Abstract
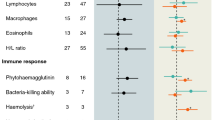
Studies of immunity in bat species are rare. However, it is important to determine immunological variations to identify factors influencing the health status of these endangered mammals from an evolutionary, ecological, conservation, and public health point of view. Immunity is highly variable and can be influenced by both internal (e.g. hormone levels, energy demand) and external factors (e.g. pathogens, climate). As bats have some peculiar ecological, energetic, and putative immunological characteristics, they are outstanding study organisms for ecoimmunological studies. We tested if (i) female bats have a higher immunity than males similar to most other mammalian species and (ii) individuals differ according to their energy demand (e.g. reproductive status). To study these questions, we sampled female and male Myotis daubentonii with different reproductive states and estimated their bacterial killing activity, hemolysis/hemagglutination titer, immunoglobulin G (IgG) concentration, and total and differential white blood cell counts. These methods characterize the cellular and humoral branches of both the adaptive and the innate immune responses of these individuals. Reproductively active males had lower cellular immunity compared to non-reproductive individuals. Pregnant females had increased IgG concentrations while hemolysis was enhanced during lactation. No clear trade-off between immunity and reproduction was found; instead immunity of males and female bats seems to be modulated differently due to varying hormonal and energetic states. Our data suggest that both adaptive and innate immunity as well as individual differences (i.e. sex and reproductive state) need to be considered to get a comprehensive overall picture of immunity in wild mammals.
Article PDF
Similar content being viewed by others
References
Adamo, S.A., 2004. How should behavioural ecologists interpret measurements of immunity? Anim. Behav. 68, 1443–1449.
Al-Afaleq, A., Homeida, A., 1998. Effects of low doses of oestradiol, testosterone and dihydrotestosterone on the immune response of broiler chicks. Immunopharmacol. Immunotoxicol. 20, 315–327.
Alexander, J., Stimson, W.H., 1988. Sex hormones and the course of parasitic infection. Parasitol. Today 4, 189–193.
Allen, L.C., Turmelle, A.S., Mendonça, M.T., Navara, K.J., Kunz, T.H., McCracken, G.F., 2009. Roosting ecology and variation in adaptive and innate immune system function in the Brazilian free-tailed bat (Tadarida brasiliensis). J. Comp. Physiol. B 179, 315–323.
Anthony, E.L., 1988. Age determination in bats. In: Kunz, T.H. (Ed.), Ecological and behavioral methods for the study of bats. Smithsonian Institution Press, Washington D.C., pp. 1–28.
Anthony, E.L.P., Kunz, T.H., 1977. Feeding strategies of the little brown bat, Myotis lucifugus, in southern New Hampshire. Ecology 58, 775–786.
Anthony, E.L.P., Stack, M.H., Kunz, T.H., 1981. Night roosting and the nocturnal time budget of the little brown bat, Myotis lucifugus: effects of reproductive status, prey density, and environmental conditions. Oecologia 51, 151–156.
Apanius, V., 1998. Stress and immune defense. Adv. Study Behav. 27, 133–153.
Bain, B.J., England, J.M., 1975. Normal haematological values: sex difference in neutrophil count. Br. Med. J. 1, 306–309.
Barclay, R.M.R., 1991. Population atructure of temperate zone insectivorous bats in relation to foraging behaviour and energy demand. J. Anim. Ecol. 60, 165–178.
Barclay, R.M.R., Harder, L.D., 2003. Life histories of bats: life in the slow lane. In: Kunz, T.H., Fenton, M.B. (Eds.), Bat Ecology. The University of Chicago Press, Chicago, pp. 209–253.
Becker, D.J., Chumchal, M.M., Bentz, A.B., Platt, S.G., Czirják, G.Á., Rainwater, T.R., Altizer, S., Streicker, D.G., 2017. Predictors and immunological correlates of sublethal mercury exposure in vampire bats. R. Soc. Open Sci. 4, 170073.
Becker, D.J., Czirják, G.Á., Volokhov, D.V., Bentz, A.B., Carrera, J.E., Camus, M.S., Navara, K.J., Chizhikov, V.E., Brock Fenton, M., Simmons, N.B., Recuenco, S.E., Gilbert, A.T., Altizer, S., Streicker, D.G., 2018. Livestock abundance predicts vampire bat demography, immune profiles and bacterial infection risk. Philos. Trans. R. Soc. Lond. B: Biol. Sci. 373, 20170089.
