properties and subcellular distribution of an N-type calcium channel alpha 1 subunit. Neuron 1992;9: 1099–1115.
Kerr LM, Filloux F, Olivera BM, Jackson H, Wamsley JK. Autoradiographic localization of calcium channels with [125I]omega-conotoxin in rat brain. Eur J Pharmacol 1988;146: 181–183.
Gohil K, Bell JR, Ramachandran J, Miljanich GP. Neuroanatomical distribution of receptors for a novel voltage-sensitive calcium-channel antagonist, SNX-230 (omega-conopeptide MVIIC). Brain Res 1994;653: 258–266.
Pertovaara A. Noradrenergic pain modulation. Prog Neurobiol 2006;80: 53–83.
Bourinet E, Soong TW, Stea A, Snutch TP. Determinants of the G protein-dependent opioid modulation of neuronal calcium channels. Proc Natl Acad Sci U S A 1996;93: 1486–1491.
Altier C, Zamponi GW. Targeting Ca2+ channels to treat pain: T-type versus N-type. Trends Pharmacol Sci 2004;25: 465–470.
Chaplan SR, Pogrel JW, Yaksh TL. Role of voltage-dependent calcium channel subtypes in experimental tactile allodynia. J Pharmacol Exp Ther 1994;269: 1117–1123.
Maggi CA, Tramontana M, Cecconi R, Santicioli P. Neurochemical evidence for the involvement of N-type calcium channels in transmitter secretion from peripheral endings of sensory nerves in guinea pigs. Neurosci Lett 1990;114: 203–206.
Santicioli P, Del Bianco E, Tramontana M, Geppetti P, Maggi CA. Release of calcitonin gene-related peptide like-immunoreactivity induced by electrical field stimulation from rat spinal afferents is mediated by conotoxin-sensitive calcium channels. Neurosci Lett 1992;136: 161–164.
Evans AR, Nicol GD, Vasko MR. Differential regulation of evoked peptide release by voltage-sensitive calcium channels in rat sensory neurons. Brain Res 1996;712: 265–273.
Saegusa H, Kurihara T, Zong S, et al. Suppression of inflammatory and neuropathic pain symptoms in mice lacking the N-type Ca2+ channel. Embo J 2001;20: 2349–2356.
Saegusa H, Matsuda Y, Tanabe T. Effects of ablation of N- and R-type Ca(2+) channels on pain transmission. Neurosci Res 2002;43: 1–7.
Hatakeyama S, Wakamori M, Ino M, et al. Differential nociceptive responses in mice lacking the alpha(1B) subunit of N-type Ca(2+) channels. Neuroreport 2001;12: 2423–2427.
Bell TJ, Thaler C, Castiglioni AJ, Helton TD, Lipscombe D. Cell-specific alternative splicing increases calcium channel current density in the pain pathway. Neuron 2004;41: 127–138.
Lin Y, McDonough SI, Lipscombe D. Alternative Splicing in the voltage-sensing region of N-type CaV2.2 channels modulates channel kinetics. J Neurophysiol 2004;92: 2820–2830.
Lin Z, Haus S, Edgerton J, Lipscombe D. Identification of functionally distinct isoforms of the N-type Ca2+ channel in rat sympathetic ganglia and brain. Neuron 1997;18: 153–166.
Miljanich GP. In: Venoms to drugs. Heron Island, Australia. 2005. Available at: http://www.venomstodrugs.com/index.html? page=107301&pid=0.
Hillyard DR, Monje VD, Mintz IM, et al. A new Conus peptide ligand for mammalian presynaptic Ca2+ channels. Neuron 1992; 9: 69–77.
Lewis RJ, Nielsen KJ, Craik DJ, et al. Novel omega-conotoxins from Conus catus discriminate among neuronal calcium channel subtypes. J Biol Chem 2000;275: 35335–35344.
Nadasdi L, Yamashiro D, Chung D, Tarczy-Homoch K, Adriaenssens P, Ramachandran J. Structure-activity analysis of a Conus peptide blocker of N-type neuronal calcium channels. Biochemistry 1995;34: 8076–8081.
