Abstract
Objective
Intrauterine infection has been linked to brain injury in human infants, although the mechanisms are not fully understood. We recently showed that repeated acute exposure of preterm fetal sheep to bacterial endotoxin (lipopoly saccharide [LPS]) results in fetal hypoxemia, hypotension, increased systemic proinflammatory cytokines, and brain damage, including white matter injury. However, it is not clear whether this injury is caused by reduced cerebral oxygen delivery or inflammatory pathways independent of hypoxia. The aim of the present study was to determine the effects on the fetal brain and placenta of a chronic intrauterine inflammatory state, induced by LPS infusion into the fetal circulation, a model that did not cause hypoxia.
Methods
At 0.65 of term, eight catheterized fetal sheep received intravenous infusions of LPS (5 to 15 μg) over 5 days; control fetuses received saline. Fetal physiologic responses were monitored throughout the infusion. Fetal brain and placental tissues were examined histologically 6 days after the conclusion of the infusion.
Results
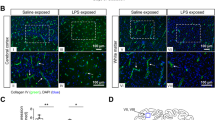
LPS infusions did not result in physiologically significant alterations to fetal blood gases or mean arterial pressure; however, plasma proinflammatory cytokine levels were elevated. Following LPS exposure there was no difference in fetal body or brain weights (P >.05); placental weight was reduced (P <.05), consistent with reduced placentome cross-sectional area (P <.05). In the cerebral hemispheres subcortical white matter injury was present in six LPS-exposed fetuses and included axonal damage, microgliosis, oligodendrocyte injury, and increased β amyloid precursor protein (β-APP) expression.
Conclusions
Chronic, systemic exposure of the fetus to LPS resulted in fetal brain damage in the absence of hypoxemia or hypotension, although the resulting injury was less severe than following repeated acute exposure, (J Soc Gynecol Investig 2006;13:87–96) Copyright © 2006 by the Society for Gynecologic Investigation.
Similar content being viewed by others
References
Volpe JJ. Perinatal brain injury: From pathogenesis to neuroprotection. Ment Retard Dev Disabil Res Rev 2001;7:56–64.
Maalouf EF, Duggan PJ, Rutherford MA, et al. Magnetic reso-nance imaging of the brain in a cohort of extremely preterm infants. J Pediatr 1999;135:351–7.
Counsell SJ, Allsop JM, Harrison MC, et al. Diffusion-weighted imaging of the brain in preterm infants with focal and diffuse white matter abnormality. Pediatrics 2003;112:1–7.
Inder TE, Anderson NJ, Spencer C, Wells S, Volpe JJ. White matter injury in the premature infant: A comparison between serial cranial sonographic and MR findings at term. AJNR Am J Neuroradiol 2003;24:805–9.
Hamrick SE, Miller SP, Leonard C, et al. Trends in severe brain injury and neurodevelopmental outcome in premature newborn infants: The role of cystic periventricular leukomalacia. J Pediatr 2004;145:593–9.
Inder TE, Huppi PS, Warfield S, et al. Periventricular white matter injury in the premature infant is followed by reduced cerebral cortical gray matter volume at term. Ann Neurol 1999;46:755–60.
Isaacs EB, Lucas A, Chong WK, et al. Hippocampal volume and everyday memory in children of very low birth weight. Pediatr Res 2000;47:713–20.
Allin M, Matsumoto H, Santhouse AM, et al. Cognitive and motor function and the size of the cerebellum in adolescents bom very preterm. Brain 2001;124:60–6.
Kinney HC. Human myelination and perinatal white matter disorders. J Neurol Sci 2005;228:190–2.
Yoon BH, Romero R, Kim CJ, et al. Amniotic fluid interleu-kin-6: A sensitive test for antenatal diagnosis of acute inflammatory lesions of preterm placenta and prediction of perinatal morbidity. Am J Obstet Gynecol 1995;172:960–70.
Yoon BH, Romero R, Yang SH, et al. Interleukin-6 concen-trations in umbilical cord plasma are elevated in neonates with white matter lesions associated with periventricular leukomalacia. Am J Obstet Gynecol 1996;174:1433–40.
Kadhim H, Tabarki B, Verellen G, De Prez C, Rona AM, Sebire G. Inflammatory cytokines in the pathogenesis of periventricular leukomalacia. Neurology 2001;56:1278–84.
Deguchi K, Mizuguchi M, Takashima S. Immunohistochemical expression of tumor necrosis factor alpha in neonatal leukomalacia. Pediatr Neurol 1996;14:13–6.
Yoon BH, Romero R, Kim CJ, Koo JN, Choe G, Syn HC, Chi JG. High expression of tumor necrosis factor-alpha and interleukin-6 in periventricular leukomalacia. Am J Obstet Gynecol 1997;177:406–11.
