Abstract
Study Design
Case report.
Introduction
Long-term survival of a patient with a Ewing sarcoma family of tumors/primitive neuroectodermal tumors of the central spine with pathologic fracture and extradural extension is presented. Literature-based evidence for a survival benefit with modern neoadjuvant chemotherapy and en bloc resection with and without radiotherapy is reviewed.
Case Report
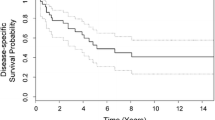
More than 10-year clinical and radiographic follow-up is given for a 14-cm-diameter tumor originating from the L4 body with pathologic fracture, unilateral pedicle involvement, and extradural canal extension. Neoadjuvant chemotherapy led to 90% tumor regression and the authors’ subsequent en bloc resection attempt. The postoperative chemotherapy was resumed early and postoperative radiotherapy was administered owing to positive microscopic margins. The patient is alive more than 10 years after completing treatment and is without evidence of recurrent disease or secondary malignancy. Late effects of chemotherapy are limited to mild cardiomyopathy controlled with medication.
Conclusions
The Ewing sarcoma family of tumors of the spinal column accounts for approximately 2% of Ewing sarcoma lesions of the skeletal system; local and systemic relapses are higher than for the extremity sites. Survival is enhanced by en bloc surgical resection in cases where clear margins are obtained, but the prognosis of patients with central (spine and pelvis) sites is typically poor. This case adds to the literature with documentation of high quality of life with long-term, disease-free survival after modern chemotherapy surgical intervention and combined radiotherapy, a finding suggested in recent large Surveillance, Epidemiology, and End Results database studies and smaller case series of this uncommon, high-grade spinal tumor. Neoadjuvant chemotherapy with attempted en bloc resection, and postoperative radiotherapy to the resected tumor bed if there are microscopic positive margins, can still lead to long-term, disease-free survival.
Similar content being viewed by others
References
Sciubba DM, Hsieh P, McLoughlin GS, et al. Pediatric tumors involving the spinal column. Neurosurg Clin N Am 2008;19:81–92.
Wold LE. Atlas of orthopedic pathology. Philadelphia: Saunders; 1990. p. 210–5.
Whitehouse GH, Griffiths GJ. Roentgenologic aspects of spinal involvement by primary and metastatic Ewing’s tumor. J Can Assoc Radiol 1976;27:290–7.
Nesbit Jr ME, Gehan EA, Burgert Jr EO, et al. Multimodal therapy for the management of primary, nonmetastatic Ewing’s sarcoma of bone: a long-term follow-up of the First Intergroup study. J Clin Oncol 1990;8:1664–74.
Nesbit Jr ME, Perez CA, Tefft M, et al. Multimodal therapy for the management of primary, nonmetastatic Ewing’s sarcoma of bone: an Intergroup Study. Natl Cancer Inst Monogr 1981;56:255–62.
Evans RG, Nesbit ME, Gehan EA, et al. Multimodal therapy for the management of localized Ewing’s sarcoma of pelvic and sacral bones: a report from the second intergroup study. J Clin Oncol 1991;9:1173–80.
Gehan EA, Nebit Jr ME, Burgert Jr EO, et al. Prognostic factors in children with Ewing’s sarcoma. National Cancer Institute Monogr 1981;56:273–8.
Rosen G, Caparros B, Nirenberg A, et al. Ewing’s sarcoma: ten-year experience with adjuvant chemotherapy. Cancer 1981;47:2204–13.
DeLaney TF, Liebsch NJ, Pedlow FX, et al. Phase II study of highdose photon/proton radiotherapy in the management of spine sarcomas. Int J Radiat Oncol Biol Phys 2009;74:732–9.
Graham-Pole J. Ewing sarcoma: treatment with high dose radiation and adjuvant chemotherapy. Med Pediatr Oncol 1979;7:1–8.
Paulino AC, Nguyen TX, Mai WY. An analysis of primary site control and late effects according to local control modality in nonmetastatic Ewing sarcoma. Pediatr Blood Cancer 2007;48:423–9.
