Abstract
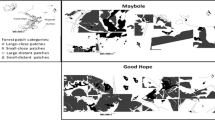
Large herbivores play crucial ecological roles, affecting the structure and function of terrestrial ecosystems. Their effect, however, depend on how they select plants and vegetation patches for foraging. At the landscape scale, succession is one of the processes that should generate vegetation patches with different nutritional quality, affecting selection by herbivores. Earlier successional stages should be preferred as they are dominated by plants with nutritious and palatable leaves. Here, we investigate if the Lowland tapir prefers early compared to late successional forest patches, aiming at contributing to the understanding of the ecological role of the largest terrestrial South American herbivore, and to conservation strategies for this endangered species. We sampled 12 vegetation patches varying in successional stages across a 20.000-ha continuously-forested landscape in the Brazilian Atlantic Forest, recording tapirs through standardized camera trap and track surveys, and quantifying vegetation structure and treefall gaps. Whereas the number of individuals using each patch was not influenced by successional stage, intensity of use was higher in patches in earlier successional stages, in particular patches with higher density of smaller trees and higher cover of treefall gaps. Although inferences on the effects of tapir on plant community depends on future, smaller-scale studies, our results suggest herbivory by tapirs affects forest regeneration, potentially contributing to the maintenance of plant diversity. Results also point out to the potential of mosaics encompassing old-growth and secondary forests for the conservation of the Lowland tapir.
Similar content being viewed by others
References
Agustí, J., Antón, M., 2002. Mammoths, Sabertooths, and Hominids: 65 Million Years of Mammalian Evolution in Europe. Columbia University Press, New York.
Bodmer, R., 1990. Fruit patch size and frugivory in the lowland tapir Tapirus terrestris. J. Zool. 222, 121–128, http://dx.doi.org/10.1111/j.1469-7998.1990.tb04034.x.
Bolker, B.M., Brooks, M.E., Clark, C.J., Geange, S.W., Poulsen, J.R., Stevens, M.H.H., White, J.-S.S., 2009. Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol. Evol. 24, 127–135, http://dx.doi.org/10.1016/j.tree.2008.10.008.
Brasil, 1994. Resoluçao CONAMA no 1, de 31 de janeiro de 1994. http://www.mma.gov.br/port/conama/legislacao/CONAMA RES CONS 1994 001.pdf.
Bryant, J., Kuropat, P., 1980. Selection of winter forage by sub-arctic browsing vertebrates—the role of plant chemistry. Annu. Rev. Ecol. Syst. 11, 261–285, http://dx.doi.org/10.1146/annurev.es.11.110180.001401.
Burnham, K., Anderson, D., 2004. Multimodel inference—understanding AIC and BIC in model selection. Sociol. Methods Res. 33, 261–304, http://dx.doi.org/10.1177/0049124104268644.
Cabrera, J.A., Molina, E., González, T., Armenteras, D., 2016. Does plan B work? Home range estimations from stored on board and transmitted data sets produced by GPS-telemetry in the Colombian Amazon. Rev. Biol. Trop. 64, 1441–1450.
Camargo-Sanabria, A.A., Mendoza, E., 2016. Interactions between terrestrial mammals and the fruits of two neotropical rainforest tree species. Acta Oecol. 73, 45–52, http://dx.doi.org/10.1016/j.actao.2016.02.005.
Canale, G.R., Peres, C.A., Guidorizzi, C.E., Gatto, C.A.F., Kierulff, M.C.M., 2012. Pervasive defaunation of forest remnants in a tropical biodiversity hotspot. PLoS One 7, e41671, http://dx.doi.org/10.1371/journal.pone.0041671.
Cates, R., Orians, G., 1975. Successional status and palatability of plants to generalized herbivores. Ecology 56, 410–418, http://dx.doi.org/10.2307/1934971.
Chandler, R.B., Royle, J.A., King, D.I., 2011. Inference about density and temporary emigration in unmarked populations. Ecology 92, 1429–1435, http://dx.doi.org/10.1890/10-2433.1.
