Abstract
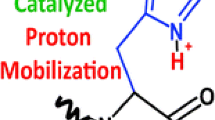
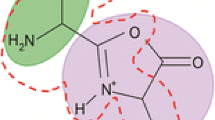
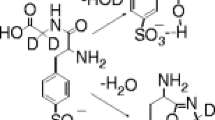
In this study, the possible influence of acidic, basic, and amide side chains on the opening of a putative macrocyclic b ion (b +5 ) intermediate was investigated. Collision induced dissociation (CID) of b 5 ions was studied using a group of hexapeptides in which amino acids with the side chains of interest occupied internal sequence positions. Further experiments were performed with permuted isomers of glutamine (Q) containing peptides to probe for sequence scrambling and whether the specific sequence site of the residues influences opening of the macrocycle. Overall, the trend for (apparent) preferential/selective opening of the cyclic b +5 , presumably due to the side chain, followed by the loss of the amino acid with active side group is: Q > K > D > N ∼ E.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hunt, D. F.; Yates, J. R. III; Shabanonowitz J.; Winston, S.; Hauer, C. R. Protein Sequencing by Tandem Mass Spectrometry. Proc. Natl. Acad. Sci. 1986, 83, 6233–6237.
Biemann, K. Contributions of Mass Spectrometry to Peptide and Protein Structure. Biomed. Environ. Mass Spectrom. 1988, 16, 99–111.
Steen, H.; Mann, M. The abc’s and the xyz’s of Peptide Sequencing. Nat. Rev. Mol. Cell. Biol. 2004, 5, 699–711.
Nesvizhskii, A. E.; Vitek, O.; Aebersold, R. Analysis and Validation of Proteomic Data Generated by Tandem Mass Spectrometry. Nat. Methods 2007, 4, 787–797.
Roepstroff, P.; Fohlmann, J. Proposal for a Common Nomenclature for Sequence Ions in Mass Spectra of Peptides. J. Biomed. Mass Spectrom. 1984, 11, 601.
Papayannopoulos, I. A. The Interpretation of Collision-Induced Dissociation Tandem Mass Spectra of Peptides. 1995, 14, 49–73.
Dongre, A. R.; Jones, J. L.; Somogyi, A.; Wysocki, V. H. Influence of Peptide Composition, Gas-Phase Basicity, and Chemical Modification on Fragmentation Efficiency: Evidence for the Mobile Proton Model. J. Am. Chem. Soc. 1996, 118, 8365–8374.
Wysocki, V. H.; Tsaprailis, G.; Smith, L. L.; Breci, L. A. Mobile and Localized Protons: A Framework for Understanding Peptide Dissociation. J. Mass Spectrom. 2000, 35, 1399–1406.
Yalcin, T.; Csizmadia, I. G.; Peterson, M. B.; Harrison, A. Why are b Ions Stable Species in Peptide Spectra? J. Am. Soc. Mass Spectrom. 1996, 7, 233–242.
Paizs, B.; Lendvay, G.; Vekey, K.; Suhai, S. Formation of b + 2 Ions from Protonated Peptides: An Ab Initio Study. Rapid Commun. Mass Spectrom. 1999, 13, 525–533.
Paizs, B.; Suhai, S. Towards Understanding the Tandem Mass Spectra of Protonated Oligopeptides. 1: Mechanism of Amide Bond Cleavage. J. Am. Soc. Mass Spectrom. 2004, 15, 103–113.
Paizs, B.; Suhai, S. Combined Quantum Chemical and RRKM Modeling of the Main Fragmentation Pathways of Protonated GGG. II. Formation of b2, y1, and y2 ions. Rapid Commun. Mass Spectrom. 2002, 16, 375–389.
Polce, M. J.; Ren, D.; Wesdemiotis, C. Dissociation of the Peptide Bond in Protonated Peptides. J. Mass Spectrom. 2000, 35(12), 1391–1398.
