Abstract
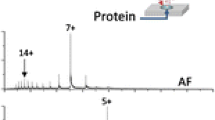
The effects of two supercharging reagents, m-nitrobenzyl alcohol (m-NBA) and sulfolane, on the charge-state distributions and conformations of myoglobin ions formed by electrospray ionization were investigated. Addition of 0.4% m-NBA to aqueous ammonium acetate solutions of myoglobin results in an increase in the maximum charge state from 9+ to 19+, and an increase in the average charge state from 7.9+ to 11.7+, compared with solutions without m-NBA. The extent of supercharging with sulfolane on a per mole basis is lower than that with m-NBA, but comparable charging was obtained at higher concentration. Arrival time distributions obtained from traveling wave ion mobility spectrometry show that the higher charge state ions that are formed with these supercharging reagents are significantly more unfolded than lower charge state ions. Results from circular dichroism spectroscopy show that sulfolane can act as chemical denaturant, destabilizing myoglobin by ∼1.5 kcal/mol/M at 25 °C. Because these supercharging reagents have low vapor pressures, aqueous droplets are preferentially enriched in these reagents as evaporation occurs. Less evaporative cooling will occur after the droplets are substantially enriched in the low volatility supercharging reagent, and the droplet temperature should be higher compared with when these reagents are not present. Protein unfolding induced by chemical and/or thermal denaturation in the electrospray droplet appears to be the primary origin of the enhanced charging observed for noncovalent protein complexes formed from aqueous solutions that contain these supercharging reagents, although other factors almost certainly influence the extent of charging as well.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Han, X. M.; Jin, M.; Breuker, K.; McLafferty, F. W. Extending Top-Down Mass Spectrometry to Proteins with Masses Greater Than 200 Kilodaltons. Science 2006, 314, 109–112.
Marshall, A. G.; Hendrickson, C. L.; Jackson, G. S. Fourier Transform Ion Cyclotron Resonance Mass Spectrometry: A Primer. Mass Spectrom. Rev. 1998, 17, 1–35.
Perry, R. H.; Cooks, R. G.; Noll, R. J. Orbitrap Mass Spectrometry: Instrumentation, Ion Motion and Applications. Mass Spectrom. Rev. 2008, 27, 661–699.
Smith, R. D.; Cheng, X.; Bruce, J. E.; Hofstadler, S. A.; Anderson, G. A. Trapping, Detection and Reaction of Very Large Single Molecular-Ions by Mass-Spectrometry. Nature 1994, 369, 137–139.
Bruce, J. E.; Cheng, X.; Bakhtiar, R.; Wu, Q.; Hofstadler, S. A.; Anderson, G. A.; Smith, R. D. Trapping, Detection, and Mass Measurement of Individual Ions in a Fourier-Transform Ion-Cyclotron Resonance Mass-Spectrometer. J. Am. Chem. Soc. 1994, 116, 7839–7847.
Chen, R. D.; Cheng, X. H.; Mitchell, D. W.; Hofstadler, S. A.; Wu, Q. Y.; Rockwood, A. L.; Sherman, M. G.; Smith, R. D. Trapping, Detection, and Mass Determination of Coliphage T4 DNA Ions of 10(8) Da by Electrospray-Ionization Fourier-Transform Ion-Cyclotron Resonance Mass-Spectrometry. Anal. Chem. 1995, 67, 1159–1163.
Fuerstenau, S. D.; Benner, W. H.; Thomas, J. J.; Brugidou, C.; Bothner, B.; Siuzdak, G. Mass Spectrometry of an Intact Virus. Angew. Chem. Int. Ed. 2001, 40, 542–544.
Chang, H. C. Ultrahigh-Mass Mass Spectrometry of Single Biomolecules and Bioparticles. Ann. Rev. Anal. Chem. 2009, 2, 169–185.
Fenn, J. B. Ion Formation from Charged Droplets — Roles of Geometry, Energy, and Time. J. Am. Soc. Mass Spectrom. 1993, 4, 524–535.
de la Mora, J. F. Electrospray Ionization of Large Multiply Charged Species Proceeds Via Dole’s Charged Residue Mechanism. Anal. Chim. Acta 2000, 406, 93–104.