Smith MT, Cabot PJ, Ross FB, Robertson AD, Lewis RJ. The novel N-type calcium channel blocker, AM336, produces potent dose-dependent antinociception after intrathecal dosing in rats and inhibits substance P release in rat spinal cord slices. Pain 2002;96: 119–127.
Wang YX, Gao D, Pettus M, Phillips C, Bowersox SS. Interactions of intrathecally administered ziconotide, a selective blocker of neuronal N-type voltage-sensitive calcium channels, with morphine on nociception in rats. Pain 2000;84: 271–281.
Wang YX, Pettus M, Gao D, Phillips C, Scott Bowersox S. Effects of intrathecal administration of ziconotide, a selective neuronal N-type calcium channel blocker, on mechanical allodynia and heat hyperalgesia in a rat model of postoperative pain. Pain 2000;84: 151–158.
Sluka KA. Blockade of N- and P/Q-type calcium channels reduces the secondary heat hyperalgesia induced by acute inflammation. J Pharmacol Exp Ther 1998;287: 232–237.
Lynch SS, Cheng CM, Yee JL. Intrathecal ziconotide for refractory chronic pain. Ann Pharmacother 2006;40: 1293–1300.
Klotz U. Ziconotide—a novel neuron-specific calcium channel blocker for the intrathecal treatment of severe chronic pain—a short review. Int J Clin Pharmacol Ther 2006;44: 478–483.
Wermeling DP. Ziconotide, an intrathecally administered N-type calcium channel antagonist for the treatment of chronic pain. Pharmacotherapy 2005;25: 1084–1094.
Atanassoff PG, Hartmannsgruber MW, Thrasher J, et al. Ziconotide, a new N-type calcium channel blocker, administered intrathecally for acute postoperative pain. Reg Anesth Pain Med 2000;25: 274–278.
Mathur VS. Ziconotide: a new pharmacological class of drug for the management of pain. Sem Anesth Periop Med Pain 2000; 19: 67–75.
Staats PS, Yearwood T, Charapata SG, et al. Intrathecal ziconotide in the treatment of refractory pain in patients with cancer or AIDS: a randomized controlled trial. JAMA 2004;291: 63–70.
Wallace MS, Charapata SG, Fisher R, et al. Intrathecal Ziconotide in the treatment of chronic non-malignant pain: a randomized, double-blind, placebo-controlled clinical trial. Neuromodulation 2006;9: 75–86.
Lyseng-Williamson KA, Perry C. Ziconotide. CNS Drugs 2006; 20: 331–341.
Rauck RL, Wallace MS, Leong MS, et al. A randomized, double-blind, placebo-controlled study of intrathecal ziconotide in adults with severe chronic pain. J Pain Symptom Manage 2006;31: 393–406.
Fisher R, Hassenbusch S, Krames E, et al. A consensus statement regarding the present suggested titration for priait (ziconotide). Neuromodulation 2005;8: 153–154.
Saulino M. Successful reduction of neuropathic pain associated with spinal cord injury via of a combination of intrathecal hydromorphone and ziconotide: a case report. Spinal Cord 2007;45: 749–752.
Staats PS, Yearwood T, Charapata SG, et al. Intrathecal ziconotide in the treatment of refractory pain in patients with cancer or AIDS: a randomized controlled trial. JAMA 2004;291: 63–70.
Snutch TP. Targeting chronic and neuropathic pain: the N-type calcium channel comes of age. NeuroRx 2005;2: 662–670.
Wermeling DP, Berger JR. Ziconotide infusion for severe chronic pain: case series of patients with neuropathic pain. Pharmacotherapy 2006;26: 395–402.
Yarotskyy V, Elmslie KS. Omega-conotoxin GVIA alters gating charge movement of N-type (CaV2.2) calcium channels. J Neurophysiol 2009;101: 332–340.
Thompson JC, Dunbar E, Laye RR. Treatment challenges and complications with ziconotide monotherapy in established pump patients. Pain Physician 2006;9: 147–152.
Vitale V, Battelli D, Gasperoni E, Monachese N. Intrathecal therapy with ziconotide: clinical experience and considerations on its use. Minerva Anestesiol 2008;74: 727–733.
Deer T, Krames ES, Hassenbusch SJ, et al. Polyanalgesic Consensus Conference 2007: recommendations for the management of pain by intrathecal (intraspinal) drug delivery: report of an interdisciplinary expert panel. Neuromodulation 2007;10: 300–328.
