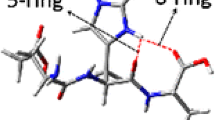
Abstract
The gas-phase H/D exchange reactions of arginine (R) and arginine-containing di- and tri-peptide (gly-arg (GR), arg-gly (RG), gly-gly-arg (GGR), gly-arg-gly (GRG) and arg-gly-gly (RGG)) [M+H]+ ions with deuterated ammonia (ND3) were investigated by using Fourier-transform ion cyclotron resonance mass spectrometry (FT-ICR), ion mobility-mass spectrometry (IM-MS), ab initio and density functional theory-based molecular orbital calculations and molecular modeling. Three exchanges are observed for arginine and arginine-containing tri-peptide [M+H]+ ions, whereas the di-peptide [M+H]+ ions undergo a single H/D exchange. In addition, C-terminal methylation blocks H/D exchange of arginine and the arginine-containing peptide [M+H]+ ions, and a single H/D exchange is observed for N-terminal acetylated arginine [M+H]+ ions. A general mechanism for H/D exchange involving a collision complex that is best described as a “solvated salt-bridge” structure is proposed.
Article PDF
Similar content being viewed by others
References
Englander, S. W. Hydrogen Exchange and Mass Spectrometry: A Historical Perspective. J. Am. Soc. Mass Spectrom. 2006, 17, 1481–1489.
Kjellsson, A.; Sethson, I.; Jonsson, B.-H. Hydrogen Exchange in a Large 29 kD Protein and Characterization of Molten Globule Aggregation by NMR. Biochemistry 2003, 42, 363–374.
Deng, Y.; Zhang, Z.; Smith, D. L. Comparison of Continuous and Pulsed Labeling Amide Hydrogen Exchange/Mass Spectrometry for Studies of Protein Dynamics. J. Am. Soc. Mass Spectrom. 1999, 10, 675–684.
Weis, D. D.; Wales, T. E.; Engen, J. R.; Hotchko, M.; Eyck, L. F. T. Identification and Characterization of EX1 Kinetics in H/D Exchange Mass Spectrometry by Peak Width Analysis. J. Am. Soc. Mass Spectrom. 2006, 17, 1498–1509.
Yan, X.; Deinzer, M. L.; Schimerlik, M. I.; Broderick, D.; Leid, M. E.; Dawson, M. I. Investigation of Ligand Interactions with Human RXRα by Hydrogen/Deuterium Exchange and Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2006, 17, 1510–1517.
Cheng, G.; Wysocki, V. H.; Cusanovich, M. A. Local Stability of Rhodobacter capsulatus Cytochrome c 2 Probed by Solution Phase Hydrogen/Deuterium Exchange and Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2006, 17, 1518–1525.
Chitta, R. K.; Rempel, D. L.; Grayson, M. A.; Remsen, E. E.; Gross, M. L. Application of SIMSTEX to Oligomerization of Insulin Analogs and Mutants. J. Am. Soc. Mass Spectrom. 2006, 17, 1526–1534.
Dai, S. Y.; Fitzgerald, M. C. Accuracy of SUPREX (Stability of Unpurified Proteins from Rates of H/D Exchange) and MALDI Mass Spectrometry-Derived Protein Unfolding Free Energies Determined Under Non-EX2 Exchange Conditions. J. Am. Soc. Mass Spectrom. 2006, 17, 1535–1542.
Abzalimov, R. R.; Kaltashov, I. A. Extraction of Local Hydrogen Exchange Data from HDX CAD MS Measurements by Deconvolution of Isotopic Distributions of Fragment Ions. J. Am. Soc. Mass Spectrom. 2006, 17, 543–1551.
Truhlar, S. M. E.; Croy, C. H.; Torpey, J. W.; Koeppe, J. R.; Komives, E. A. Solvent Accessibility of Protein Surfaces by Amide H/2H Exchange MALDI-TOF Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2006, 17, 1490–1497.