Cech, N. B.; Enke, C. G. Practical Implications of Some Recent Studies in Electrospray Ionization Fundamentals. Mass Spectrom. Rev. 2001, 20, 362–387.
Cech, N. B.; Enke, C. G. Effect of Affinity for Droplet Surfaces on the Fraction of Analyte Molecules Charged During Electrospray Droplet Fission. Anal. Chem. 2001, 73, 4632–4639.
Iavarone, A. T.; Williams, E. R. Mechanism of Charging and Supercharging Molecules in Electrospray Ionization. J. Am. Chem. Soc. 2003, 125, 2319–2327.
Hogan, C. J.; Carroll, J. A.; Rohrs, H. W.; Biswas, P.; Gross, M. L. Charge Carrier Field Emission Determines the Number of Charges on Native State Proteins in Electrospray Ionization. J. Am. Chem. Soc. 2008, 130, 6926–6927.
Kebarle, P.; Verkerk, U. H. Electrospray: From Ions in Solution to Ions in the Gas Phase, What We Know Now. Mass Spectrom. Rev. 2009, 28, 898–917.
Grandori, R. Origin of the Conformation Dependence of Protein Charge-State Distributions in Electrospray Ionization Mass Spectrometry. J. Mass Spectrom. 2003, 38, 11–15.
Chowdhury, S. K.; Katta, V.; Chait, B. T. Probing Conformational Changes in Proteins by Mass Spectrometry. J. Am. Chem. Soc. 1990, 112, 9012–9013.
Konermann, L.; Douglas, D. J. Equilibrium Unfolding of Proteins Monitored by Electrospray Ionization Mass Spectrometry: Distinguishing Two-State from Multi-State Transitions. Rapid Commun. Mass Spectrom. 1998, 12, 435–442.
Wang, G. D.; Cole, R. B. Disparity Between Solution-Phase Equilibria and Charge-State Distributions in Positive-Ion Electrospray Mass Spectrometry. Org. Mass Spectrom. 1994, 29, 419–427.
Page, J. S.; Kelly, R. T.; Tang, K.; Smith, R. D. Ionization and Transmission Efficiency in an Electrospray Ionization-Mass Spectrometry Interface. J. Am. Soc. Mass Spectrom. 2007, 18, 1582–1590.
Iavarone, A. T.; Jurchen, J. C.; Williams, E. R. Effects of Solvent on the Maximum Charge State and Charge State Distribution of Protein Ions Produced by Electrospray Ionization. J. Am. Soc. Mass Spectrom. 2000, 11, 976–985.
Williams, E. R. Proton Transfer Reactivity of Large Multiply Charged Ions. J. Mass Spectrom. 1996, 31, 831–842.
Loo, R. R. O.; Smith, R. D. Proton-Transfer Reactions of Multiply-Charged Peptide and Protein Cations and Anions. J. Mass Spectrom. 1995, 30, 339–347.
Scalf, M.; Westphall, M. S.; Smith, L. M. Charge Reduction Electrospray Mass Spectrometry. Anal. Chem. 2000, 72, 52–60.
Stephenson, J. L.; McLuckey, S. A. Charge Manipulation for Improved Mass Determination of High-Mass Species and Mixture Components by Electrospray Mass Spectrometry. J. Mass Spectrom. 1998, 33, 664–672.
Iavarone, A. T.; Jurchen, J. C.; Williams, E. R. Supercharged Protein and Peptide Ions Formed by Electrospray Ionization. Anal. Chem. 2001, 73, 1455–1460.
Iavarone, A. T.; Williams, E. R. Supercharging in Electrospray Ionization: Effects on Signal and Charge. Int. J. Mass Spectrom. 2002, 219, 63–72.
Sze, S. K.; Ge, Y.; Oh, H.; McLafferty, F. W. Top-Down Mass Spectrometry of a 29-kDa Protein for Characterization of Any Post-Translational Modification to within One Residue. Proc. Natl. Acad. Sci. U.S.A. 2002, 99, 1774–1779.
Iavarone, A. T.; Williams, E. R. Collisionally Activated Dissociation of Supercharged Proteins Formed by Electrospray Ionization. Anal. Chem. 2003, 75, 4525–4533.