Smith HS, Deer TR, Staats PS, Singh V, Sehgal N, Cordner H. Intrathecal drug delivery. Pain Physician 2008;11: S89-S104.
Pin JP, Bockaert J. Omega-conotoxin GVIA and dihydropyridines discriminate two types of Ca2+ channels involved in GABA release from striatal neurons in culture. Eur J Pharmacol 1990;188: 81–84.
Zhang SP, Kauffman J, Yagel SK, Codd EE. High-throughput screening for N-type calcium channel blockers using a scintillation proximity assay. J Biomol Screen 2006;11: 672–677.
Scott DA, Wright CE, Angus JA. Actions of intrathecal omega-conotoxins CVID, GVIA, MVIIA, and morphine in acute and neuropathic pain in the rat. Eur J Pharmacol 2002;451: 279–286.
Malmberg AB, Yaksh TL. Effect of continuous intrathecal infusion of omega-conopeptides, N-type calcium-channel blockers, on behavior and antinociception in the formalin and hot-plate tests in rats. Pain 1995;60: 83–90.
Cousins M, Goucke R, Cher L, Brooker C. A phase I clinical trial of AM336, a novel N-type calcium channel blocker. IASP Press: 10th World Congress on Pain 2002;200: 615-P249.
Knutsen LJ, Hobbs CJ, Earnshaw CG, et al. Synthesis and SAR of novel 2-arylthiazolidinones as selective analgesic N-type calcium channel blockers. Bioorg Med Chem Lett 2007;17: 662–667.
Herlitze S, Garcia DE, Mackie K, Hille B, Scheuer T, Catterall WA. Modulation of Ca2+ channels by G-protein beta gamma subunits. Nature 1996;380: 258–262.
Pirec V, Laurito CE, Lu Y, Yeomans DC. The combined effects of N-type calcium channel blockers and morphine on A delta versus C fiber mediated nociception. Anesth Analg 2001;92: 239–243.
Martin TJ, Eisenach JC. Pharmacology of opioid and nonopioid analgesics in chronic pain states. J Pharmacol Exp Ther 2001; 299: 811–817.
Sharpe IA, Gehrmann J, Loughnan ML, et al. Two new classes of conopeptides inhibit the alphal-adrenoceptor and noradrenaline transporter. Nat Neurosci 2001;4: 902–907.
Paczkowski FA, Sharpe IA, Dutertre S, Lewis RJ. chi-Conotoxin and tricyclic antidepressant interactions at the norepinephrine transporter define a new transporter model. J Biol Chem 2007; 282: 17837–17844.
Nelson MT, Joksovic PM, Perez-Reyes E, Todorovic SM. The endogenous redox agent L-cysteine induces T-type Ca2+ channel-dependent sensitization of a novel subpopulation of rat peripheral nociceptors. J Neurosci 2005;25: 8766–8775.
Todorovic SM, Jevtovic-Todorovic V. The role of T-type calcium channels in peripheral and central pain processing. CNS Neurol Disord Drug Targets 2006;5: 639–653.
Sekizawa SI, French AS, Torkkeli PH. Low-voltage-activated calcium current does not regulate the firing behavior in paired mechanosensory neurons with different adaptation properties. J Neurophysiol 2000;83: 746–753.
Jagodic MM, Pathirathna S, Nelson MT, et al. Cell-specific alterations of T-type calcium current in painful diabetic neuropathy enhance excitability of sensory neurons. J Neurosci 2007;27: 3305–3316.
Jagodic MM, Pathirathna S, Joksovic PM, et al. Upregulation of the T-type calcium current in small rat sensory neurons after chronic constrictive injury of the sciatic nerve. J Neurophysiol 2008;99: 3151–3156.
Dogrul A, Gardell LR, Ossipov MH, Tulunay FC, Lai J, Porreca F. Reversal of experimental neuropathic pain by T-type calcium channel blockers. Pain 2003;105: 159–168.
Flatters SJ, Bennett GJ. Ethosuximide reverses paclitaxel- and vincristine-induced painful peripheral neuropathy. Pain 2004;109: 150–161.