Chazdon, R.L., Peres, C.A., Dent, D., Sheil, D., Lugo, A.E., Lamb, D., Stork, N.E., Miller, S.E., 2009. The potential for species conservation in tropical secondary forests. Conserv. Biol. 23, 1406–1417, http://dx.doi.org/10.1111/j.1523-1739.2009.01338.x.
Clark, D., 1996. Abolishing virginity. J. Trop. Ecol. 12, 735–739, http://dx.doi.org/10.1017/S0266467400009937.
Clauss, M., Wilkins, T., Hartley, A., Hatt, J.-M., 2009. Diet composition, food intake, body condition, and fecal consistency in captive tapirs (Tapirus spp.) in UK Collections. Zoo Biol. 28, 279–291, http://dx.doi.org/10.1002/zoo.20225.
Coley, P., Barone, J., 1996. Herbivory and plant defenses in tropical forests. Annu. Rev. Ecol. Syst. 27, 305–335, http://dx.doi.org/10.1146/annurev.ecolsys.27.1.
Crawley, M.J., 2007. The R Book. John Wiley & Sons, Ltd, Chichester, UK, http://dx.doi.org/10.1002/9780470515075.
Danell, K., Bergstrom, R., Duncan, P., Pastor, J. (Eds.), 2006. Large Herbivore Ecology, Ecosystem Dynamics and Conservation. Cambridge University Press, Cambridge, http://dx.doi.org/10.1017/CBO9780511617461.
Foerster, C.R., Vaughan, C., 2002. Home range, habitat use, and activity of Baird’s tapir in Costa Rica. Biotropica 34, 423, http://dx.doi.org/10.1646/0006-3606(2002)034[0423:HRHUAA]2.0.CO;2.
Gill, R., 2006. The influence of large herbivores on tree recruitment and forest dynamics. In: Danell, K., Bergstrom, R., Duncan, P., Pastor, J. (Eds.), Large Herbivore Ecology, Ecosystem Dynamics and Conservation. Cambridge University Press, Cambridge, pp. 170–202, http://dx.doi.org/10.1017/CBO9780511617461.008.
Guariguata, M.R., Ostertag, R., 2001. Neotropical secondary forest succession: changes in structural and functional characteristics. For. Ecol. Manage. 148, 185–206, http://dx.doi.org/10.1016/S0378-1127(00)00535-1.
Harrison, X.A., 2014. Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ 2, e616, http://dx.doi.org/10.7717/peerj.616.
Herms, D., Mattson, W., 1992. The dilemma of plants—to grow or defend. Q. Rev. Biol. 67, 283–335, http://dx.doi.org/10.1086/417659.
Hester, A.J., Bergman, M., Iason, G.R., Moen, J., 2006. Impacts of large herbivores on plant community structure and dynamics. In: Danell, K., Bergstrom, R., Duncan, P., Pastor, J. (Eds.), Large Herbivore Ecology, Ecosystem Dynamics and Conservation. Cambridge University Press, Cambridge, pp. 97–141, http://dx.doi.org/10.1017/CBO9780511617461.006.
Hibert, F., Sabatier, D., Andrivot, J., Scotti-Saintagne, C., Gonzalez, S., Prévost, M.-F., Grenand, P., Chave, J., Caron, H., Richard-Hansen, C., 2011. Botany, genetics and ethnobotany: a crossed investigation on the elusive tapir’s diet in French Guiana. PLoS One 6, e25850, http://dx.doi.org/10.1371/journal.pone.0025850.
Kuijper, D.P.J., Cromsigt, J.P.G.M., Churski, M., Adam, B., Jędrzejewska, B., Jędrzejewski, W., 2009. Do ungulates preferentially feed in forest gaps in European temperate forest? For. Ecol. Manage. 258, 1528–1535, http://dx.doi.org/10.1016/j.foreco.2009.07.010.