Davies, N. W.; Wiese, M. D.; Browne, S. G. A. Characterization of Major Peptides in ‘Jack Jumper’ Ant Venom by Mass Spectrometry. Toxicon 2004, 43, 173–183.
Kjeldsen, F.; Giessing, A. M. B.; Ingrell, C. R.; Jensen, O. N. Peptide Sequencing and Characterization of Post-Translational Modifications by Enhanced Ion-Charging and Liquid Chromatography Electron-Transfer Dissociation Tandem Mass Spectrometry. Anal. Chem. 2007, 79, 9243–9252.
Madsen, J. A.; Brodbelt, J. S. Comparison of Infrared Multiphoton Dissociation and Collision-Induced Dissociation of Supercharged Peptides in Ion Traps. J. Am. Soc. Mass Spectrom. 2009, 20, 349–358.
Grimm, R. L.; Beauchamp, J. L. Evaporation and Discharge Dynamics of Highly Charged Multicomponent Droplets Generated by Electrospray Ionization. J. Phys. Chem. A 2010, 114, 1411–1419.
Sterling, H. J.; Williams, E. R. Origin of Supercharging in Electrospray Ionization of Noncovalent Complexes from Aqueous Solution. J. Am. Soc. Mass Spectrom. 2009, 20, 1933–1943.
Lord, Rayleigh. Phils. Mag. 1882, 14, 184–186.
Dole, M.; Mack, L. L.; Hines, R. L. Molecular Beams of Macroions. J. Chem. Phys. 1968, 49, 2240–2249.
Iribarne, J. V.; Thomson, B. A. Evaporation of Small Ions from Charged Droplets. J. Chem. Phys. 1976, 64, 2287–2294.
CRC Handbook of Chemistry and Physics. In [Online] 89th ed.; D. R. Lide, Ed. 2008–2009.
Scherrenberg, R.; Coussens, B.; van Vliet, P.; Edouard, G.; Brackman, J.; de Brabander, E.; Mortensen, K. The Molecular Characteristics of Poly(Propyleneimine) Dendrimers as Studied with Small-Angle Neutron Scattering, Viscosimetry, and Molecular Dynamics. Macromolecules 1998, 31, 456–461.
Samalikova, M.; Grandori, R. Protein Charge-State Distributions in Electrospray-Ionization Mass Spectrometry Do Not Appear to be Limited by the Surface Tension of the Solvent. J. Am. Chem. Soc. 2003, 125, 13352–13353.
Samalikova, M.; Grandori, R. Testing the Role of Solvent Surface Tension in Protein Ionization by Electrospray. J. Mass Spectrom. 2005, 40, 503–510.
Samalikova, M.; Matecko, I.; Muller, N.; Grandori, R. Interpreting Conformational Effects in Protein Nano-ESI-MS Spectra. Anal. Bioanal. Chem. 2004, 378, 1112–1123.
Lomeli, S. H.; Peng, I. X.; Yin, S.; Loo, R. R. O.; Loo, J. A. New Reagents for Increasing ESI Multiple Charging of Proteins and Protein Complexes. J. Am. Soc. Mass Spectrom. 2010, 21, 127–131.
Lomeli, S. H.; Yin, S.; Loo, R. R. O.; Loo, J. A. Increasing Charge While Preserving Noncovalent Protein Complexes for ESI-MS. J. Am. Soc. Mass Spectrom. 2009, 20, 593–596.
Clemmer, D. E.; Jarrold, M. F. Ion Mobility Measurements and Their Applications to Clusters and Biomolecules. J. Mass Spectrom. 1997, 32, 577–592.
Wyttenbach, T.; Bowers, M. T. In Modern Mass Spectrometry; Vol. 225; Springer-Verlag: Berlin, 2003; p. 207–232.
Kanu, A. B.; Dwivedi, P.; Tam, M.; Matz, L.; Hill, H. H. Ion Mobility-Mass Spectrometry. J. Mass Spectrom. 2008, 43, 1–22.
Giles, K.; Pringle, S. D.; Worthington, K. R.; Little, D.; Wildgoose, J. L.; Bateman, R. H. Applications of a Traveling Wave-Based Radio-Frequency Only Stacked Ring Ion Guide. Rapid Commun. Mass Spectrom. 2004, 18, 2401–2414.