Todorovic SM, Meyenburg A, Jevtovic-Todorovic V. Redox modulation of peripheral T-type Ca2+ channels in vivo: alteration of nerve injury-induced thermal hyperalgesia. Pain 2004; 109: 328–339.
Todorovic SM, Meyenburg A, Jevtovic-Todorovic V. Mechanical and thermal antinociception in rats following systemic administration of mibefradil, a T-type calcium channel blocker. Brain Res 2002;951: 336–340.
Choi S, Na HS, Kim J, et al. Attenuated pain responses in mice lacking Ca(V)3.2 T-type channels. Genes Brain Behav 2007;6: 425–431.
Bourinet E, Alloui A, Monteil A, et al. Silencing of the Cav3.2 T-type calcium channel gene in sensory neurons demonstrates its major role in nociception. Embo J 2005;24: 315–324.
Shin HS, Cheong EJ, Choi S, Lee J, Na HS. T-type Ca2+ channels as therapeutic targets in the nervous system. Curr Opin Pharmacol 2008;8: 33–41.
Todorovic SM, Jevtovic-Todorovic V, Meyenburg A, et al. Redox modulation of T-type calcium channels in rat peripheral nociceptors. Neuron 2001;31: 75–85.
Todorovic SM, Jevtovic-Todorovic V. Regulation of T-type calcium channels in the peripheral pain pathway. Channels (Austin) 2007;1: 238–245.
Dworkin RH, O’Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain 2007;132: 237–251.
Raskin P, Donofrio PD, Rosenthal NR, et al. Topiramate vs placebo in painful diabetic neuropathy: analgesic and metabolic effects. Neurology 2004;63: 865–873.
Thienel U, Neto W, Schwabe SK, Vijapurkar U. Topiramate in painful diabetic polyneuropathy: findings from three double-blind placebo-controlled trials. Acta Neurol Scand 2004;110: 221–231.
Khoromi S, Patsalides A, Parada S, Salehi V, Meegan JM, Max MB. Topiramate in chronic lumbar radicular pain. J Pain 2005; 6: 829–836.
Fowler JA, Shen JY, Bettinger TL. Successful use of topiramate in a patient with severe postherpetic neuralgia. Ann Pharmacother 2009;43: 139–142.
Chong MS, Libretto SE. The rationale and use of topiramate for treating neuropathic pain. Clin J Pain 2003;19: 59–68.
Ophoff RA, Terwindt GM, Vergouwe MN, et al. Familial hemiplegic migraine and episodic ataxia type-2 are caused by mutations in the Ca2+ channel gene CACNL1A4. Cell 1996;87: 543–552.
Pietrobon D, Striessnig J. Neurobiology of migraine. Nat Rev Neurosci 2003;4: 386–398.
Pietrobon D. Familial hemiplegic migraine. Neurotherapeutics 2007;4: 274–284.
Murakami M, Nakagawasai O, Suzuki T, et al. Antinociceptive effect of different types of calcium channel inhibitors and the distribution of various calcium channel alpha 1 subunits in the dorsal horn of spinal cord in mice. Brain Res 2004;1024: 122–129.
Luo ZD, Chaplan SR, Higuera ES, et al. Upregulation of dorsal root ganglion (alpha)2(delta) calcium channel subunit and its correlation with allodynia in spinal nerve-injured rats. J Neurosci 2001;21: 1868–1875.
Luo ZD, Calcutt NA, Higuera ES, et al. Injury type-specific calcium channel alpha 2 delta-1 subunit up-regulation in rat neuropathic pain models correlates with antiallodynic effects of gabapentin. J Pharmacol Exp Ther 2002;303: 1199–1205.
Newton RA, Bingham S, Case PC, Sanger GJ, Lawson SN. Dorsal root ganglion neurons show increased expression of the calcium channel alpha2delta-l subunit following partial sciatic nerve injury. Brain Res Mol Brain Res 2001;95: 1–8.
Wang H, Sun H, Della Penna K, et al. Chronic neuropathic pain is accompanied by global changes in gene expression and shares pathobiology with neurodegenerative diseases. Neuroscience 2002;114: 529–546.