Paizs, B.; Suhai, S. Fragmentation Pathways of Protonated Peptides. Mass Spectrom. Rev. 2004, 24, 508–548.
Yalcin, T.; Khouw, C.; Csizmadia, I. G.; Peterson, M. R.; Harrison, A. G. The Structure and Fragmentation of b n (n < 3) Ions in Peptide Spectra. J. Am. Soc. Mass Spectrom. 1995, 6, 1165–1174.
Harrison, A. G.; Young, A. B.; Bleiholder, B.; Suhai, S.; Paizs, B. Scrambling of Sequence Information in Collision-Induced Dissociation of Peptides. J. Am. Chem. Soc. 2006, 128, 10364–10365.
Riba-Garcia, F.; Giles, K.; Bateman, R. H.; Gaskell, S. J. Evidence for Structural Variants of a-and b-Type Peptide Fragment Ions Using Combined Ion Mobility/Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2008, 19, 609–613.
Bleiholder, C.; Osburn, S.; Williams, T. D.; Suhai, S.; Van Stipdonk, M.; Harrison, A. G.; Paizs, B. Sequence Scrambling Fragmentation Pathways of Protonated Peptides. J. Am. Chem. Soc. 2008, 130, 17774–17789.
Jia, C.; Qi, W.; He, Z. Cyclization Reaction of Peptide Fragment Ions during Multistage Collisionally Activated Decomposition: An Inducement to Lose Internal Amino-Acid Residues. J. Am. Soc. Mass Spectrom. 2007, 18, 663–667.
Farrugia, J. M.; O’Hair, R. A. J.; Reid, G. E.; Do All b2 Ions Have Oxazolone Structures? Multistage Mass Spectrometry and Ab Initio Studies on Protonated N-Acyl Amino Acid Methyl Ester Model Systems. Int. J. Mass Spectrom. 2001, 210/211, 71–87.
Yalcin, T.; Harrison, A. G. Ion Chemistry of Protonated Lysine Derivatives. J. Mass Spectrom. 1996, 31, 1237–1243.
Kish, M. M.; Wesdemiotis, C. Selective Cleavage at Internal Lysine Residues in Protonated Versus Metalated Peptides. Int. J. Mass Spectrom. 2003, 227, 191–203.
Farrugia, J. M.; O’Hair, R. A. J. Involvement of Salt Bridges in a Novel Gas Phase Rearrangement of Protonated Arginine-Containing Dipeptides That Precedes Fragmentation. Int. J. Mass Spectrom. 2003, 222, 229–242.
Paizs, B.; Suhai, S.; Hargittai, B.; Hruby, V. J.; Somogyi, Á. Ab Initio and MS/MS Studies on Protonated Peptides Containing Basic and Acidic Amino Acid Residues: I. Solvated proton Versus Salt-Bridged Structures and the Cleavage of the Terminal Amide Bond of Protonated RD-NH2. Int. J. Mass Spectrom. 2002, 219, 203–232.
Jonsson, A. P.; Bergman, T.; Jörnvall, H.; Griffiths, W. J.; Bratt, P.; Strömberg, N. Gln-Gly Cleavage: Correlation Between CollisionInduced Dissociation and Biological Degradation. J. Am. Soc. Mass Spectrom. 2001, 12, 337–342.
Lee, Y. J.; Lee, Y. M. Formation of c1 Fragment Ions in Collision-Induced Dissociation of Glutamine-Containing Peptide Ions: A Tip for De Novo Sequencing. Rapid Commun. Mass Spectrom. 2004, 18, 2069–2076.
Harrison, A. G. Fragmentation Reactions of Protonated Peptides Containing Glutamine or Glutamic Acid. JMS 2003, 38, 174–187.
Baldwin, M. A.; Falick, A. M.; Gibson, B. W.; Prusiner, S. B.; Stahl, N.; Burlingame, A. L. Tandem Mass Spectrometry of Peptides with N-Terminal Glutamine: Studies on a Prion Protein Peptide. J. Am. Soc. Mass Spectrom. 1990, 1, 258–264.