Giles, K.; Williams, J. P.; Pringle, S. D.; Wildgoose, J. L.; Slade, S. E.; Thalassinos, K.; Bateman, R. H.; Bowers, M. T.; Scrivens, J. H. An Investigation of the Mobility Separation of Some Peptide and Protein Ions Using a New Hybrid Quadrupole/Traveling Wave IMS/oa-TOF Instrument. Int. J. Mass Spectrom. 2007, 261, 1–12.
Ruotolo, B. T.; Giles, K.; Campuzano, I.; Sandercock, A. M.; Bateman, R. H.; Robinson, C. V. Evidence for Macromolecular Protein Rings in the Absence of Bulk Water. Science 2005, 310, 1658–1661.
Ruotolo, B. T.; Hyung, S. J.; Robinson, P. M.; Giles, K.; Bateman, R. H.; Robinson, C. V. Ion Mobility-Mass Spectrometry Reveals Long-Lived, Unfolded Intermediates in the Dissociation of Protein Complexes. Angew. Chem. Int. Ed. 2007, 46, 8001–8004.
Lorenzen, K.; Olia, A. S.; Uetrecht, C.; Cingolani, G.; Heck, A. J. R. Determination of Stoichiometry and Conformational Changes in the First Step of the P22 Tail Assembly. J. Mol. Biol. 2008, 379, 385–396.
Scarff, C. A.; Patel, V. J.; Thalassinos, K.; Scrivens, J. H. Probing Hemoglobin Structure by Means of Traveling-Wave Ion Mobility Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2009, 20, 625–631.
Hopper, J. T. S.; Oldham, N. J. Collision Induced Unfolding of Protein Ions in the Gas Phase Studied by Ion Mobility-Mass Spectrometry: The Effect of Ligand Binding on Conformational Stability. J. Am. Soc. Mass Spectrom. 2009, 20, 1851–1858.
Shelimov, K. B.; Clemmer, D. E.; Hudgins, R. R.; Jarrold, M. F. Protein Structure in Vacuo: Gas-Phase Confirmations of BPTI and Cytochrome c. J. Am. Chem. Soc. 1997, 119, 2240–2248.
Shvartsburg, A. A.; Smith, R. D. Fundamentals of Traveling Wave Ion Mobility Spectrometry. Anal. Chem. 2008, 80, 9689–9699.
Shelimov, K. B.; Jarrold, M. F. Conformations, Unfolding, and Refolding of Apomyoglobin in Vacuum: An Activation Barrier for Gas-Phase Protein Folding. J. Am. Chem. Soc. 1997, 119, 2987–2994.
Van Holde, K. E.; Johnson, W. C.; Ho, P. Principles of Physical Biochemistry. Prentice-Hall, Inc.: Upper Saddle River, NJ, 1998; p. 434.
Chanthai, S.; Ogawa, M.; Tamiya, T.; Tsuchiya, T. Studies on Thermal Denaturation of Fish Apomyoglobins Using Differential Scanning Calorimetry, Circular Dichroism, and Fluorescence. Fisheries Sci. 1996, 62, 933–937.
Awad, E. S.; Deranleau, D. A. Thermal Denaturation of Myoglobin. I. Kinetic Resolution of Reaction Mechanism. Biochemistry 1968, 7, 1791–1795.
Konermann, L.; Douglas, D. J. Unfolding of Proteins Monitored by Electrospray Ionization Mass Spectrometry: A Comparison of Positive and Negative Ion Modes. J. Am. Soc. Mass Spectrom. 1998, 9, 1248–1254.
Kelly, M. A.; Vestling, M. M.; Fenselau, C. C.; Smith, P. B. Electrospray Analysis of Proteins—a Comparison of Positive-Ion and Negative-Ion Mass-Spectra at High and Low pH. Org. Mass Spectrom. 1992, 27, 1143–1147.
Sterling, H. J.; Williams, E. R. Proceedings of the 58th ASMS Conference on Mass Spectrometry and Allied Topics; Salt Lake City, UT, May, 2010.
Chanthai, S.; Ogawa, M.; Tamiya, T.; Tsuchiya, T. Studies on Thermal Denaturation Profiles of Holo- and Reconstituted Myoglobins from Bonito and Sperm Whale. Fisheries Sci. 1998, 64, 411–414.