Folkerth RD, Keefe RJ, Haynes RL, Trachtenberg FL, Volpe JJ, Kinney HC. Interferon-gamma expression in periventricular leukomalacia in the human brain. Brain Pathol 2004;14:265–74.
Wu YW. Systematic review of chorioamnionitis and cerebral palsy. Ment Retard Dev Disabil Res Rev 2002;8:25–9.
Baud O, Emilie D, Pelletier E, et al. Amniotic fluid concentrations of interleukin-lbeta, interleukin-6 and TNF-alpha in chorioamnionitis before 32 weeks of gestation: Histological associations and neonatal outcome. Br J Obstet Gynaecol 1999;106:72–7.
Nelson KB, Grether JK, Dambrosia JM, et al. Neonatal cytokines and cerebral palsy in very preterm infants. Pediatr Res 2003;53:600–7.
Rothwell NJ, Strijbos PJ. Cytokines in neurodegeneration and repair. Int J Dev Neurosci 1995;13:179–85.
Duncan JR, Cock M, Scheerlinck JP, et al. White matter injury after repeated endotoxin exposure in the preterm ovine fetus. Pediatr Res 2002;52:941–9.
Bell MJ, Hallenbeck JM. Effects of intrauterine inflammation on developing rat brain. J Neurosci Res 2002;70:570–9.
Gilles FH, Leviton A, Kerr CS. Endotoxin leucoencephalopathy in the telencephalon of the newborn kitten. J Neurol Sci 1976;27:183–91.
Ornoy A, Altshuler G. Maternal endotoxemia, fetal anomalies, and central nervous system damage: A rat model of a human problem. Am J Obstet Gynecol 1976;124:196–204.
Cai Z, Pan ZL, Pang Y, Evans OB, Rhodes PG. Cytokine induction in fetal rat brains and brain injury in neonatal rats after maternal lipopolysaccharide administration. Pediatr Res 2000;47:64–72.
Mallard C, Welin AK, Peebles D, Hagberg H, Kjellmer I. White matter injury following systemic endotoxemia or asphyxia in the fetal sheep. Neurochem Res 2003;28:215–23.
Yoon BH, Kim CJ, Romero R, Jun JK, Park KH, Choi ST, Chi JG. Experimentally induced intrauterine infection causes fetal brain white matter lesions in rabbits. Am J Obstet Gynecol 1997;177:797–802.
Debillon T, Gras-Leguen C, Verielle V, et al. Intrauterine infection induces programmed cell death in rabbit periventricular white matter. Pediatr Res 2000;47:736–42.
Kinney HC, Back SA. Human oligodendroglial development: Relationship to periventricular leukomalacia. Semin Pediatr Neurol 1998;5:180–9.
McCrabb GJ, Harding R. Cerebral blood flow is increased throughout 12h of hypoxemia in the midgestation ovine fetus. Reprod Fertil Dev 1995;7:463–7.
Egan PJ, Rothel JS, Andrews AE, Seow HF, Wood PR, Nash AD. Characterization of monoclonal antibodies to ovine tumor necrosis factor-alpha and development of a sensitive immunoassay. Vet Immunol Immunopathol 1994;41:259–74.
McWaters P, Hurst L, Chaplin PJ, Collins RA, Wood PR, Scheerlinck JP. Characterisation of monoclonal antibodies to ovine interleukin-6 and the development of a sensitive capture ELISA. Vet Immunol Immunopathol 2000;73:155–65.
Geddes JF, Whitwell HL, Graham DI. Traumatic axonal injury: Practical issues for diagnosis in medicolegal cases. Neuropathol Appl Neurobiol 2000;26:105–16.
Nitsos I, Rees S. The effects of intrauterine growth retardation on the development of neuroglia in fetal guinea pigs. An immu-nohistochemical and an ultrastructural study. Int J Dev Neurosci 1990;8:233–44.
Rungby J, Kassem M, Eriksen EF, Danscher G. The von Kossa reaction for calcium deposits: Silver lactate staining increases sensitivity and reduces background. Histtochem J 1993;25:446–51.
Duncan JR, Cock ML, Rees S, Harding R. The effects of repeated endotoxin exposure on placental structure in sheep. Placenta 2003;24:786–9.
Dalitz P, Harding R, Rees SM, Cock ML. Prolonged reductions in placental blood flow and cerebral oxygen delivery in preterm fetal sheep exposed to endotoxin: Possible factors in white matter injury after acute infection. J Soc Gynecol Investig 2003;10:283–90.
Rothel JS, Hurst L, Seow HF, et al. Analysis of ovine IL-1 beta production in vivo and in vitro by enzyme immunoassay and immunohistochemistry. Vet Immunol Immunopathol 1997;57:267–78.
