Abstract
Objective
We examined whether estrogen action in human parturition is regulated by an intracrine mechanism mediated by target tissue expression of specific 17β-hydroxysteroid dehydrogenase (17βHSD) isozymes that interconvert estrone (El) and estradiol (E2), such that the onset of labor is associated with an increase in local E2 bioavailability.
Methods
The extent of 17βHSD-1, -2, -3, -4, -5, and -7 expression (measured by quantitative reverse transcriptase polymerase chain reaction) and the capacity to interconvert E1 and E2 were compared in amnion, chorion, placenta, decidua, and myometrium obtained from women at term before (n = 6) and after (n = 6) the onset of labor.
Results
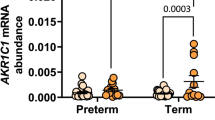
In chorion, abundance of 17βHSD-1 (converts E1 to E2) mRNA decreased 2.7-fold (P <.05) in association with labor onset. In myometrium, 17βHSD-1 and 17βHSD-4 (converts E2 to E1) mRNAs increased two-fold and five-fold, respectively, with the onset of labor (P <.05 for each). No other statistically significant labor-associated change in 17βHSD expression was observed. In chorion, 17βHSD oxidative (E2 to E1) and reductive (E1 to E2) activities and the net E2 synthetic capacity increased with labor. In decidua, both activities decreased with the onset of labor, but there was no change in net E2 synthetic capacity. The capacity to interconvert E1 and E2 did not change in the other tissues.
Conclusion
The increase in E2 synthetic capacity in the chorion might contribute to an increase in local estrogen bioactivity in association with the onset of labor. However, it cannot be explained by changes in 17βHSD isozyme expression and is unlikely to account for the increased estrogen action at parturition. These data show that intracrine mechanisms based on 17βHSD isozyme expression play a minor role, if any, in controlling estrogen action in gestational tissues during human parturition.
Similar content being viewed by others
References
Mesiano S. Roles of estrogen and progesterone in human parturition. Front Horm Res 2001;27:86–104.
Pepe G, Albrecht E. Actions pf placental and fetal adrenal steroid hormones in primate pregnancy. Endocr Rev 1995;16:608–48.
Siiteri PK, MacDonald PC. Placental estrogen biosynthesis during human pregnancy. J Clin Endocrinol Metab 1966;7:751–61.
Yen SSC. Endocrine-metabolic adaptations in pregnancy. In: Yen SSC, Jaffe RJB, eds. Reproductive endocrinology. 3rd ed. Philadelphia: WB Saunders, 1991:936–81.
Labrie F, Luu-The V, Lin SX, et al. Intracrinology: Role of the family of 17 beta-hydroxysteroid dehydrogenases in human physiology and disease. J Mol Endocrinol 2000;1:1–16.
Labrie F, Luu-The V, Lin SX, Simard J, Labrie C. Role of 17 beta-hydroxysteroid dehydrogenases in sex steroid formation in peripheral intracrine tissues. Trends Endocrinol Metab 2000;10:421–7.
Mitchell BF, Wong S. Changes in 17 beta,20 alpha-hydroxy-steroid dehydrogenase activity supporting an increase in the estrogen/progesterone ratio of human fetal membranes at parturition. Am J Obstet Gynecol 1993;5:1377–85.
Peltoketo H, Vihko P, Vihko R. Regulation of estrogen action: Role of 17 beta-hydroxysteroid dehydrogenases. Vitam Horm 1999;55:353–98.
Luu-The V. Analysis and characteristics of multiple types of human 17beta-hydroxysteroid dehydrogenase. J Steroid Bio-chem Mol Biol 2001;76:143–51.
Penning TM. Molecular endocrinology of hydroxysteroid dehydrogenases. Endocr Rev 1997;3:281–305.
Labrie F, Luu-The V, Lin SX, et al. The key role of 17 beta-hydroxysteroid dehydrogenases in sex steroid biology. Steroids 1997;1:148–58.
Poutanen M, Isomaa V, Peltoketo H, Vihko R. Regulation of oestrogen action: Role of 17 beta-hydroxysteroid dehydrogenases. Ann Med 1995;6:675–82.
Fournet-Dulguerov N, MacLusky NJ, Leranth CZ, et al. Immu-nohistochemical localization of aromatase cytochrome P-450 and estradiol dehydrogenase in the syncytiotrophoblast of the human placenta. J Clin Endocrinol Metab 1987;4:757–64.
Tremblay Y, Ringler GE, Morel Y, et al. Regulation of the gene for estrogenic 17-ketosteroid reductase lying on chromosome 17cen-q25. J Biol Chem 1989;34:20458–62.
Ghersevich S, Nokelainen P, Poutanen M, et al. Rat 17 beta-hydroxysteroid dehydrogenase type 1: Primary structure and regulation of enzyme expression in rat ovary by diethylstilbestrol and gonadotropins in vivo. Endocrinology 1994;4:1477–87.