Pflumm, M.; Luchins, J.; Beychok, S. Stopped-Flow Circular-Dichroism. Methods Enzymol. 1986, 130, 519–534.
Chanthai, S.; Ogawa, M.; Tamiya, T.; Tsuchiya, T. Studies on Thermal Denaturation of Fish Myoglobins Using Differential Scanning Calorimetry, Circular Dichroism, and Tryptophan Fluorescence. Fisheries Sci. 1996, 62, 927–932.
Giacometti, G. M.; Antonini, E.; Brunori, M. Kinetics of Reversible Protein Denaturation — Study on Aplysia Myoglobin. Biophys. Chem. 1979, 10, 119–127.
Abbruzzetti, S.; Sottini, S.; Viappiani, C.; Corrie, J. E. T. Acid-Induced Unfolding of Myoglobin Triggered by a Laser pH Jump Method. Photochem. Photobiol. Sci. 2006, 5, 621–628.
Shen, L. L.; Hermans, J. Kinetics of Conformation Change of Sperm-Whale Myoglobin. 1. Folding and Unfolding of Metmyoglobin Following pH Jump. Biochemistry 1972, 11, 1836–1841.
Goodman, J. M.; Kirby, P. D.; Haustedt, L. O. Some Calculations for Organic Chemists: Boiling Point Variation, Boltzmann Factors and the Eyring Equation. Tetrahedron Lett. 2000, 41, 9879–9882.
Benkestock, K.; Sundqvist, G.; Edlund, P. O.; Roeraade, J. Influence of Droplet Size, Capillary-Cone Distance and Selected Instrumental Parameters for the Analysis of Noncovalent Protein-Ligand Complexes by Nano-Electrospray Ionization Mass Spectrometry. J. Mass Spectrom. 2004, 39, 1059–1067.
Charlebois, J. P.; Patrie, S. M.; Kelleher, N. L. Electron Capture Dissociation and C-13, N-15 Depletion for Deuterium Localization in Intact Proteins after Solution-Phase Exchange. Anal. Chem. 2003, 75, 3263–3266.
Pan, J.; Han, J.; Borchers, C. H.; Konermann, L. Electron Capture Dissociation of Electrosprayed Protein Ions for Spatially Resolved Hydrogen Exchange Measurements. J. Am. Chem. Soc. 2008, 130, 11574–11575.
Rand, K. D.; Adams, C. M.; Zubarev, R. A.; Jørgensen, T. J. D. Electron Capture Dissociation Proceeds with a Low Degree of Intramolecular Migration of Peptide Amide Hydrogens. J. Am. Chem. Soc. 2008, 130, 1341–1349.
Pan, J. X.; Han, J.; Borchers, C. H.; Konermann, L. Hydrogen/Deuterium Exchange Mass Spectrometry with Top-Down Electron Capture Dissociation for Characterizing Structural Transitions of a 17 kDa Protein. J. Am. Chem. Soc. 2009, 131, 12801–12808.
Zehl, M.; Rand, K. D.; Jensen, O. N.; Jorgensen, T. J. D. Electron Transfer Dissociation Facilitates the Measurement of Deuterium Incorporation into Selectively Labeled Peptides with Single Residue Resolution. J. Am. Chem. Soc. 2008, 130, 17453–17459.
Abzalimov, R. R.; Kaplan, D. A.; Easterling, M. L.; Kaltashov, I. A. Protein Conformations Can Be Probed in Top-Down HDX MS Experiments Utilizing Electron Transfer Dissociation of Protein Ions without Hydrogen Scrambling. J. Am. Soc. Mass Spectrom. 2009, 20, 1514–1517.
Rand, K. D.; Zehl, M.; Jensen, O. N.; Jorgensen, T. J. D. Protein Hydrogen Exchange Measured at Single-Residue Resolution by Electron Transfer Dissociation Mass Spectrometry. Anal. Chem. 2009, 81, 5577–5584.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sterling, H.J., Daly, M.P., Feld, G.K. et al. Effects of supercharging reagents on noncovalent complex structure in electrospray ionization from aqueous solutions. J Am Soc Mass Spectrom 21, 1762–1774 (2010). https://doi.org/10.1016/j.jasms.2010.06.012
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2010.06.012