Becker, N.I., Encarnação, J.A., Tschapka, M., Kalko, E.K.V., 2013a. Energetics and life-history of bats in comparison to small mammals. Ecol. Res. 28, 249–258.
Becker, N.I., Tschapka, M., Kalko, E.K.V., Encarnação, J.A, 2013b. Balancing the energy budget in free-ranging male Myotis daubentonii bats. Physiol. Biochem. Zool. 86, 361–369.
Boonstra, R., McColl, C.J., Karels, T.J., 2001. Reproduction at all costs: the adaptive stress response of male arctic ground squirrels. Ecology 82, 1930–1946.
Borghesi, J., Mario, L.C., Nogueira, M.R., Favaron, P.O., Miglino, M.A., 2014. Immunoglobulin transport during gestation in domestic animals and humans—a review. Open J. Anim. Sci. 4, 323.
Bouma, H.R., Carey, H.V., Kroese, F.G.M., 2010. Hibernation: the immune system at rest? J. Leukoc. Biol. 88, 619–624.
Bouman, A., Heineman, M.J., Faas, M.M., 2005. Sex hormones and the immune response in humans. Hum. Reprod. Update 11, 411–423.
Buchanan, K., Evans, M., Goldsmith, A., 2003. Testosterone, dominance signalling and immunosuppression in the house sparrow, Passer domesticus. Behav. Ecol. Sociobiol. 55, 50–59.
Butterworth, M., McClellan, B., Aklansmith, M., 1967. Influence of sex on immunoglobulin levels. Nature 214, 1224–1225.
Christe, P., Giorgi, M.S., Vogel, P., Arlettaz, R., 2000. Variation in intensity of a parasitic mite (Spinturnix myoti) in relation to the reproductive cycle and immunocompetence of its bat host (Myotis myotis). Ecol. Lett. 3, 207–212.
Da Silva, J.A.P., 1999. Sex hormones and glucocorticoids: interactions with the immune system. Ann. N.Y.Acad. Sci. 876, 102–118.
Demas, G.E., Greives, T., Chester, E., French, S., 2012. The energetics of immunity. In: Demas, G.E., Nelson, R.J. (Eds.), Ecoimmunology. Oxford University Press, New York, pp. 259–296.
Demas, G.E., Zysling, D.A., Beechler, B.R., Muehlenbein, M.P., French, S.S., 2011. Beyond phytohaemagglutinin: assessing vertebrate immune function across ecological contexts. J. Anim. Ecol. 80, 710–730.
Dempster, G., Grodums, E.I., Spencer, W.A., 1966. Experimental coxsackie B-3 virus infection in Citellus lateralis. J. Cell. Physiol. 67, 443–453.
Dietz, C., Nill, D., von Helversen, O., 2009. Bats of Britain, Europe and Northwest Africa. A & C Black, London.
Drazen, D.L., Trasy, A., Nelson, R.J., 2003. Photoperiod differentially affects energetics of immunity in pregnant and lactating Siberian hamsters (Phodopus sungorus). Can. J. Zool. 81, 1406–1413.
Drexler, J.F., Corman, V.M., Wegner, T., Tateno, A.F., Zerbinati, R.M., Gloza-Rausch, F., Seebens, A., Müller, M.A., Drosten, C., 2011. Amplification of emerging viruses in a bat colony. Emerg. Infect. Dis. 17, 449–456.
Duffy, D.L., Bentley, G.E., Drazen, D.L., Ball, G.F., 2000. Effects of testosterone on cell-mediated and humoral immunity in non-breeding adult European starlings. Behav. Ecol. 11, 654–662.
Ellison, P.T., 2003. Energetics and reproductive effort. Am. J. Hum. Biol. 15, 342–351.
Encarnação, J.A, 2012. Spatiotemporal pattern of local sexual segregation in a tree-dwelling temperate bat Myotis daubentonii. J. Ethol. 30, 271–278.
Encarnação, J.A, Dietz, M., 2006. Estimation of food intake and ingested energy in Daubenton’s bats (Myotis daubentonii) during pregnancy and spermatogenesis. Eur. J. Wildl. Res. 52, 221–227.
Encarnação, J.A, Dietz, M., Kierdorf, U., 2004. Reproductive condition and activity pattern of male Daubenton’s bats (Myotis daubentonii) in the summer habitat. Mamm. Biol. 69, 163–172.
