Abstract
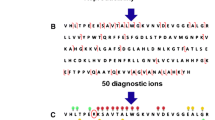
Population migration has led to the global dispersion of human hemoglobinopathies and has precipitated a need for their identification. An effective mass spectrometry-based procedure involves analysis of the intact α- and β-globin chains to determine their mass, followed by location of the variant amino acid residue by direct analysis of the enzymatically digested chains and low-energy collision induced dissociation of the variant peptide. Using this procedure, a variant was identified as either β54Val→Leu or β54Val→Ile, since the amino acids leucine and isoleucine cannot be distinguished using low-energy collisions. Here, we describe how hot electron capture dissociation on a Fourier transform-ion cyclotron resonance mass spectrometer was used to distinguish isoleucine from leucine and identify the mutation as β54(D5)Val→Ile. This is a novel variant, and we have named it Hb Askew.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hardison, R. C.; Chui, D. H. K.; Giardine, B.; Riemer, C.; Patrinos, G. P.; Anagnou, N.; Millar, W. HbVar: A relational database of human hemoglobin variants and thalassemia mutations at the globin gene server. Human Mutat. 2002, 19, 225–233. (http://globin.cse.psu.edu).
Wild, B. J.; Green, B. N.; Cooper, E. K.; Lalloz, M. R. A.; Erten, S.; Stephens, A. D.; Layton, D. M. Rapid identification of hemoglobin variants by electrospray ionization mass spectrometry. Blood Cells Mol. Dis. 2001, 27, 691–704.
Rai, D. K.; Griffiths, W. J.; Landin, B.; Wild, B. J.; Alvelius, G.; Green, B. N. Accurate mass measurement by electrospray ionization quadrupole mass spectrometry: Detection of variants differing by <6 Da from normal in human hemoglobin heterozygotes. Anal. Chem. 2003, 75, 1978–1982.
Bateman, R. H.; Green, B. N.; Morris, M. Electrospray ionization mass spectrometric analysis of the globin chains in hemoglobin heterozygotes can detect the variants HbC, D, and E. Clin. Chem. 2008, 54, 1256–1257.
Roberts, N. B.; Green, B. N.; Morris, M. Potential of electrospray mass spectrometry for quantifying glycohemoglobin. Clin. Chem. 1997, 43, 771–778.
Roberts, N. B.; Amara, A. B.; Morris, M.; Green, B. N. Long-term evaluation of electrospray ionization mass spectrometric analysis of glycated hemoglobin. Clin. Chem. 2001, 47, 316–321.
Williams, J. P.; Jackson, H.; Green, B. N. Hb Belleville [β10(A7)Ala→Thr] affects the determination of Hb A1C by routine cation exchange high performance liquid chromatography. Hemoglobin 2009, 33, 45–50.
Williams, J. P.; Scrivens, J. H.; Green, B. N.; Farrar, L. M.; Sutcliffe, M. Hb Leeds [β56(D7)Gly→Cys]: A new hemoglobin that aggravates anemia in a child with β 0-thalassemia trait. Hemoglobin 2007, 31, 367–373.
Roepstorff, P.; Fohlman, J. Proposal for a common nomenclature for sequence ions in mass spectra of peptides. Biol. Mass Spectrom. 1984, 11, 601.
Kjeldsen, F.; Haselmann, K. F.; Budnik, B. A.; Jensen, F.; Zubarev, R. A. Dissociative capture of hot (3–13 eV electrons by polypeptide polycations: An efficient process accompanied by secondary fragmentation. Chem. Phys. Lett. 2002, 356, 201–206.
Cooper, H. J.; Hakansson, K.; Marshall, A. G. The role of electron capture dissociation in biomolecular analysis. Mass Spectrom. Rev. 2005, 24, 201–222.
Zubarev, R. A.; Kelleher, N. L.; McLafferty, F. W. ECD of multiply charged protein cations: A nonergodic process. J. Am. Chem. Soc. 1998, 120, 3265–3266.
Kjeldsen, F.; Haselmann, K. F.; Sorensen, E. S.; Zubarev, R. A. Distinguishing of Ile/Leu amino acid residues in the PP3 protein by (hot) electron capture dissociation in Fourier transform ion cyclotron resonance mass spectrometry. Anal. Chem. 2003, 75, 1267–1274.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published online May 18, 2009
Rights and permissions
About this article
Cite this article
Williams, J.P., Creese, A.J., Roper, D.R. et al. Hot electron capture dissociation distinguishes leucine from isoleucine in a novel hemoglobin variant, Hb Askew, β54(D5)Val→Ile. J Am Soc Mass Spectrom 20, 1707–1713 (2009). https://doi.org/10.1016/j.jasms.2009.05.002
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2009.05.002