Katta, V.; Chait, B. T. Hydrogen/Deuterium Exchange Electrospray Ionization Mass Spectrometry: A Method for Probing Protein Conformational Changes in Solution. J. Am. Chem. Soc. 1993, 115, 6317–6321.
Figueroa, I. D.; Russell, D. H. Matrix-Assisted Laser Desorption Ionization Hydrogen/Deuterium Exchange Studies to Probe Peptide Conformational Changes. J. Am. Soc. Mass Spectrom. 1999, 10, 719–731.
Winger, B. E.; Light-Wahl, K. J.; Rockwood, A. L.; Smith, R. D. Probing Qualitative Conformation Differences of Multiply Protonated Gas-Phase Proteins Via Hydrogen/Deuterium Isotopic Exchange with Water-d2. J. Am. Chem. Soc. 1992, 114, 5897–5898.
Schaaff, T. G.; Stephenson, J. L.; McLuckey, S. A. Gas Phase H/D Exchange Kinetics: DI Versus D2O. J. Am. Soc. Mass Spectrom. 2000, 11, 167–171.
Freitas, M. A.; Marshall, A. G. Rate and Extent of Gas-Phase Hydrogen/Deuterium Exchange of Bradykinins: Evidence for Peptide Zwitterions in the Gas Phase. Int. J. Mass Spectrom. 1999, 182/183, 221–231.
Valentine, S. J.; Clemmer, D. E. Temperature-Dependent H/D Exchange of Compact and Elongated Cytochrome c Ions in the Gas Phase. J. Am. Soc. Mass Spectrom. 2002, 13, 506–517.
Valentine, S. J.; Clemmer, D. E. H/D Exchange Levels of Shape-Resolved Cytochrome c Conformers in the Gas Phase. J. Am. Chem. Soc. 1997, 119, 3558–3566.
Dookeran, N. N.; Harrison, A. G. Gas-phase H-D Exchange Reactions of Protonated Amino Acids and Peptides with ND3. J. Mass Spectrom. 1995, 30, 666–674.
Grad, E.; Willard, D.; Bregar, J.; Green, M. K.; Lebrilla, C. B. Site Specificity in the H-D Exchange Reactions of Gas-Phase Protonated Amino Acids with CH3OD. Org. Mass Spectrom. 1993, 28, 1632–1639.
KovaĈević, B.; Rožman, M.; Klasinc, L.; Srzić, D.; Maksić, Z. B.; Yáñez, M. Gas-Phase Structure of Protonated Histidine and Histidine Methyl Ester: Combined Experimental Mass Spectrometry and Theoretical ab Initio Study. J. Phys. Chem. A 2005, 109, 8329–8335.
Campbell, S.; Rodgers, M. T.; Marzluff, E. M.; Beauchamp, J. L. Deuterium Exchange Reactions as a Probe of Biomolecule Structure: Fundamental Studies of Gas Phase H/D Exchange Reactions of Protonated Glycine Oligomers with D2O, CD3OD, CD3CO2D, and ND3. J. Am. Chem. Soc. 1995, 117, 12840–12854.
Lee, S.-W.; Lee, H.-N.; Kim, H.-S.; Beauchamp, J. L. Selective Binding of Crown Ethers to Protonated Peptides Can Be Used to Probe Mechanisms of H/D Exchange and Collision-Induced Dissociation Reactions in the Gas Phase. J. Am. Chem. Soc. 1998, 120, 5800–5805.
Linstrom, P. J.; Mallard, W. G. Eds., NIST Chemistry Web Book, NIST Stantdard Reference Database Number 69; June 2005, National Institute of Standard and Technology: Gaithersburg MD, 20889 (http://webbook.nist.gov).
Zhu, C.; Balta, B.; Aviyente, V.; Lifshitz, C. The Interaction of Protonated Diglycine with Ammonia: A Density Functional Theory Model Study. J. Phys. Chem. A 2000, 104, 7061–7067.