Wagner TD, Kobayashi W, Dean S, et al. Combination short-course preoperative irradiation, surgical resection, and reduced-field high-dose postoperative irradiation in the treatment of tumors involving the bone. Int J Radiat Oncol Biol Phys 2009;73:259–66.
Bacci G, Ferrari S, Longhi A, et al. Local and systemic control in Ewing’s sarcoma of the femur treated with chemotherapy, and locally by radiotherapy and/or surgery. J Bone Joint Surg Br 2003;85:107–14.
Burdach S, Jurgens H, Peters C, et al. Myeloablative radiochemotherapy and hematopoietic stem-cell rescue in poor-prognosis Ewing’s sarcoma. J Clin Oncol 1993;11:1482–8.
Gibbs IC, Tuamokumo N, Yock TI. Role of radiation therapy in pediatric cancer. Hematol Oncol Clin N Am 2006;20:455–70.
Goldman A. Ewing’s sarcoma: treatment with high-dose radiation and adjuvant chemotherapy. Recent Results Cancer Res 1982;80:115–9.
Horowitz ME, Neff JR, Kun LE. Ewing’s sarcoma: radiotherapy versus surgery for local control. Pediatr Clin N Am 1991;38:365–80.
Hristov B, Shokek O, Frassica DA. The role of radiation treatment in the contemporary management of bone tumors. J Natl Comp Cancer Network 2007;5:456–66.
Indelicato DJ, Keole SR, Shahlaee AH, et al. Impact of local management on long-term outcomes in Ewing tumors of the pelvis and sacral bones: the University of Florida experience. Int J Radiat Oncol Biol Phys 2008;72:41–8.
Krasin MJ, Rodriguez-Galindo C, Billups CA, et al. Definitive irradiation in multidisciplinary management of localized Ewing sarcoma family of tumors in pediatric patients: outcome and prognostic factors. Int J Radiat Oncol Biol Phys 2004;60:830–8.
La TH, Meyers PA, Wexler LH, et al. Radiation therapy for Ewing’s sarcoma: results from Memorial Sloan-Kettering in the modern era. Int J Radiat Oncol Biol Phys 2006;64:544–50.
Perez CA, Tefft M, Nesbit Jr ME, et al. Radiation therapy in the multimodal management of Ewing’s sarcoma of bone: report of the Intergroup Ewing’s Sarcoma Study. Natl Cancer Inst Monogr 1981;56:263–71.
Perez CA, Tefft M, Nesbit M, et al. The role of radiation therapy in the management of non-metastatic Ewing’s sarcoma of bone: report of the Intergroup Ewing’s Sarcoma Study. Int J Radiat Oncol Biol Phys 1981;7:141–9.
Pilepich MV, Vietti TJ, Nesbit ME, et al. Radiotherapy and combination chemotherapy in advanced Ewing’s Sarcoma-Intergroup study. Cancer 1981;47:1930–6.
Sharafuddin MJ, Haddad FS, Hitchon PW, et al. Treatment options in primary Ewing’s sarcoma of the spine: report of seven cases and review of the literature. Neurosurgery 1992;30:610–8; discussion 618–9.
Shi W, Indelicato DJ, Keole SR, et al. Radiation treatment for Ewing family of tumors in adults: the University of Florida experience. Int J Radiat Oncol Biol Phys 2008;72:1140–5.
Thomas PR, Perez CA, Neff JR, et al. The management of Ewing’s sarcoma: role of radiotherapy in local tumor control. Cancer Treat Rep 1984;68:703–10.
Vlasak R, Sim FH. Ewing’s sarcoma. Orthop Clin N Am 1996;27: 591–603.
Halm H. [Augmentation of VDS (ventral derotation spondylodesis) using double rod instrumentation: surgical method and early results]. Z Orthop Ihre Grenzgeb 1994;132:383–9.
