Abstract
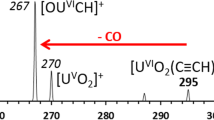
The following ions [UO2(NO3)3]−, [UO2(ClO4)3]−, [UO2(CH3COO)3]− were generated from respective salts (UO2(NO3)2, UO2(ClO4)3, UO2(CH3COO)2) by laser desorption/ionization (LDI). Collision induced dissociation of the ions has led, among others, to the formation of UO4 − ion (m/z 302). The undertaken quantum mechanical calculations showed this ion is most likely to possess square planar geometry as suggested by MP2 results or strongly deformed geometry in between tetrahedral and square planar as indicated by DFT results. Interestingly, geometrical parameters and analysis of electron density suggest it is an UVI compound, in which oxygen atoms bear unpaired electron and negative charge.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Emsley, J. The Elements, Oxford Chemistry Guides, 2nd ed.; Clarendon Press: Oxford, 1991, p. 214.
Mizuoka, K.; Tsushima, S.; Hasegawa, M.; Hoshi, T.; Ikeda, Y. Electronic Spectra of Pure Uranyl(V) Complexes: Characteristic Absorption Bands Due to a UvO2 + Core in Visible and Near-Infrared Regions. Inorg. Chem. 2005, 44, 6211–6218.
Kubatko, K.-A.; Helean, K. B.; Navrotsky, A.; Burns, P. C. Stability of Peroxide-Containing Uranyl Minerals. Science 2003, 302, 648, 1191–1193.
Sigmon, G. E.; Ling, J.; Unruh, D. K.; Moore-Shay, L.; Ward, M.; Weaver, B.; Burns, P. C. Uranyl-Peroxide Interactions Favor Nanocluster Self Assembly. J. Am. Chem. Soc. 2009, 131, 16648–16649.
Kubatko, K.-A.; Forbes, T. Z.; Klingensmith, A. L.; Burns, P. C. Expanding the Crystal Chemistry of Uranyl Peroxides: Synthesis and Structures of Di- and Triperoxodioxouranium(VI) Complexes. Inorg. Chem. 2007, 46, 3657–3662.
Kubatko, K.-A.; Burns, P. C. Expanding the Crystal Chemistry of Actinyl Peroxides: Open Sheets of Uranyl Polyhedra in Na5[(UO2)3(O2)4(OH)3](H2O)13. Inorg. Chem. 2006, 45, 6096–6098.
Masci, B.; Thuéry, P. Uranyl Complexes with the Pyridine-2,6-dicarboxylato Ligand: New Dinuclear Species with μ-η2,η2-Peroxide, μ2-Hydroxide or μ2-Methoxide Bridges. Polyhedron 2005, 24, 229–237.
Kemp, T. J.; Jennings, K. R.; Read, P. A. Formation and Decomposition of Uranium-Oxygen Clusters in Fast Atom Bombardment of Dioxouranium(v1) Salts. J. Chem. Soc. Dalton Trans. 1995, (5), 885–889.
Tsierkezos, N. G.; Roithová, J.; Schröder, D.; Onîčák, M.; Slavíîcek, P. Can Electrospray Mass Spectrometry Quantitatively Probe Speciation? Hydrolysis of Uranyl Nitrate Studied by Gas-Phase Methods. Inorg. Chem. 2009, 48, 6287–6296.
Pasilis, S.; Somogyi, Á.; Herrmann, K.; Pemberton, J. E. Ions Generated from Uranyl Nitrate Solutions by Electrospray Ionization (ESI) and Detected with Fourier Transform Ion-Cyclotron Resonance (FT-ICR) Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2006, 17, 230–240.
Chien, W.; Anbalagan, V.; Zandler, M.; Hanna, D.; Gresham, G.; Groenewold, G. Intrinsic Hydration of Monopositive Uranyl Hydroxide, Nitrate, and Acetate Cations. J. Am. Soc. Mass Spectrom. 2004, 15, 777–783.
Van Stipdonk, M. J.; Chien, W.; Anbalagan, V.; Hanna, D.; Groenewold, G. S. Gas-Phase Complexes Containing the Uranyl Ion and Acetone. J. Phys. Chem. A 2004, 108, 10448–10457.