Cox, H. A.; Julian, R. R.; Lee, S.; Beauchamp, J. L. Gas-Phase H/D Exchange of Sodiated Glycine Oligomers with ND3: Exchange Kinetics Do Not Reflect Parent Ion Structures. J. Am. Chem. Soc. 2004, 126, 6485–6490;
Geller, O.; Lifshitz, C. Gas Phase H/D Exchange of Protonated Arginine Monomers and Dimers. J. Phys. Chem. A. 2003, 107, 5654–5659.
Wyttenbach, T.; Bowers, M. T. Gas Phase Conformations of Biological Molecules: The Hydrogen/Deuterium Exchange Mechanism. J. Am. Soc. Mass Spectrom. 1999, 10, 9–14.
Wyttenbach, T.; Paizs, B.; Barran, P.; Breci, L.; Liu, D.; Suhai, S.; Wysocki, V. H.; Bowers, M. T. The Effect of the Initial Water of Hydration on the Energetics, Structures, and H/D Exchange Mechanism of a Family of Pentapeptides: An Experimental and Theoretical Study. J. Am. Chem. Soc. 2003, 125, 13768–13775.
Strittmatter, E. F.; Williams, E. R. Structures of Protonated Arginine Dimer and Bradykinin Investigated by Density Functional Theory: Further Support for Stable Gas-Phase Salt Bridges. J. Phys. Chem. A 2000, 104, 6069–6076.
Jockusch, R. A.; Price, W. D.; Williams, E. R. Structure of Cationized Arginine (Arg·M+, M=H, Li, Na, K, Rb, and Cs) in the Gas Phase: Further Evidence for Zwitterionic Arginine. J. Phys. Chem. A 1999, 103, 9266–9274.
Schnier, P. D.; Price, W. D.; Jockusch, R. A.; Williams, E. R. Blackbody Infrared Radiative Dissociation of Bradykinin and Its Analogues: Energetics, Dynamics, and Evidence for Salt-Bridge Structures in the Gas Phase. J. Am. Chem. Soc. 1996, 118, 7178–7189.
Rak, J.; Skurski, P.; Simons, J.; Gutowski, M. Low-Energy Tautomers and Conformers of Neutral and Protonated Arginine. J. Am. Chem. Soc. 2001, 123, 11695–11707.
Julian, R. R.; Hodyss, R.; Beauchamp, J. L. Salt Bridge Stabilization of Charged Zwitterionic Arginine Aggregates in the Gas Phase. J. Am. Chem. Soc. 2001, 123, 3577–3583.
Price, W. D.; Jockusch, R. A.; Williams, E. R. Is Arginine a Zwitterion in the Gas Phase?. J. Am. Chem. Soc. 1997, 119, 11988–11989.
Chapo, C. J.; Paul, J. B.; Provencal, R. A.; Roth, K.; Saykally, R. J. Is Arginine Zwitterionic or Neutral in the Gas Phase?: Results from IR Cavity Ringdown Spectroscopy. J. Am. Chem. Soc. 1998, 120, 12956–12957.
Julian, R. R.; Beauchamp, J. L.; Goddard, III W. A. Cooperative Salt Bridge Stabilization of Gas-Phase Zwitterions in Neutral Arginine Clusters. J. Phys. Chem. A 2002, 106, 32–34.
Kerley, E. L.; Russell, D. H. Mass and Energy Selective Ion Partitioning in a Two-Section Fourier-Transform Ion Cyclotron Resonance Spectrometer Cell. Anal. Chem. 1989, 61, 53–57.
Bartmess, J. E.; Georgiadis, R. M. Empirical Method for the Determination of Ion Gauge Sensitivities to Different Gases. Vacuum 1983, 33, 149–153.
Chen, L.; Wang, T.-C. L.; Ricca, T. L.; Marshall, A. G. Phase-Modulated Stored Waveform Inverse Fourier Transform Excitation for Trapped Ion Mass Spectrometry. Anal. Chem. 1987, 59, 449–454.