Talac R, Yaszemski MJ, Currier BL, et al. Relationship between surgical margins and local recurrence in sarcomas of the spine. Clin Orthop Relat Res 2002;397:127–32.
Mukherjee D, Chaichana KL, Gokaslan ZL, et al. Survival of patients with malignant primary osseous spinal neoplasms: results from the Surveillance, Epidemiology, and End Results (SEER) database from 1973 to 2003. J Neurosurg Spine 2011;14:143–50.
Mukherjee D, Chaichana KL, Adogwa O, et al. Association of extent of local tumor invasion and survival in patients with malignant primary osseous spinal neoplasms from the surveillance, epidemiology, and end results (SEER) database. World Neurosurg 2011;76:580–5.
Chaichana KL, Parker SL, Mukherjee D, et al. Assessment of the extent of surgical resection as a predictor of survival in patients with primary osseous spinal neoplasms. Clin Neurosurg 2011;58:117–21.
Parker SL. Prolonged survival achieved with surgical resection for spinal tumor patients. Orthop Today 2011;31(3):48–50.
Emery SE, Gocke RR. The textbook of spinal surgery. 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2011. p. 1553–9.
Boriani S, Weinstein JN, Biagini R. Primary bone tumors of the spine: terminology and surgical staging. Spine (Phila Pa 1976) 1997;22:1036–44.
Weinstein SL. The pediatric spine: principles and practice. New York: Raven Press; 1994. p. 900–1.
Bacci G, Longhi A, Briccoli A, et al. The role of surgical margins in treatment of Ewing’s sarcoma family tumors: experience of a single institution with 512 patients treated with adjuvant and neoadjuvant chemotherapy. Int J Radiat Oncol Biol Phys 2006;65:766–72.
Indelicato DJ. Spinal and paraspinal Ewing tumors. Int J Radiat Oncol Biol Phys 2010;76:1463–71.
Boriani S, Amendola L, Corghi A, et al. Ewing’s sarcoma of the mobile spine. Eur Rev Med Pharmacol Sci 2011;15:831–9.
Marco RA, Gentry JB, Rhines LD, et al. Ewing’s sarcoma of the mobile spine. Spine (Phila Pa 1976) 2005;30:769–73.
Nathoo N, Mendel E. The National Cancer Institute’s SEER Registry and primary malignant osseous spine tumors. World Neurosurg 2011;76:531–2.
Vogin G, Helfre S, Glorion C, et al. Local control and sequelae in localised Ewing tumours of the spine: a French retrospective study. Eur J Cancer 2013;49:1314–23.
Haeusler J, Ranft A, Boelling T, et al. The value of local treatment in patients with primary, disseminated, multifocal Ewing sarcoma (PDMES). Cancer 2010;116:443–50.
Maheshwari AV, Cheng EY. Ewing sarcoma family of tumors. J Am Acad Orthop Surg 2010;18:94–107.
Shamberger RC, LaQuaglia MP, Gebhardt MC, et al. Ewing sarcoma/primitive neuroectodermal tumor of the chest wall: impact of initial versus delayed resection on tumor margins, survival, and use of radiation therapy. Ann Surg 2003;238:563–7; discussion 567–8.
Gibbs CP, Kukekov VG, Reith JD, et al. Stem-like cells in bone sarcomas: implications for tumorigenesis. Neoplasia 2005;7:967–76.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author disclosures: EPS (consultancy for Pioneer Surgical Technologies; expert testimony 2007–13; travel/accommodations/meeting expenses from Alphatec; stock from Pioneer Surgical Technologies; committee member for SRS Growing Spine Committee); SH (none); IS (none); JZF (none).
Rights and permissions
About this article
Cite this article
Southern, E.P., Hohl, S., Singh, I. et al. Long-Term, Disease-Free Survival of a Patient With a Primitive Neuroectodermal/Ewing Sarcoma in the Mobile Spine With Extracompartmental Extension. Spine Deform 2, 158–164 (2014). https://doi.org/10.1016/j.jspd.2013.12.001
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jspd.2013.12.001