Lizcano, D., Cavelier, J., 2000. Daily and seasonal activity of the Mountain tapir (Tapirus pinchaque) in the Central Andes of Colombia. J. Zool. 252, 429–435, http://dx.doi.org/10.1111/j.1469-7998.2000.tb01225.x.
MacFadden, B., 2000. Cenozoic mammalian herbivores from the Americas: reconstructing ancient diets and terrestrial communities. Annu. Rev. Ecol. Syst. 31, 33–59, http://dx.doi.org/10.1146/annurev.ecolsys.31.1.33.
MacKenzie, D., Nichols, J., Lachman, G., Droege, S., Royle, J., Langtimm, C., 2002. Estimating site occupancy rates when detection probabilities are less than one. Ecology 83, 2248–2255, http://dx.doi.org/10.2307/3072056.
McCook, L., 1994. Understanding ecological community succession—causal-models and theories, a review. Vegetatio 110, 115–147, http://dx.doi.org/10.1007/BF00033394.
Medici, E.P., 2010. Assessing the Viability of Lowland Tapir Populations in a Fragmented Landscape (PhD). University of Kent, Canterbury, United Kingdom.
Naranjo, E.J., 1995. Abundancia y uso de hábitat del tapir (Tapirus bairdii) en un bosque tropical húmedo de Costa Rica. Vida Silv. Neotopical 4, 20–31.
Norris, D., Peres, C.A., Michalski, F., Hinchsliffe, K., 2008. Terrestrial mammal responses to edges in Amazonian forest patches: a study based on track stations. Mammalia 72, http://dx.doi.org/10.1515/mamm.2008.002.
Olff, H., Vera, F., Bokdam, J., Bakker, E., Gleichman, J., de Maeyer, K., Smit, R., 1999. Shifting mosaics in grazed woodlands driven by the alternation of plant facilitation and competition. Plant Biol. 1, 127–137, http://dx.doi.org/10.1055/s-2007-978499.
Padilla, M., Dowler, R.C., 1994. Tapirus terrestris. Mamm. Species 1. 10.2307/3504109.
Parry, L., Barlow, J., Peres, C.A., 2007. Large-vertebrate assemblages of primary and secondary forests in the Brazilian Amazon. J. Trop. Ecol. 23, 653–662, http://dx.doi.org/10.1017/S0266467407004506.
Pastor, J., Naiman, R., 1992. Selective foraging and ecosystem processes in Boreal forest. Am. Nat. 139, 690–705, http://dx.doi.org/10.1086/285353.
Peel, M.C., Finlayson, B.L., McMahon, T.A., 2007. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 11, 1633–1644, http://dx.doi.org/10.5194/hess-11-1633-2007.
Pinotti, B.T., Pagotto, C.P., Pardini, R., 2012. Habitat structure and food resources for wildlife across successional stages in a tropical forest. For. Ecol. Manage. 283, 119–127, http://dx.doi.org/10.1016/j.foreco.2012.07.020.
Poorter, L., Bongers, F., 2006. Leaf traits are good predictors of plant performance across 53 rain forest species. Ecology 87, 1733–1743, http://dx.doi.org/10.1890/0012-9658(2006)87[1733:LTAGPO]2.0.CO;2.
Poorter, L., de Plassche, M., Willems, S., Boot, R., 2004. Leaf traits and herbivory rates of tropical tree species differing in successional status. Plant Biol. 6, 746–754, http://dx.doi.org/10.1055/s-2004-821269.
R Development Core Team, 2016. R: A Language and Environment for Statistical Computing. the R Foundation for Statistical Computing, Vienna, Austria.
Reich, P.B., Walters, M.B., Ellsworth, D.S., 1992. Leaf life-span in relation to leaf, plant, and stand characteristics among diverse ecosystems. Ecol. Monogr. 62, 365–392, http://dx.doi.org/10.2307/2937116.