Su, T.; Chesnavich, W. J. Parameterization of the Ion-Polar Molecule Collision Rate Constant by Trajectory Calculations. J. Chem. Phys. 1982, 76, 5183–5185;
Chesnavich, W. J.; Su, T.; Bowers, M. T. Collisions in a Noncentral Field: A Variational and Trajectory Investigation of Ion-Dipole Capture. J. Chem. Phys. 1980, 72, 2641–2655.
Hettick, J. M.; Kashon, M. L.; Simpson, J. P.; Siegel, P. D.; Mazurek, G. H.; Weissman, D. N. Proteomic Profiling of Intact Mycobacteria by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. Anal. Chem. 2004, 76(19), 5769–5776.
Reid, G. E.; Simpson, R. J.; O’Hair, R. A. J. A Mass Spectrometric and ab Initio Study of the Pathways for Dehydration of Simple Glycine and Cysteine-Containing Peptide [M+H]+ Ions. J. Am. Soc. Mass Spectrom. 1998, 9(9), 945–956.
McLean, J. A.; Ruotolo, B. T.; Gillig, K. J.; Russell, D. H. Ion Mobility-Mass Spectrometry: A New Paradigm for Proteomics. Int. J. Mass Spectrom. 2005, 240, 301–315.
Frisch, M. J.; Gaussian, Inc.: Pittsburgh, PA, 2001.
It is important to note that in some areas of the potential energy surface calculations, a specific transition state is not directly associated with the given complex. That is, some geometric rearrangements rotation of ammonia relative to the peptide occurs within the complex before the system experiences the transition state. In some cases, such rearrangements lower the energy of the transition state relative to the complex. In those cases, it is assumed that the transition state is associated with a reactant-like and a product-like complex, both with energy lower than the transition state, but their geometries are unknown. In addition, if the energy barrier is insignificant, the zero point correction can eliminate the energy barrier due to the relatively small size of the basis set.
Mason, E. A.; McDaniels, E. W. Transport Properties of Ions in Gases, John Wiley and Sons: New York, 1988; pp. 230–235.
Sawyer, H. A.; Marini, J. T.; Stone, E. G.; Ruotolo, B. T.; Gillig, K. J.; Russell, D. H. The Structure of Gas-Phase Bradykinin Fragment 1–5 (RPPGF) Ions: An Ion Mobility Spectrometry and H/D Exchange Ion-Molecule Reaction Chemistry Study. J. Am. Soc. Mass Spectrom. 2005, 16(6), 893–905.
Ruotolo, B. T.; Gillig, K. J.; Woods, A. S.; Egan, T. F.; Ugarov, M. V.; Schultz, J. A.; Russell, D. H. Analysis of Phosphorylated Peptides by Ion Mobility-Mass Spectrometry. Anal. Chem. 2004, 76, 6727–6733.
Ruotolo, B. T.; Verbeck, I. V.; Thomson, L. M.; Woods, A. S.; Gillig, K. J.; Russell, D. H. Distinguishing Between Phosphorylated and Nonphosphorylated Peptides by Ion Mobility-Mass Spectrometry. J. Proteome Res. 2002, 1, 303–306.
Shvartsburg, A. A.; Jarrold, M. F. An Exact Hard-Spheres Scattering Model for the Mobilities of Polyatomic Ions. Chem. Phys. Lett. 1996, 261, 86–91.
Marini, J. T. Development and Implementation of FT- ICR Mass Spectrometry for the Investigation of Peptide Ion Conformations of Peptide Sequence Isomers Containing Basic Amino Acid Residues by Gas-Phase Hydrogen/Deuterium Exchange; Ph. D. Dissertation, Texas A and M University: College Station, TX, 2003.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online July 30, 2009
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Huang, Y., Marini, J.A., McLean, J.A. et al. A mechanistic study of the H/D exchange reactions of protonated arginine and arginine-containing Di- and tripeptides. J Am Soc Mass Spectrom 20, 2049–2057 (2009). https://doi.org/10.1016/j.jasms.2009.07.015
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2009.07.015