Valder CR, Liu JJ, Song YH, Luo ZD. Coupling gene chip analyses and rat genetic variances in identifying potential target genes that may contribute to neuropathic allodynia development. J Neurochem 2003;87: 560–573.
Li CY, Song YH, Higuera ES, Luo ZD. Spinal dorsal horn calcium channel alpha2delta-1 subunit upregulation contributes to peripheral nerve injury-induced tactile allodynia. J Neurosci 2004;24: 8494–8499.
Gee NS, Brown JP, Dissanayake VU, Offord J, Thurlow R, Woodruff GN. The novel anticonvulsant drug, gabapentin (Neurontin), binds to the alpha2delta subunit of a calcium channel. J Biol Chem 1996;271: 5768–5776.
Hwang JH, Yaksh TL. Effect of subarachnoid gabapentin on tactile-evoked allodynia in a surgically induced neuropathic pain model in the rat. Regional Anesthesia 1997;22: 249–256.
Abdi S, Lee DH, Chung JM. The anti-allodynic effects of amitriptyline, gabapentin, and lidocaine in a rat model of neuropathic pain. Anesth Analg 1998;87: 1360–1366.
Field MJ, McCleary S, Hughes J, Singh L. Gabapentin and pregabalin, but not morphine and amitriptyline, block both static and dynamic components of mechanical allodynia induced by streptozocin in the rat. Pain 1999;80: 391–398.
Backonja M, Beydoun A, Edwards KR, et al. Gabapentin for the symptomatic treatment of painful neuropathy in patients with diabetes mellitus: a randomized controlled trial. JAMA 1998;280: 1831–1836.
Rosner H, Rubin L, Kestenbaum A. Gabapentin adjunctive therapy in neuropathic pain states. Clin J Pain 1996;12: 56–58.
Rosenberg JM, Harrell C, Ristic H, Werner RA, de Rosayro AM. The effect of gabapentin on neuropathic pain. Clin J Pain 1997; 13: 251–255.
Nudler S, Piriz J, Urbano FJ, Rosato-Siri MD, Renteria ES, Uchitel OD. Ca2+ channels and synaptic transmission at the adult, neonatal, and P/Q-type deficient neuromuscular junction. Ann N Y Acad Sci 2003;998: 11–17.
Perrier JF, Alaburda A, Hounsgaard J. Spinal plasticity mediated by postsynaptic L-type Ca2+ channels. Brain Res Brain Res Rev 2002;40: 223–229.
Westenbroek RE, Hoskins L, Catterall WA. Localization of Ca2+ channel subtypes on rat spinal motor neurons, interneurons, and nerve terminals. J Neurosci 1998;18: 6319–6330.
Kato A, Ohkubo T, Kitamura K. Algogen-specific pain processing in mouse spinal cord: differential involvement of voltage-dependent Ca(2+) channels in synaptic transmission. Br J Pharmacol 2002;135: 1336–1342.
Reid CA, Bekkers JM, Clements JD. Presynaptic Ca2+ channels: a functional patchwork. Trends Neurosci 2003;26: 683–687.
Talley EM, Cribbs LL, Lee JH, Daud A, Perez-Reyes E, Bayliss DA. Differential distribution of three members of a gene family encoding low voltage-activated (T-type) calcium channels. J Neurosci 1999;19: 1895–1911.
Lambert RC, McKenna F, Maulet Y, et al. Low-voltage-activated Ca2+ currents are generated by members of the CavT subunit family (alpha1G/H) in rat primary sensory neurons. J Neurosci 1998;18: 8605–8613.
McCallum JB, Kwok WM, Mynlieff M, Bosnjak ZJ, Hogan QH. Loss of T-type calcium current in sensory neurons of rats with neuropathic pain. Anesthesiology 2003;98: 209–216.
Boroujerdi A, Kim HK, Lyu YS, et al. Injury discharges regulate calcium channel alpha-2-delta-1 subunit upregulation in the dorsal horn that contributes to initiation of neuropathic pain. Pain 2008.
Zareba G. New treatment options in the management of fibromyalgia: role of pregabalin. Neuropsychiatr Dis Treat 2008;4: 1193–1201.
Stahl SM. Anticonvulsants and the relief of chronic pain: pregabalin and gabapentin as alpha(2)delta ligands at voltage-gated calcium channels. J Clin Psychiatry 2004;65: 596–597.