Sawetawan C, Milewich L, Word RA, Carr BR, Rainey WE. Compartmentalization of type I 17 beta-hydroxysteroid oxi-doreductase in the human ovary. Mol Cell Endocrinol 1994;2:161–8.
Luu-The V, Dufort I, Pelletier G, Labrie F. Type 5 17beta-hydroxysteroid dehydrogenase: Its role in the formation of androgens in women. Mol Cell Endocrinol 2001;171:77–82.
Dufort I, Rheault P, Huang XF, Soucy P, Luu-The V. Characteristics of a highly labile human type 5 17beta-hydroxysteroid dehydrogenase. Endocrinology 1999;2:568–74.
Schwabe I, Husen B, Einspanier A. Expression of the estradiol-synthesizing 17beta-hydroxysteroid dehydrogenases type 1 and type 7 in the nonhuman primate Callithrix jacchus. Mol Cell Endocrinol 2001;171:187–92.
Breitling R, Krazeisen A, Moller G, Adamski J. 17Beta-hydroxy-steroid dehydrogenase type 7—An ancient 3-ketosteroid reductase of cholesterogenesis. Mol Cell Endocrinol 2001;171:199–204.
Geissler WM, Davis DL, Wu L, et al. Male pseudohermaphroditism caused by mutations of testicular 17 beta-hydroxysteroid dehydrogenase 3. Nat Genet 1994;1:34–9.
Husen B, Adamski J, Rune GM, Einspanier A. Mechanisms of estradiol inactivation in primate endometrium. Mol Cell Endocrinol 2001;171:179–85.
Casey ML, MacDonald PC, Andersson S. 17 Beta-hydroxysteroid dehydrogenase type 2: Chromosomal assignment and progestin regulation of gene expression in human endometrium. J Clin Invest 1994;5:2135–41.
Wu L, Einstein M, Geissler WM, Chan HK, EUiston KO, Andersson S. Expression cloning and characterization of human 17 beta-hydroxysteroid dehydrogenase type 2, a microsomal enzyme possessing 20 alpha-hydroxysteroid dehydrogenase activity. J Biol Chem 1993;17:12964–9.
Breitling R, Marijanovic Z, Perovic D, Adamski J. Evolution of 17beta-HSD type 4, a multifunctional protein of beta-oxidation. Mol Cell Endocrinol 2001; 1-2:205–10.
Kikuti YY, Tamiya G, Ando A, et al. Physical mapping 220 kb centromeric of the human MHC and DNA sequence analysis of the 43-kb segment including the RING1, HKE6, and HKE4 genes. Genomics 1997;3:422–35.
Fomitcheva J, Baker ME, Anderson E, Lee GY, Aziz N. Characterization of Ke 6, a new 17beta-hydroxysteroid dehydrogenase, and its expression in gonadal tissues. J Biol Chem 1998;35:22664–71.
Adamski J, Jakob FJ. A guide to 17beta-hydroxysteroid dehydrogenases. Mol Cell Endocrinol 2001;171:1–4.
Li KX, Smith RE, Krozowski ZS. Cloning and expression of a novel tissue specific 17beta-hydroxysteroid dehydrogenase. Endocr Res 1998;24:663–7.
He XY, Merz G, Yang YZ, et al. Function of human brain short chain L-3-hydroxyacyl coenzyme A dehydrogenase in androgen metabolism. Biochim Biophys Acta 2000;1484:267–77.
Chai Z, Brereton P, Suzuki T, et al. 17 Beta-hydroxysteroid dehydrogenase type XI localizes to human steroidogenic cells. Endocrinology 2003;5:2084–91.
Martel C, Rheaume E, Takahashi M, et al. Distribution of 17 beta-hydroxysteroid dehydrogenase gene expression and activity in rat and human tissues. J Steroid Biochem Mol Biol 1992;41:597–603.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of proteindye binding. Anal Biochem 1976;72:248–54.
Mustonen MV, Isomaa W, Vaskivuo T, et al. Human 17beta-hydroxysteroid dehydrogenase type 2 messenger ribonucleic acid expression and localization in term placenta and in endometrium during the menstrual cycle. J Clin Endocrinol Metab 1998;4:1319–24.
Peltoketo H, Luu-The V, Simard J, Adamski J. 17Beta-hydroxy-steroid dehydrogenase (HSD)/17-ketosteroid reductase (KSR) family; Nomenclature and main characteristics of the 17HSD/ KSR enzymes. J Mol Endocrinol 1999;1:1–11.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported in part by grants to S.M. from the National Health and Medical Research Council of Australia (#9902490), The Hunter Medical Research Institute, The Mothers and Babies Research Centre and the University of Newcastle.
Rights and permissions
About this article
Cite this article
Madsen, G., Zakar, T., Manuelpillai, U. et al. Intracrine Control of Estrogen Action in Human Gestational Tissues at Parturition. Reprod. Sci. 11, 213–219 (2004). https://doi.org/10.1016/j.jsgi.2003.12.003
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jsgi.2003.12.003