Bell MJ, Hallenbeck JM, Gallo V. Determining the fetal inflam-matory response in an experimental model of intrauterine inflammation in rats. Pediatr Res 2004;56:541–6.
Yan E, Castillo-Melendez M, Nicholls T, Hirst J, Walker D. Cerebrovascular responses in the fetal sheep brain to lowdose endotoxin. Pediatr Res 2004;55:855–63.
Briscoe T, Duncan J, Cock M, Rice GE, Harding R, Scheerlinck JP, Rees S. Activation of NF-kappaB transcription factor in the preterm ovine brain and placenta after acute LPS-exposure. J Neurosci Res (in press).
Reichard RR, White CL 3rd, Hladik CL, Dolinak D. Beta-amyloid precursor protein staining of nonaccidental central nervous system injury in pediatric autopsies. J Neurotrauma 2003;20:347–55.
Saljo A, Bao F, Shi J, Hamberger A, Hansson HA, Haglid KG. Expression of c-Fos and c-Myc and deposition of beta-APP in neurons in the adult rat brain as a result of exposure to short-lasting impulse noise. J Neurotrauma 2002;19:379–85.
Yoshikawa K, Aizawa T, Hayashi Y. Degeneration in vitro of post-mitotic neurons overexpressing the Alzheimer amyloid protein precursor. Nature 1992;359:64–7.
Marin-Padilla M. Developmental neuropathology and impact of perinatal brain damage. Ill: Gray matter lesions of the neocortex. J Neuropathol Exp Neurol 1999;58:407–29.
Gonzalez-Scarano F, Baltuch G. Microglia as mediators of inflammatory and degenerative diseases. Ann Rev Neurosc 1999;22:219–40.
Giulian D, Woodward J, Young DG, Krebs JF, Lachman LB. Interleukin-1 injected into mammalian brain stimulates astrogliosis and neovascularization. J Neurosci 1988;8:2485–90.
Olson EE, McKeon RJ. Characterization of cellular and neuro-logical damage following unilateral hypoxia/ischemia. J Neurol Sci 2004;227:7–19.
Tahraoui SL, Marret S, Bodenant C, et al. Central role of microglia in neonatal excitotoxic lesions of the murine periven-tricular white matter. Brain Pathol 2001;11:56–71.
Gao HM, Jiang J, Wilson B, Zhang W, Hong JS, Liu B. Microglial activation-mediated delayed and progressive degeneration of rat nigral dopaminergic neurons: Relevance to Parkinson’s disease. J Neurochem 2002;81:1285–97.
Boelen A, Kwakkel J, Thijssen-Timmer DC, Alkemade A, Fliers E, Wiersinga WM. Simultaneous changes in central and peripheral components of the hypothalamus-pituitary-thyroid axis in lipopolysaccharide-induced acute illness in mice. J Endocrinol 2004;182:315–23.
Peebles DM, Miller S, Newman JP, Scott R, Hanson MA. The effect of systemic administration of lipopolysaccharide on cerebral haemodynamics and oxygenation in the 0.65 gestation ovine fetus in utero. BJOG 2003;110:735–43.
Sherwin C, Fern R. Acute lipopolysaccharide-mediated injury in neonatal white matter glia: Role of TNF-{alpha}, IL-l{beta}, and calcium. J Immunol 2005;175:155–61.
Rees S, Stringer M, Just Y, Hooper SB, Harding R. The vulnerability of the fetal sheep brain to hypoxemia at mid-gestation. Dev Brain Res 1997;103:103–18.
Rees S, Breen S, Loeliger M, McCrabb G, Harding R. Hypoxemia near mid-gestation has long-term effects on fetal brain development. J Neuropathol Exp Neurol 1999;58:932–45.
Eklind S, Mallard C, Leverin AL, et al. Bacterial endotoxin sensitizes the immature brain to hypoxic-ischaemic injury. Eur J Neurosci 2001;13:1101–6.
Author information
Authors and Affiliations
Corresponding author
Additional information
Joint senior authors.
Supported by the National Health and Medical Research Council of Australia and the Australian Research Council.
The authors are grateful to Alex Satragno and Todd Briscoe for technical assistance and to Dr Roberto Cappai (University of Melbourne, Parkville, Victoria, Australia) for his kind donation of the β-APP antibody.
Rights and permissions
About this article
Cite this article
Duncan, J.R., Cock, M.L., Suzuki, K. et al. Chronic Endotoxin Exposure Causes Brain Injury in the Ovine Fetus in the Absence of Hypoxemia. Reprod. Sci. 13, 87–96 (2006). https://doi.org/10.1016/j.jsgi.2005.12.003
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jsgi.2005.12.003