Epstein, J.H., Baker, M.L., Zambrana-Torrelio, C., Middleton, D., Barr, J.A., DuBovi, E., Boyd, V., Pope, B., Todd, S., Crameri, G., 2013. Duration of maternal antibodies against canine distemper virus and Hendra virus in pteropid bats. PLoS One 8, e67584.
Fokidis, B.H., Greiner, E.C., Deviche, P., 2008. Interspecific variation in avian blood parasites and haematology associated with urbanization in a desert habitat. J. Avian Biol. 39, 300–310.
Folstad, I., Karter, A.J., 1992. Parasites, bright males, and the immunocompetence handicap. Am. Nat. 139, 603–622.
French, S.S., DeNardo, D.F., Moore, M.C., 2007. Trade-offs between the reproductive and immune systems: facultative responses to resources or obligate responses to reproduction? Am. Nat. 170, 79–89.
French, S.S., Neuman-Lee, L.A., 2012. Improved ex vivo method for microbiocidal activity across vertebrate species. Biol. Open 1, 482–487.
Geiser, F., 2004. Metabolic rate and body temperature reduction during hibernation and daily torpor. Annu. Rev. Physiol. 66, 239–274.
George, D.B., Webb, C.T., Farnsworth, M.L., O’Shea, T.J., Bowen, R.A., Smith, D.L., Stanley, T.R., Ellison, L.E., Rupprecht, C.E., 2011. Host and viral ecology determine bat rabies seasonality and maintenance. Proc. Natl. Acad. Sci. U. S. A. 108, 10208–10213.
Gittleman, J.L., Thompson, S.D., 1988. Energy allocation in mammalian reproduction. Integr. Comp. Biol. 28, 863–875.
Gloza-Rausch, F., Ipsen, A., Seebens, A., Göttsche, M., Panning, M., Drexler, J.F., Petersen, N., Annan, A., Grywna, K., Müller, M., 2008. Detection and prevalence patterns of group I coronaviruses in bats, northern Germany. Emerg. Infect. Dis. 14, 626–631.
Greiner, S., Stefanski, V., Dehnhard, M., Voigt, C.C., 2010. Plasma testosterone levels decrease after activation of skin immune system in a free-ranging mammal. Gen. Comp. Endocrinol. 168, 466–473.
Grindstaff, J.L., 2008. Maternal antibodies reduce costs of an immune response during development. J. Exp. Biol. 211, 654–660.
Hammond, K.A., Diamond, J., 1997. Maximal sustained energy budgets in humans and animals. Nature 386, 457–462.
Hayman, D., Bowen, R., Cryan, P., McCracken, G.F., O’shea, T., Peel, A.J., Gilbert, A., Webb, C., Wood, J., 2013. Ecology of zoonotic infectious diseases in bats: current knowledge and future directions. Zoonoses Public Health 60, 2–21.
Heinrich, S.K., Hofer, H., Courtiol, A., Melzheimer, J., Dehnhard, M., Czirják, G.Á., Wachter, B., 2017. Cheetahs have a stronger constitutive innate immunity than leopards. Sci. Rep. 7, 44837.
Heinrich, S.K., Wachter, B., Aschenborn, O.H.K., Thalwitzer, S., Melzheimer, J., Hofer, H., Czirják, G.Á., 2016. Feliform carnivoes have a distinguished constitutive innate immune response. Biol. Open 5, 550–555.
Hsieh, M.M., Everhart, J.E., Byrd-Holt, D.D., Tisdale, J.F., Rodgers, G.P., 2007. Prevalence of neutropenia in the U.S. population: age, sex, smoking status, and ethnic differences. Ann. Intern. Med. 146, 486–492.
Klein, S.L., 2000. The effects of hormones on sex differences in infection: from genes to behavior. Neurosci. Biobehav. Rev. 24, 627–638.
Krutzsch, P.H., 2000. Anatomy, physiology and cyclicity of the male reproductive tract. In: Crichton, E.G., Krutzsch, P.H. (Eds.), Reproductive Biology of Bats. Academic Press, London, pp. 92–137.
Kunz, T.H., Pierson, E.D., 1994. Bats of the world: an introduction. In: Nowak, R.M. (Ed.), Bats of the World. John Hopkins University, Baltimore, pp. 1–46.
Kunz, T.H., Wrazen, J.A., Burnett, C.D., 1998. Changes in body mass and fat reserves in pre-hibernating little brown bats (Myotis lucifugus). Ecoscience 5, 8–17.
Lee, K.A., 2006. Linking immune defenses and life history at the levels of the individual and the species. Integr. Comp. Biol. 46, 1000–1015.