Anbalagan, V.; Chien, W.; Gresham, G. L.; Groenewold, G. S.; Van Stipdonk, M. J. Production and Collision-induced Dissociation of Gasphase, Water- and Alcohol-Coordinated Uranyl Complexes Containing Halide or Perchlorate Anions. Rapid Commun. Mass Spectrom. 2004, 18, 3028–3034.
Moulin, C.; Charron, N.; Plancque, G.; Virelizier, H. Speciation of Uranium by Electrospray Ionization Mass Spectrometry: Comparison with Time-Resolved Laser-Induced Fluorescence. Appl. Spectrosc. 2000, 54, 843–848.
Henderson, W.; McIndoe, J. S. Mass Spectrometry of Inorganic Coordination and Organometallic Compounds; John Wiley and Sons, Ltd.: Chichester, 2005, p. 113.
Schnaars, D. D.; Wu, G.; Hayton, R. W. Reduction of Pentavalent Uranyl to U(IV) Facilitated by Oxo Functionalization. J. Am. Chem. Soc. 2009, 131, 17532–17533.
Khan, A. S. A.; Ahmed, R.; Mirza, M. L. Evaluation of Kinetic Parameters of Uranyl Acetate Complexes in Ethanolic Solution by Cyclic Voltammetry. J. Radioanal. Nucl. Chem. 2010, 283, 527–531.
Groenewold, G. S.; Van Stipdonk, M. J.; De Jong, W. A.; Oomens, J.; Gresham, G. L.; McIlwain, M. A.; Gao, D.; Siboulet, B.; Visscher, L.; Kullman, M.; Polfer, N. Infrared Spectroscopy of Dioxouranium(V) Complexes with Solvent Molecules: Effect of Reduction. Chem. Phys. Chem. 2008, 9, 1278–1285.
Pyykko, P.; Zhaot, Y. The Large Range of Uranyl Bond Lengths: Ab Initio Calculations on Simple Uranium-Oxygen Clusters. Inorg. Chem. 1991, 30, 3787–3788.
King, R. B. Some Aspects of Structure and Bonding in Binary and Ternary Uranium(VI) Oxides. Chem. Mater. 2002, 14, 3628–3635.
Bolvin, H.; Wahlgren, U.; Gropen, O.; Marsden, C. Ab Initio Study of the Two Iso-Electronic Molecules NpO4 − and UO4 2−. J. Phys. Chem. A 2001, 105, 10570–10576.
Bryantsev, V. S.; De Jong, W. A.; Cossel, K. C.; Diallo, M. S.; Goddard, W. A.; Groenewold, G. S.; Chien, W.; Van Stipdonk, M. J. Two-Electron Three-Centered Bond in Side-On (η2) Uranyl(V) Superoxo Complexes. J. Phys. Chem. A 2008, 112, 5777–5780.
Groenewold, G. S.; Cossel, K. C.; Gresham, G. L.; Gianotto, A. K.; Appelhans, A. D.; Olson, J. E.; Van Stipdonk, M. J.; Chien, W. Binding of Molecular O2 to Di- and Triligated [UO2]+. J. Am. Chem. Soc. 2006, 128, 3075–3084.
Joaquim Marcalo, J.; Santos, M.; De Matos, A. P.; Gibson, J. K. Molecular Uranates: Laser Synthesis of Uranium Oxide Anions in the Gas Phase. Inorg. Chem. 2009, 48, 5055–5057.
Michelini, M. C.; Marcalo, J.; Russo, N.; Gibson, J. K. Gas-Phase Reactions of Uranate Ions, UO2 −, UO3 −, UO4 −, and UO4H−, with Methanol: A Convergence of Experiment and Theory. Inorg. Chem. 2010, 49, 3836–3850.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sokalska, M., Prussakowska, M., Hoffmann, M. et al. Unusual ion UO4 − formed upon collision induced dissociation of [UO2(NO3)3]−, [UO2(ClO4)3]−, [UO2(CH3COO)3]− ions. J Am Soc Mass Spectrom 21, 1789–1794 (2010). https://doi.org/10.1016/j.jasms.2010.06.018
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2010.06.018