Rosenthal, J., Kotanen, P., 1994. Terrestrial plant tolerance to herbivory. Trends Ecol. Evol. 9, 145–148, http://dx.doi.org/10.1016/0169-5347(94)90180-5.
Royle, J.A., Nichols, J.D., 2003. Estimating abundance from repeated presence-absence data or point counts. Ecology 84, 777–790, http://dx.doi.org/10.1890/0012-9658(2003)084[0777:EAFRPA]2.0. CO;2.
Ruiz-Guerra, B., Guevara, R., Mariano, N.A., Dirzo, R., 2010. Insect herbivory declines with forest fragmentation and covaries with plant regeneration mode: evidence from a Mexican tropical rain forest. Oikos 119, 317–325, http://dx.doi.org/10.1111/j.1600-0706.2009.17614.x.
Salas, L., 1996. Habitat of the Lowland tapir (Tapirus terrestris) in the Tabaro River valley, southern Venezuela. Can. J. Zool. 74, 1452–1458, http://dx.doi.org/10.1139/z96-160.
Schulze, E.-D., Beck, E., Müller-Hohenstein, K., 2005. Plant Ecology. Springer, Berlin, http://dx.doi.org/10.1093/aob/mcj018.
Senft, R., Coughenour, M., Bailey, D., Rittenhouse, L., Sala, O., Swift, D., 1987. Large herbivore foraging and ecological hierarchies. Bioscience 37, 789-, http://dx.doi.org/10.2307/1310545.
Shipley, L., Illius, A., Danell, K., Hobbs, N., Spalinger, D., 1999. Predicting bite size selection of mammalian herbivores: a test of a general model of diet optimization. Oikos 84, 55–68, http://dx.doi.org/10.2307/3546866.
Simpson, B.K., Shukor, M.N., Magintan, D., Ling, W.S., 2013. Food selection of the Malayan Tapir (Tapirus indicus) under semi-wild conditions. In: Murad, A.M.H.A., Yen, C.C., Ismail, E.S., Maskat, M.Y., Noorani, M.S.M., Ibrahim, N., Karim, N.H.B.A., Yahya, R., Khalid, R.M., Ismail, W.R., Ibrahim, Z. (Eds.), The 2013 UKM FST Postgraduate Colloquium Presented at the AIP Conference Proceedings. AIP, Publishing, Selangor Malaysia, pp. 317–324, http://dx.doi.org/10.1063/1.4858676.
Stephens, D.W., Krebs, J.R., 1986. Foraging Theory, Monographs in Behavior and Ecology. Princeton University Press, Princeton, N.J (978-0-691-08441-1978-0-691-08442-8).
Tobler, M.W., Carrillo-Percastegui, S.E., Powell, G., 2009. Habitat use, activity patterns and use of mineral licks by five species of ungulate in south-eastern Peru. J. Trop. Ecol. 25, 261–270, http://dx.doi.org/10.1017/S0266467409005896.
Tobler, M., 2002. Habitat use and diet of Baird’s tapirs (Tapirus bairdii) in a montane cloud forest of the Cordillera de Talamanca, Costa Rica. Biotropica 34, 468–474, http://dx.doi.org/10.1646/0006-3606(2002)034[0468:HUADOB]2.0.CO;2.
Vancleve, K., Oliver, L., Schlentner, R., Viereck, L., Dyrness, C., 1983. Productivity and nutrient cycling in Taiga forest ecosystems. Can. J. For. Res. 13, 747–766, http://dx.doi.org/10.1139/x83-105.
Wright, S.J., 2010. The future of tropical forests: future tropical forests. Ann. N. Y. Acad. Sci. 1195, 1–27, http://dx.doi.org/10.1111/j.1749-6632.2010.05455.x.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
de Luca, J.R., Pardini, R. Use of early and late successional forest patches by the endangered Lowland tapir Tapirus terrestris (Perissodactyla: Tapiridae). Mamm Biol 86, 107–114 (2017). https://doi.org/10.1016/j.mambio.2017.08.001
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.mambio.2017.08.001