Rowbotham M, Harden N, Stacey B, Bernstein P, Magnus-Miller L. Gabapentin for the treatment of postherpetic neuralgia: a randomized controlled trial. JAMA 1998;280: 1837–1842.
Rice AS, Maton S. Gabapentin in postherpetic neuralgia: a randomised, double blind, placebo controlled study. Pain 2001;94: 215–224.
Anhut H, Ashman P, Feuerstein TJ, Sauermann W, Saunders M, Schmidt B. Gabapentin (Neurontin) as add-on therapy in patients with partial seizures: a double-blind, placebo-controlled study. The International Gabapentin Study Group. Epilepsia 1994;35: 795–801.
Garcia-Borreguero D, Larrosa O, de la Llave Y, Verger K, Masramon X, Hernandez G. Treatment of restless legs syndrome with gabapentin: a double-blind, cross-over study. Neurology 2002;59: 1573–1579.
Pollack MH, Matthews J, Scott EL. Gabapentin as a potential treatment for anxiety disorders. Am J Psychiatry 1998;155: 992–993.
Bockbrader H. Clinical pharmacokinetics of gabapentin. Drugs Today 1995;31: 613–619.
McLean MJ. Clinical pharmacokinetics of gabapentin. Neurology 1994;44: S17–22; discussion S31-12.
Stewart BH, Kugler AR, Thompson PR, Bockbrader HN. A saturable transport mechanism in the intestinal absorption of gabapentin is the underlying cause of the lack of proportionality between increasing dose and drug levels in plasma. Pharm Res 1993;10: 276–281.
Nicholson B. Gabapentin use in neuropathic pain syndromes. Acta Neurol Scand 2000;101: 359–371.
Hemstreet B, Lapointe M. Evidence for the use of gabapentin in the treatment of diabetic peripheral neuropathy. Clin Ther 2001; 23: 520–531.
Todorov AA, Kolchev CB, Todorov AB. Tiagabine and gabapentin for the management of chronic pain. Clin J Pain 2005;21: 358–361.
Arnold LM, Goldenberg DL, Stanford SB, et al. Gabapentin in the treatment of fibromyalgia: a randomized, double-blind, placebo-controlled, multicenter trial. Arthritis Rheum 2007;56: 1336–1344.
Matthews EA, Dickenson AH. A combination of gabapentin and morphine mediates enhanced inhibitory effects on dorsal horn neuronal responses in a rat model of neuropathy. Anesthesiology 2002;96: 633–640.
Gilron I, Bailey JM, Tu D, Holden RR, Weaver DF, Houlden RL. Morphine, gabapentin, or their combination for neuropathic pain. N Engl J Med 2005;352: 1324–1334.
Hanna M, O’Brien C, Wilson MC. Prolonged-release oxycodone enhances the effects of existing gabapentin therapy in painful diabetic neuropathy patients. Eur J Pain 2008;12: 804–813.
Gu R, Gusler GM, Hou SYE, et al. Extended release and dose-proportionality of a novel gabapentin formulation. The 88th Annual Meeting of the Endocrine Society; 2006:319 (abstract).
Gordi T, Hou E, Kasichayanula S, Berner B. Pharmacokinetics of gabapentin after a single day and at steady state following the administration of gastric-retentive-extended-release and immediate-release tablets: a randomized, open-label, multiple-dose, three-way crossover, exploratory study in healthy subjects. Clin Ther 2008;30: 909–916.
Sabatowski R, Gálvez R, Cherry DA, et al. Pregabalin reduces pain and improves sleep and mood disturbances in patients with post-herpetic neuralgia: results of a randomised, placebo-controlled clinical trial. Pain 2004;109: 26–35.
Irving G, Jensen M, Cramer M, et al. Efficacy and tolerability of gastric-retentive gabapentin for the treatment of postherpetic neuralgia: results of a double-blind, randomized, placebo-controlled clinical trial. Clin J Pain 2009;25: 185–192.
Cundy KC, Sastry S, Luo W, Zou J, Moors TL, Canafax DM. Clinical pharmacokinetics of XP13512, a novel transported prodrug of gabapentin. J Clin Pharmacol 2008;48: 1378–1388.