Rozman, M. Aspartic Acid Side Chain Effect—Experimental and Theoretical Insight. J. Am. Soc. Mass Spectrom. 2007, 18, 121–127.
Gu, C.; Tsaprailis, G.; Breci, L.; Wysocki, V. H. Selective Gas-Phase Cleavage at the Peptide Bond C-Terminal to Aspartic Acid in Fixed-Charge Derivatives of Asp-Containing Peptides. Anal. Chem. 2000, 72, 5904–5813.
Tsaprailis, G.; Somogyi, Á.; Nikolaev, E. N.; Wysocki, V. H. Refining the Model for Selective Cleavage at Acidic Residues in Arginine-Containing Protonated Peptides. Int. J. Mass Spectrom. 2000, 195/196, 467–479.
Herrmann, K. A.; Wysocki, V. H.; Vorpagel, E. R. Computational Investigation and Hydrogen/Deuterium Exchange of the Fixed Charge Derivative Tris(2,4,6-Trimethoxyphenyl) Phosphonium: Implications for the Aspartic Acid Cleavage Mechanism. J. Am. Soc. Mass Spectrom. 2005, 16, 1067–1080.
Lee, S.; Kim, H. S.; Beauchamp, J. L. Salt Bridge Chemistry Applied to Gas-Phase Peptide Sequencing: Selective Fragmentation of Sodiated Gas-Phase Peptide Ions Adjacent to Aspartic Acid Residues. J. Am. Chem. Soc. 1998, 120, 3188–3195.
Gonzalez, L. J.; Shimizu, T.; Satomi, Y.; Betancourt, L.; Besada1, V.; Padron, G.; Orlando, R.; Shirasawa, T.; Shimonishi, Y.; Takao, T. Differentiating α- and β-Aspartic Acids by Electrospray Ionization and Low-Energy Tandem Mass Spectrometry. Rapid Commun. Mass Spectrom. 2000, 14, 2092–2102.
Yu, W.; Vath, J. E.; Huberty, M. C.; Martin, S. A. Identification of the Facile Gas-Phase Cleavage of the Asp-Pro and Asp-Xxx Peptide Bonds in Matrix-Assisted Laser Desorption Time-of-Flight. Mass Spectrom. Anal. Chem. 1993, 65, 3015–3023.
Price, W. D.; Schnier, P. D.; Jockusch, R. A.; Strittmatter, E. F.; Williams, E. R. Unimolecular Reaction Kinetics in the High-Pressure Limit without Collisions. J. Am. Chem. Soc. 1996, 118, 10640–10644.
Sun, W.; Kinsel, G. R.; Marynick, D. S. Computational Estimates of the Gas-Phase Basicity and Proton Affinity of Glutamic Acid. J. Phys. Chem. A 1999, 103, 4113–4117.
Chan, W. C.; White, P. D. Fmoc Solid Phase Peptide Synthesis—a Practical Approach; Oxford University Press: New York, 2000.
Molesworth, S.; Osburn, S.; Van Stipdonk, M. Influence of Size on Apparent Scrambling of Sequence During CID of b-Type Ions. J. Am. Soc. Mass Spectrom. 2009, 20, 2174–2184.
Molesworth, S.; Leavitt, C. M.; Groenewold, G. S.; Oomens, J.; Steill, J.; Van Stipdonk, M. Spectroscopic Evidence for Mobilization of Amide Position Protons During CID of Model Peptide Ions. J. Am. Soc. Mass Spectrom. 2009, 20, 1841–1845.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Molesworth, S., Osburn, S. & Van Stipdonk, M. Influence of amino acid side chains on apparent selective opening of cyclic b 5 ions. J Am Soc Mass Spectrom 21, 1028–1036 (2010). https://doi.org/10.1016/j.jasms.2010.02.011
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2010.02.011