Lemke, H., Hansen, H., Lange, H., 2003. Non-genetic inheritable potential of maternal antibodies. Vaccine 21, 3428–3431.
Lochmiller, R.L., Deerenberg, C., 2000. Trade-offs in evolutionary immunology: just what is the cost of immunity? Oikos 88, 87–98.
Love, O.P., Salvante, K.G., Dale, J., Williams, T.D., 2008. Sex-specific variability in the immune system across life-history stages. Am. Nat. 172, E99–112.
Lucan, R.K., Hanák, V., Horáček, I., 2009. Long-term re-use of tree roosts by European forest bats. For. Ecol. Manag. 258, 1301–1306.
Martin, L., Bernhard, R.T.F., 2000. Endocrine regulation of reproduction in bats: the role of circulating gonadal hormones. In: Crichton, E.G., Krutzsch, P.H. (Eds.), Reproductive Biology of Bats. Academic Press, San Diego, pp. 27–64.
Martin, L.B., Weil, Z.M., Nelson, R.J., 2008. Seasonal changes in vertebrate immune activity: mediation by physiological trade-offs. Philos. Trans. R. Soc. Lond. B: Biol. Sci. 363, 321–339.
Matson, K.D., Cohen, A.A., Klasing, K.C., Ricklefs, R.E., Scheuerlein, A., 2006. No simple answers for ecological immunology: relationships among immune indices at the individual level break down at the species level in waterfowl. Proc. R. Soc. Lond. B: Biol. Sci. 273, 815–822.
Matson, K.D., Ricklefs, R.E., Klasing, K.C., 2005. A hemolysis-hemagglutination assay for characterizing constitutive innate humoral immunity in wild and domestic birds. Dev. Comp. Immunol. 29, 275–286.
Millet, S., Bennett, J., Lee, K.A., Hau, M., Klasing, K.C., 2007. Quantifying and comparing constitutive immunity across avian species. Dev. Comp. Immunol. 31, 188–201.
Moore, I.T., Hopkins, W.A., 2009. Interactions and trade-offs among physiological determinants of performance and reproductive success. Integr. Comp. Biol. 49, 441–451.
Moore, M.S., Reichard, J.D., Murtha, T.D., Zahedi, B., Fallier, R.M., Kunz, T.K., 2011. Specific alterations in complement protein activity of little brown myotis (Myotis lucifugus) hibernating in white-nose syndrome affected sites. PLoS One 6, e27430.
Mühldorfer, K., Speck, S., Kurth, A., Lesnik, R., Freuling, C., Müller, T., Kramer-Schadt, S., Wibbelt, G., 2011. Diseases and causes of death in European bats: dynamics in disease susceptibility and infection rates. PLoS One 6, e29773.
Nelson, R.J., Demas, G.E., Klein, S.L., Kriegsfeld, L.J., 2002. Seasonal patterns of stress, immune function, and disease. Cambridge University Press, New York.
Norris, K., Evans, M.R., 2000. Ecological immunology: life history trade-offs and immune defense in birds. Behav. Ecol. 11, 19–26.
O’Shea, T.J., Cryan, P.M., Cunningham, A.A., Fooks, A.R., Hayman, D.T.S., Luis, A.D., Peel, A.J., Plowright, R.K., Wood, J.L.N., 2014. Bat flight and zoonotic viruses. Emerg. Infect. Dis. 20, 741.
Ochsenbein, A.F., Fehr, T., Lutz, C., Suter, M., Brombacher, F., Hengartner, H., Zinkernagel, R.M., 1999. Control of early viral and bacterial distribution and disease by natural antibodies. Science 286, 2156–2159.
Otto, M.S., Becker, N.I., Encarnação, J.A., 2013. Cool gleaners: thermoregulation in sympatric bat species. Mamm. Biol. 78, 212–215.
Pap, P.L., Czirják, G.Á., Vágási, C.I., Barta, Z., Hasselquist, D., 2010. Sexual dimorphism in immune function changes during the annual cycle in house sparrows. Naturwissenschaften 97, 891–901.
Plowright, R.K., Field, H.E., Smith, C., Divljan, A., Palmer, C., Tabor, G., Daszak, P., Foley, J.E., 2008. Reproduction and nutritional stress are risk factors for Hendra virus infection in little red flying foxes (Pteropus scapulatus). Proc. R. Soc. Lond. B: Biol. Sci. 275, 861–869.