Kushida CA, Becker PM, Ellenbogen AL, Canafax DM, Barrett RW. Randomized, double-blind, placebo-controlled study of XP13512/GSK1838262 in patients with RLS. Neurology 2009; 72: 439–446.
Gajraj NM. Pregabalin: its pharmacology and use in pain management. Anesth Analg 2007;105: 1805–1815.
Dworkin RH, Kirkpatrick P. Pregabalin. Nat Rev Drug Discov 2005;4: 455–456.
Frampton JE, Foster RH. Pregabalin: in the treatment of postherpetic neuralgia. Drugs 2005;65: 111–120.
Gray P. Pregabalin in the management of central neuropathic pain. Expert Opin Pharmacother 2007;8: 3035–3041.
Zareba G. Pregabalin: a new agent for the treatment of neuropathic pain. Drugs Today 2007;41: 509–516.
Owen RT. Pregabalin: its efficacy, safety and tolerability profile in fibromyalgia syndrome. Drugs Today (Barc) 2007;43: 857–863.
Stacey BR, Swift JN. Pregabalin for neuropathic pain based on recent clinical trials. Curr Pain Headache Rep 2006;10: 179–184.
Crofford LJ, Rowbotham MC, Mease PJ, et al. Pregabalin for the treatment of fibromyalgia syndrome: results of a randomized, double-blind, placebo-controlled trial. Arthritis Rheum 2005;52: 1264–1273.
Calandre EP, Morillas-Arques P, Rodriguez-Lopez CM, Rico-Villademoros F, Hidalgo J. Pregabalin augmentation of quetiapine therapy in the treatment of fibromyalgia: an open-label, prospective trial. Pharmacopsychiatry 2007;40: 68–71.
Mease PJ, Russell IJ, Arnold LM, et al. A randomized, double-blind, placebo-controlled, phase III trial of pregabalin in the treatment of patients with fibromyalgia. J Rheumatol 2008;35: 502–514.
Arnold LM, Russell IJ, Diri EW, et al. A 14-week, randomized, double-blinded, placebo-controlled monotherapy trial of pregabalin in patients with fibromyalgia. J Pain 2008;9: 792–805.
Crofford LJ, Mease PJ, Simpson SL, et al. Fibromyalgia relapse evaluation and efficacy for durability of meaningful relief (FREEDOM): a 6-month, double-blind, placebo-controlled trial with pregabalin. Pain 2008;136: 419–431.
Recla JM, Sarantopoulos CD. Combined use of pregabalin and memantine in fibromyalgia syndrome treatment: a novel analgesic and neuroprotective strategy? Med Hypotheses 2009;73: 177–189.
De Lange FP, Knoop H, Bleijenberg G, Van der Meer JW, Hagoort P, Toni I. Gray matter volume reduction in the chronic fatigue syndrome. Neuroimage 2005;26: 777–781.
Kuchinad A, Schweinhardt P, Seminowicz DA, Wood PB, Chizh BA, Bushnell MC. Accelerated brain gray matter loss in fibromyalgia patients: premature aging of the brain? J Neurosci 2007; 27: 4004–4007.
Okada T, Tanaka M, Kuratsune H, Watanabe Y, Sadato N. Mechanisms underlying fatigue: a voxel-based morphometric study of chronic fatigue syndrome. BMC Neurol 2004;4: 14.
Schmidt-Wilcke T, Luerding R, Weigand T, et al. Striatal grey matter increase in patients suffering from fibromyalgia--a voxel-based morphometry study. Pain 2007;132(suppl 1): S109–116.
Schmidt-Wilcke T, Leinisch E, Straube A, et al. Gray matter decrease in patients with chronic tension type headache. Neurology 2005;65: 1483–1486.
Villarreal G, Hamilton DA, Petropoulos H, et al. Reduced hippocampal volume and total white matter volume in posttraumatic stress disorder. Biol Psychiatry 2002;52: 119–125.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Perret, D., Luo, Z.D. Targeting voltage-gated calcium channels for neuropathic pain management. Neurotherapeutics 6, 679–692 (2009). https://doi.org/10.1016/j.nurt.2009.07.006
Issue Date:
DOI: https://doi.org/10.1016/j.nurt.2009.07.006