Prendergast, B.J., Freeman, D.A, Zucker, I., Nelson, R.J., 2002. Periodic arousal from hibernation is necessary for initiation of immune responses in ground squirrels. Am. J. Physiol. Regul. Integr. Comp. Physiol. 282, R1054–R1062.
Racey, P.A., 2009. Reproductive assessment of bats. In: Kunz, T.H., Parsons, S. (Eds.), Ecological and Behavioral Methods for the Study of Bats. The John Hopkins University Press, Baltimore, MD.
Roberts, M.L., Buchanan, K.L., Evans, M., 2004. Testing the immunocompetence handicap hypothesis: a review of the evidence. Anim. Behav. 68, 227–239.
Schneeberger, K., Czirják, G.Á., Voigt, C.C., 2013a. Inflammatory challenge increases measures of oxidative stress in a free-ranging, long-lived mammal. J. Exp. Biol. 216, 4514–4519.
Schneeberger, K., Czirják, G.Á., Voigt, C.C., 2013b. Measures of the constitutive immune system are linked to diet and roosting habits of neotropical bats. PLoS One 8, e54023.
Schneeberger, K., Courtiol, A., Czirják, G.Á., Voigt, C.C., 2014. Immune profile predicts survival and reflects senescence in a small, long-lived mammal, the greater sac-winged bat (Saccopteryx bilineata). PLoS One 9, e108268.
Schuurs, A.H.W.M., Verheul, H.A.M., 1990. Effects of gender and sex steroids on the immune response. J. Steroid Biochem. Mol. Biol. 35, 157–172.
Seltmann, A., Czirják, G.Á., Courtiol, A., Bernhard, H., Struebig, M.J., Voigt, C.C., 2017. Habitat disturbance results in chronic stress and impaired health status in forest-dwelling paleotropical bats. Conserv. Physiol. 5, cox020.
Sparkman, A.M., Palacios, M.G., 2009. A test of life-history theories of immune defence in two ecotypes of the garter snake, Thamnophis elegans. J. Anim. Ecol. 78, 1242–1248.
Speakman, J.R., 2008. The physiological costs of reproduction in small mammals. Philos. Trans. R. Soc. Lond. B: Biol. Sci. 363, 375–398.
Strobel, S., Becker, N.I., Encarnação, J.A., 2015. No short-term effect of handling and capture stress on immune responses of bats assessed by bacterial killing assay. Mamm. Biol. 80, 312–315.
Stubbe, M., Ariunbold, J., Buuveibaatar, V., Dorjderem, S., Monkhzul, T., Otgonbaatar, M., Tsogbadrakh, M., Hutson, A.M., Spitzenberger, F., Aulagnier, S., Juste, J., Coroiu, I., Paunovic, M., Karatas, A., 2008. Myotis daubentonii. The IUCN Red List ofThreatened Species 2008 e.T14128A4400742., https://doi.org/10.2305/IUCN.UK.2008.RLTS.T14128A4400742.en, Downloaded on 14 December 2016.
Telfer, S., Bennett, M., Bown, K., Carslake, D., Cavanagh, R., Hazel, S., Jones, T., Begon, M., 2005. Infection with cowpox virus decreases female maturation rates in wild populations of woodland rodents. Oikos 109, 317–322.
Thompson, S.D., 1993. Gestation and lactation in small mammals: basal metabolic rate and the limits of energy use. In: Tomasi, T.E., Horton, T.H. (Eds.), Mammalian Energetics: Interdisciplinary Views of Metabolism and Reproduction. Cornell University, Ithaca, New York, pp. 213–260.
Valdimarsson, H., Mulholland, C., Fridriksdottir, V., Coleman, D., 1983. A longitudinal study of leucocyte blood counts and lymphocyte responses in pregnancy: a marked early increase of monocyte-lymphocyte ratio. Clin. Exp. Immunol. 53, 437.
Zipfel, P.F., 2009. Complement and immune defense: from innate immunity to human diseases. Immunol. Lett. 126, 1–7.
Zuk, M., McKean, K.A., 1996. Sex differences in parasite infections: patterns and processes. Int. J. Parasitol. 26, 1009–1023.
Zuk, M., Stoehr, A.M., 2002. Immune defense and host life history. Am. Nat. 160, 9–22.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ruoss, S., Becker, N.I., Otto, M.S. et al. Effect of sex and reproductive status on the immunity of the temperate bat Myotis daubentonii. Mamm Biol 94, 120–126 (2019). https://doi.org/10.1016/j.mambio.2018.05.010
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.mambio.2018.05.010